Winter G, Gildea RJ, Paterson NG, Beale J, Gerstel M, Axford D, Vollmar M, McAuley KE, Owen RL, Flaig R, Ashton AW, Hall DR
How best to use photons
Acta Crystallogr D Struct Biol. (2019) Mar 1;75(Pt 3):242-261. doi: 10.1107/S2059798319003528
Dittrich, B., Lübben, J., Mebs, S., Wagner, A., Luger, P., Flaig, R.
Accurate Bond Lengths to Hydrogen Atoms from Single-Crystal X-ray Diffraction by Including Estimated Hydrogen ADPs and Comparison to Neutron and QM/MM Benchmarks.
Chemistry – a European Journal 23(19), 4605-4614 (2017)
Woińska M, Jayatilaka D, Dittrich B, Flaig R, Luger P, Woźniak K, Dominiak PM, Grabowsky S
Validation of X-ray wavefunction refinement
Chemphyschem. (2017) Dec 6;18(23):3334-3351. doi: 10.1002/cphc.201700810
Robaa, D., Wagner, T., Luise, C., Carlino, L., McMillan, J., Flaig, R., Schüle, R., Jung, M., Sippl, W.
Identification and Structure-Activity Relationship Studies of Small-Molecule Inhibitors of the Methyllysine Reader Protein Spindlin1
ChemMedChem, 11, 2327-2338 (2016)
doi: 10.1002/cmdc.201600362
Roatsch, M., Robaa, D., Pippel, M., Nettleship, JE., Reddivari, Y., Bird, LE., Hoffmann, I., Franz, H., Owens, RJ., Schüle, R., Flaig, R., Sippl, W., & Jung, M.
Substituted 2-(2-aminopyrimidin-4-yl)pyridine-4-carboxylates as potent inhibitors of JumonjiC domain-containing histone demethylases
Future Medicinal Chemistry, 8, 1553-1571 (2016)
doi: 10.4155/fmc.15.188
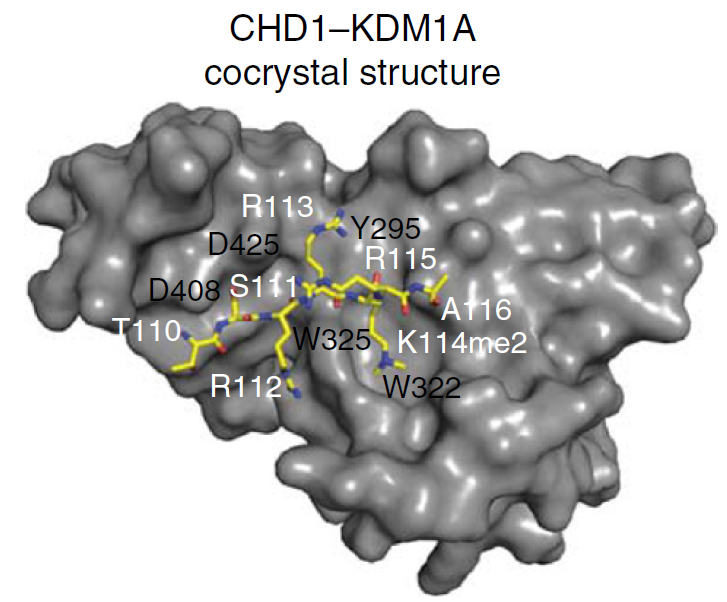
Metzger, E., Willmann, D., McMillan, J., Forne, I., Metzger, P., Gerhardt, S., Petroll, K., von Maessenhausen, A., Urban, S., Schott, A-K., Espejo, A., Eberlin,A., Wohlwend, D., Schüle, KM., Schleicher, M., Perner, S., Bedford,MT., Jung, M., Dengjel, J., Flaig, R., Imhof, A.,Einsle, O. & Schüle, R.
Assembly of methylated KDM1A and CHD1 drives androgen receptor–dependent transcription and translocation
Nature Structural & Molecular Biology, 23, 132–139 (2016)
doi: 10.1038/nsmb.3153
Aller, P., Sanchez-Weatherby, J., Foadi, J., Winter, G., Lobley, C.M., Axford, D., Ashton, A.W., Bellini, D., Brandao-Neto, J., Culurgioni, S., Douangamath, A., Duman, R., Evans, G., Fisher, S., Flaig, R., Hall, D.R., Lukacik, P., Mazzorana, M., McAuley, K.E., Mykhaylyk, V., Owen, R.L., Paterson, N.G., Romano, P., Sandy, J., Sorensen, T., von Delft, F., Wagner, A., Warren, A., Williams, M., Stuart, D.I., Walsh, M.A.
Application of in situ diffraction in high-throughput structure determination platforms
Methods Mol Biol., 1261, 233-53 (2015). doi: 10.1007/978-1-4939-2230-7_13
Duke, E.H.M., Evans, G., Flaig, R., Hall, D.R., Latchem, M., McAuley, K.E., Sandy, D.J., Sorensen, T.L-M., Waterman, D., Johnson, L.N.
The Phase I MX Beam lines at Diamond Light Source.
AIP Conference Proceedings 1234, 165-168 (2010)
Drew, D., Klebsch, M.M., Newstead, S., Flaig, R., De Gier, J-W., Iwata, S., Beis, K.
The structure of the efflux pump AcrB in complex with bile acid.
Molecular Membrane Biology 25 677-682 (2008)
doi: 10.1080/09687680802552257
Greschik, H., Althage, M., Flaig, R., Sato, Y., Chavant, V., Peluso-Iltis, C., Choulier, L., Cronet, P., Rochel, N., Schüle, R., Strömstedt, P-E., Moras, D.
Communication between the ERR-alpha homodimer interface and the PGC-1-alpha
binding surface via the helix 8-9 loop.
J. Biol. Chem. 283 20220-20230 (2008)
Sorensen, T.L-M., McAuley, K.E., Flaig, R., Duke, E.H.M.
New light for science: synchrotron radiation in structural medicine.
Trends in Biotechnology 24(11) 500-508 (2006)
doi:10.1016/j.tibtech.2006.09.006
Flaig, R., Greschik, H., Peluso-Iltis, C., Moras, D.
Structural Basis for the Cell-specific Activities of the NGFI-B and the Nurr1 Ligand-binding Domain.
J. Biol. Chem. 280 19250-19258 (2005)
doi: 10.1074/jbc.M413175200
Kammerer, S., Germain, P., Flaig, R., Peluso-Iltis, C., Mitschler, A., Rochel, N., Gronemeyer, H., Moras, D.
RAR-beta ligand-binding domain bound to an SRC-1 co-activator peptide: purification, crystallization and preliminary X-ray diffraction analysis.
Acta Crystallogr. D60 2048-2050 (2004)
Greschik, H., Flaig, R., Renaud, J-P., Moras, D.
Structural basis for the Deactivation of the Estrogen-related Receptor gamma by Diethylstilbestrol or 4-Hydroxytamoxifen and Determinants of Selectivity
J. Biol. Chem. 279, 33639-33646 (2004)
Wagner, A., Flaig, R., Dittrich, B., Schmidt, H., Koritsanszky, T., Luger, P.
Charge Density and Electrostatic Potentials of two Penicillin Derivatives
Chem. Eur. J. 10, 2977-2982 (2004)
Wagner, A., Flaig, R., Zobel, D., Dittrich, B., Bombicz, P., Strumpel, M., Luger, P., Koritsanszky, T., Krane, H.G.
Structure and Charge Density of a C60-Fullerene Derivative Based on a High Resolution Synchrotron Diffraction Experiment at 100 K
J. Phys. Chem. A106, 6581-6590 (2002)
Kingsford-Adaboh, R., Dittrich, B., Wagner, A., Messerschmidt, M., Flaig, R., Luger, P.
Topological Analysis of D,L-Argininine Monohydrate at 100K
Z. Kristallogr. 217, 168-173 (2002)
Dittrich, B., Koritsanszky, T., Grosche, M., Scherer, W., Flaig, R., Wagner, A., Krane, H.G., Kessler, H., Riemer, C., Schreurs, A.M.M., Luger, P.
Reproducibility and Transferability of Topological Properties: Experimental Charge Density of the Hexapeptide cyclo-(D,L-Pro)2-(L-Ala)4 Monohydrate
Acta Crystallogr. B58, 721-727 (2002)
Flaig, R., Koritsanszky, T., Dittrich, B., Wagner, A., Luger, P.
Intra and Intermolecular Topological Properties of Amino Acids - A Comparative Study of Experimental and Theoretical Results
J. Am. Chem. Soc. 124, 3407-3417 (2002)
Flaig, R., Koritsanszky, T., Soyka, R., Häming, L., Luger, P.
Electronic Insight into an AntiThrombotic Agent by High-resolution X-ray Crystallography
Einblick in die elektronische Struktur eines antithrombotischen Wirkstoffs durch hochaufgelöste Röntgenbeugung
Angew. Chem. Int. Ed. 40, 355-359 (2001), Angew. Chem. 113, 368-371 (2001)
Dittrich, B., Flaig, R., Koritsanszky, T., Krane, H.G., Morgenroth, W., Luger, P.
Topological Properties of the Peptide Bond in Glycyl-L-threonine Dihydrate Based on a Fast Synchrotron/CCD-Diffraction Experiment at 100K
Chem. Eur. J. 6, 2582-2589 (2000)
Flaig, R., Koritsanszky, T., Janczak, J., Krane, H.G., Morgenroth, W., Luger, P.
Fast Experiments for Charge Density Determination: Topological Analysis and Electrostatic Potential of the Amino Acids L-Asn, DL-Glu, DL-Ser, and L-Thr
Schnelle Experimente zur Ladungsdichtebestimmung: topologische Analyse und elektrostatisches Potential der Aminosäuren L-Asn, DL-Glu, DL-Ser und L-Thr
Angew. Chem. Int. Ed. 38, 1397-1400 (1999), Angew. Chem. 111, 1494-1497 (1999)
Flaig, R., Koritsanszky, T., Zobel, D., Luger, P.
Topological Analysis of the Experimental Electron Densities of Amino Acids. 1. DL-Aspartic Acid at 20K
J. Am. Chem. Soc. 120, 2227-2238 (1998)
Koritsanszky, T., Flaig, R., Zobel, D., Krane, H.G., Morgenroth, W., Luger, P.
Accurate Experimental Electronic Properties of DL-Proline Monohydrate Obtained Within 1 Day
Science 279, 356-358 (1998)
Dissertation/Thesis
Neue experimentelle Methoden der Ladungsdichtebestimmung - New experimental methods of charge density determination, FU Berlin (Germany) (2000)