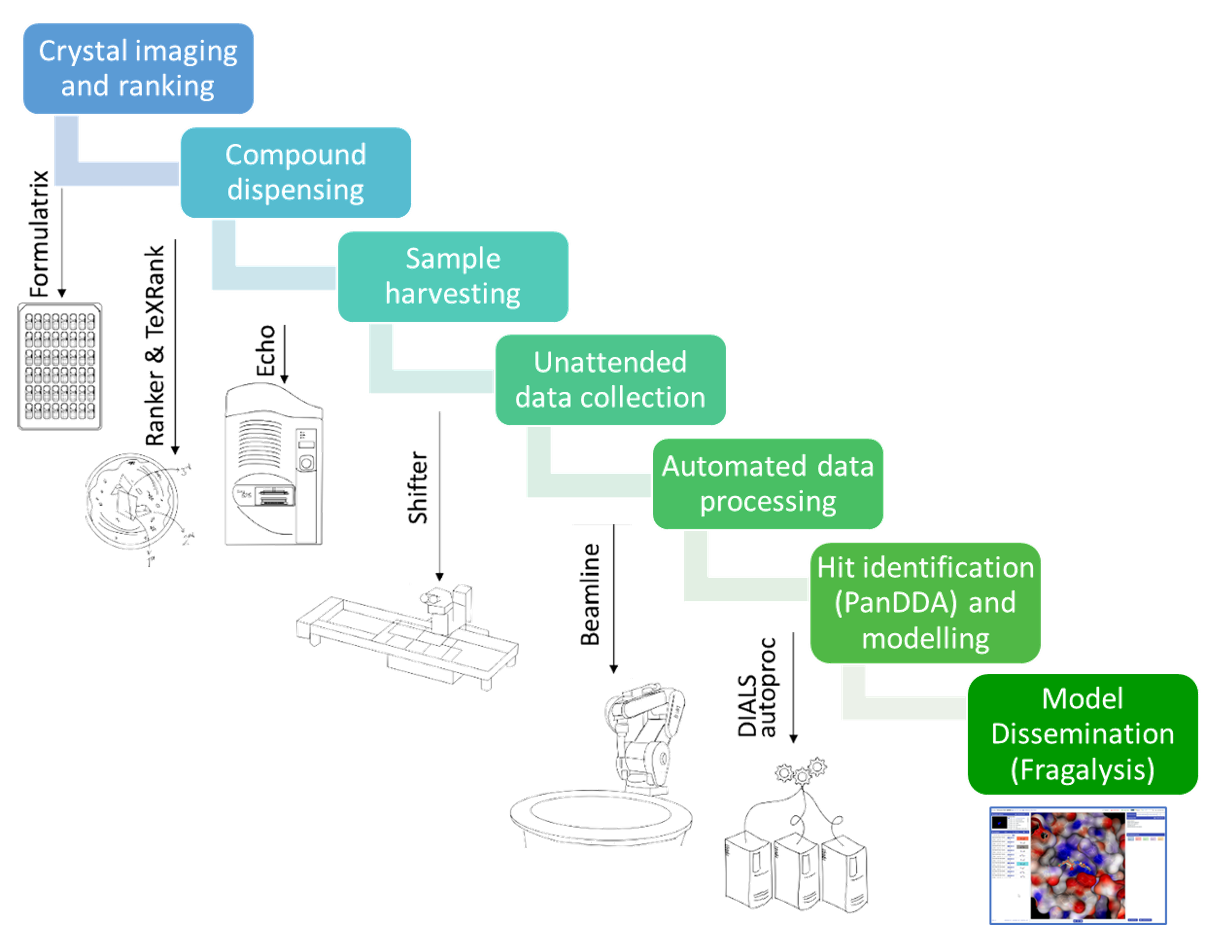
Instruments by Science Group
XChem Key Contacts
Principal Beamline Scientist:
Frank von Delft
Tel: +44 (0) 1235 778997
E-mail: [email protected]
Senior Beamline Scientist:
Blake Balcomb
Tel: +44 (0) 1235 778394
E-mail: [email protected]
Science Group Leader
Dave Hall
Email: [email protected]
Tel: +44 (0) 1235 778926
Table of Contents
Crystal Plate Imaging in the Research Complex at Harwell

Images of each individual crystal droplet are recorded using Formulatrix imagers in the Research Complex at Harwell on the RAL site.
Prior to your visit you will need to request acces to RCaH through your local contact.
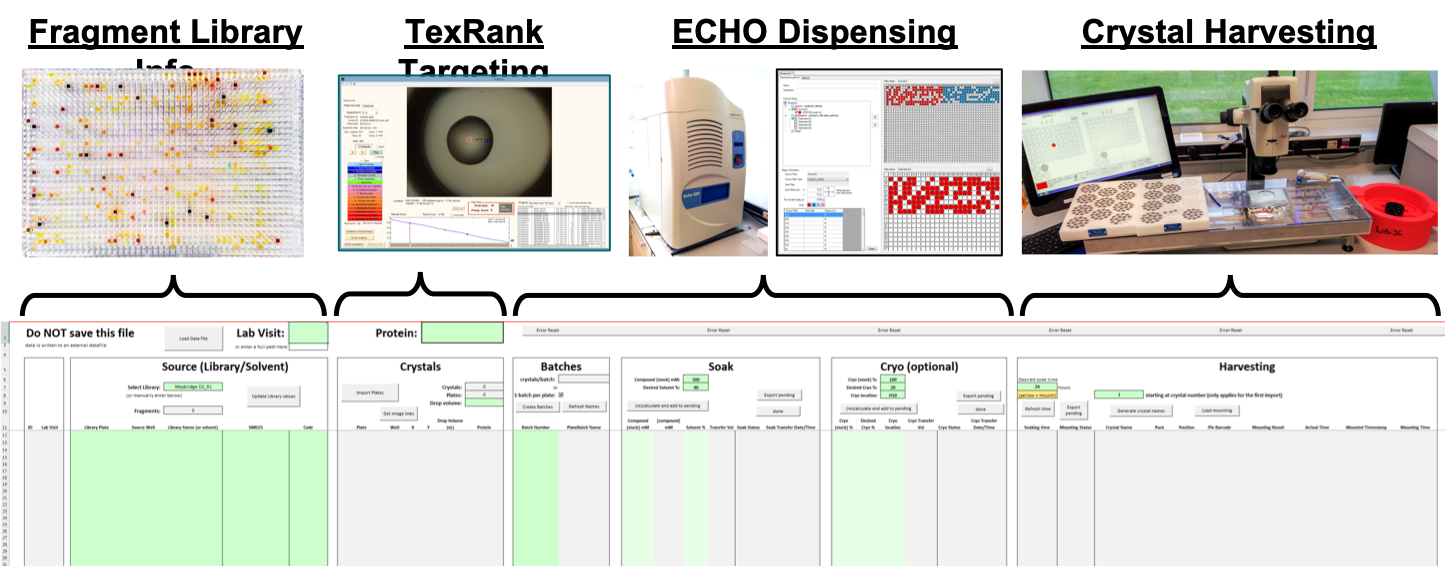
TeXRank: Crystal Targeting
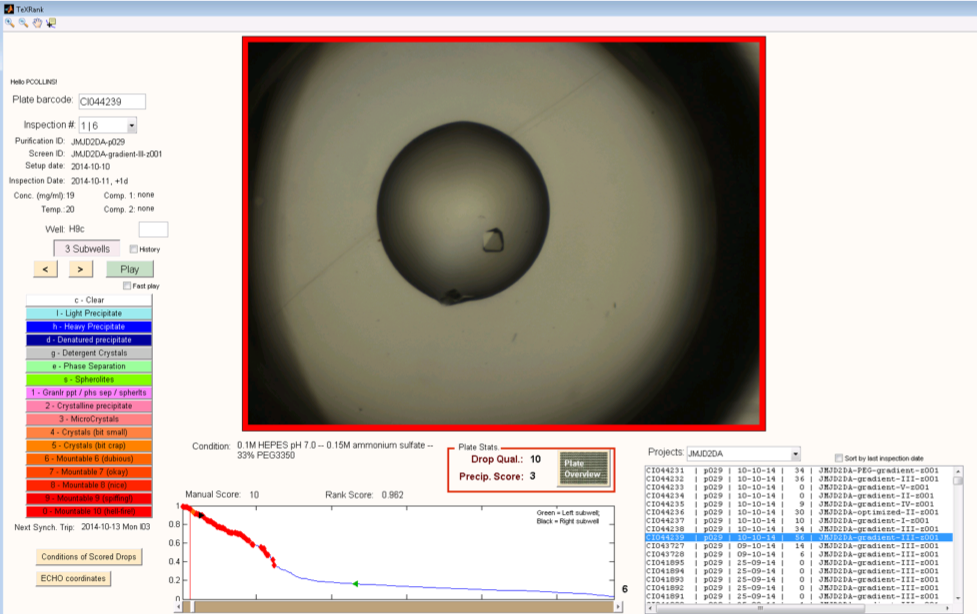
The TeXRank program takes the crystal droplet images and places them in an inspection queue with droplets that are likely to contain crystals placed at the top of the queue to be viewed first. The target location for the dispensed compound solution is done by simply right-clicking on the droplet image at the desired location and the co-ordinates are stored. Using this method, targeting a 3-droplet plate takes a matter of minutes.
Our initial anecdotal evidence is that shooting directly at the crystal will damage it, but we are evaluating various different targeting protocols.
Where do I get it: TeXRank is fully open source and available from GitHub here. If you want it deployed with your own imagers, it may require a bit of work to get everything integrated - it's do-able but a lot of faff, so if you have money but no time, contact Frank von Delft who can put you in touch with someone that can help.
ECHO Liquid Handler: Solvent, Fragment and Cryoprotectant Dispensing

Compounds are transferred to the crystals using an ECHO (Labcyte) acoustic liquid handler. The crystal plate is inverted over the plate with the compounds, and the ECHO uses ultrasonic pulses to shoot series of 2.5 nL droplets of solution at the crystal drops, as many as required to make up the required dispense volume. The dispense coordinates are uploaded from the TeXRank output. Dispensing is very fast: ~30sec for 100s of compounds.
If your system need cryoprotectant we can also dispense that into your droplets prior to the addition of the fragments. A list of the cryoptectants we have in-house is available to download here. Alternatively, you can bring your own cryoprotectant in a tube (it will be transferred to an 384 well ECHO-compatible plate for dispensing on site).
Where do I get one: ECHO liquid handlers are available from Labcyte
This guide details how to setup SwissCI-3drop plate definitions on the ECHO plate reformat software.
Incubation

Once fragment libraries have been dispensed the crystal plates are incubated to allow the compound solution to diffuse through the droplet and soak into the crystal.
Soaking incubation times are dictated by how well the crystals survive at a given solvent concentration; one hour is probably sufficient, but it's often convenient to do longer - systems that can't tolerate more than a few seconds needs special treatment.
The OLT Shifter: Crystal Harvesting

Crystals are harvested manually, but the pain of doing this for 100s of crystals has been greatly reduced by the Shifter, a microscope x-y stage that also handles unsealing and resealing as well as automatically tracking samples. This trick is surprisingly effective, and typically >100 crystals/hour can be harvested. (Current record: 200 xtals per hour, sustained.)

Where do I get one: The Shifter is now commercially available from Oxford Lab Technologies. It is ridiculously cheap (seriously), fits on your favourite stereo microscope, and you can drive it with CSV files.
Unitrays, wands and pucks

We have a well-curated collection of loops, with tools that allow them to be cleaned both rapidly and with minimal damage. This certainly speeds up harvesting, but more importantly, it makes the loop centring algorithm on the beamline very reliable indeed (>97%).
Where do I get them: The Uniwand and Unitray are now official Mitegen products, and we highly recommend it for any lab setting.
The Watershed

If your crystal droplets dehydrate, precipitate forms or protein is unstable during the harvesting process as a results of dehydration we can offer use of the WatershedTM to help control humidity levels during harvesting.
Where do I get one: The Watershed is an official Mitegen product.
Barcode Reader: Sample Information Transfer
Once your crystals have been harvested the sample information is transferred to iSPyB following scanning using a puck and pin barcode reader.
The software connects to a camera which is positioned on a 3D printed mount to allow quick alignment of the puck in the cameras field of view. The barcodes on the pins and side of the pucks are read and transferred to a clipboard at their coresponding locations in the puck. This information is then transferred to iSPyB so you don't have to manually input sample IDs for hundreds of samples.
Where do I get one: Executables can be downloaded from GitHub, here.
3D print files for the stand can be downloaded here.
Some instructions can be found here.
We recommend using it with any camera with manual focus.
SoakDB: Experiment Manager
SoakDB is a LIMS-in-a-box for the entire soaking and harvesting experiment. From here, the experiment is designed, the fragments are tracked, the ouptut of the various programs and machines gathered into one place and the input files for others are created. It's an Excel interface with an SQLite database file SoakDB; with permissions being dealt with by the file-system, the db file can be emailed around, and some wicked VBA magic means the data persists only in the db file: Excel is essentially just the database UI.
Where do I get it? You don't need to download it for the XChem experiment but if you want to use it at home it is available for to download here. There are reports that it does work outside the XChem ecosystem, but you may need to contact with questions. It's very powerful but it isn't a bells-and-whistles user interface and comes with no guarantees you'd know how to use it without training.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.