
Electron Microscope Scientist at the electron Bio-Imaging Centre (eBIC)
Weng joined eBIC in September 2023.
Email: [email protected]
Electron Microscope Scientist at the electron Bio-Imaging Centre (eBIC)
Weng joined eBIC in September 2023.
Email: [email protected]Weng obtained his DPhil from the University of Oxford (2021) in the laboratories of Professor Juha Huiskonen and Professor Thomas Bowden, where he used X-ray crystallography and cryogenic electron microscopy to characterise the structures, functions, and antibody-mediated neutralisations of arenaviral glycoproteins.
Following his doctoral studies, Weng moved to The Jenner Institute as a Postdoctoral Researcher in Structural Vaccinology in Dr Sandy Douglas’s laboratory where he studied rabies virus. During his time at the Institute, Weng contributed to determining the first high-resolution structure of a lyssavirus glycoprotein, revealing the neutralisation mechanism of a licensed protective antibody, and identifying key mutations that lock the therapeutic target in pre-fusion conformation for immunogen design.
In April 2023, Weng was awarded the Wellcome Early-Career Award (2023-2028) to initiate his own research. He joined eBIC in September 2023 as an Electron Microscope Scientist, concurrently starting an independent group at Research Complex at Harwell (RCaH). Weng’s research focuses on the structural and functional characterisation of viral glycoproteins through the application of structural biology, immunology, and cell biology to understand the pathobiology of enveloped RNA viruses and inform the rational development of therapeutics and vaccines.
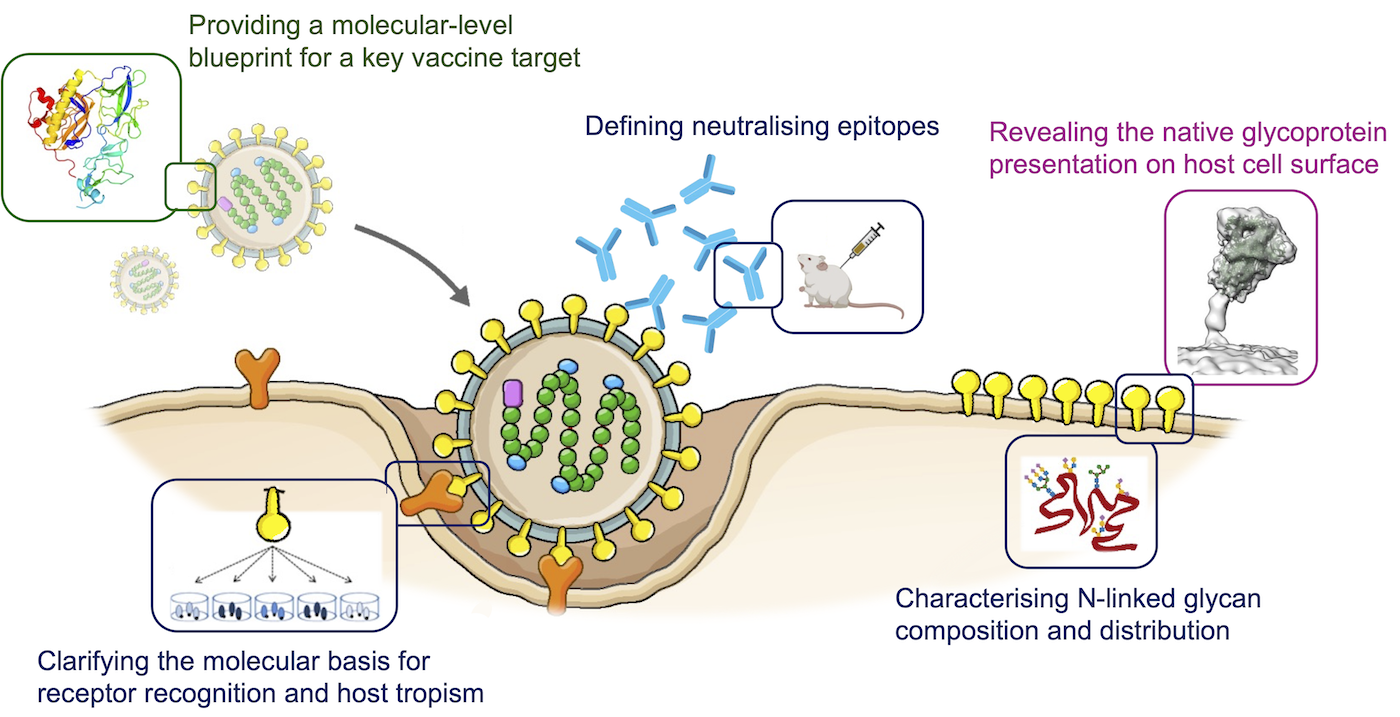
Dr Weng Ng’s research employs integrative and complementary structural and functional methodologies to provide a comprehensive understanding of viral glycoprotein native architectures and functionalities.
1. High-resolution structures of mature, oligomeric viral glycoproteins
Weng’s research focuses on the glycoproteins presented by enveloped RNA viruses (e.g., bornavirus and lyssavirus). Through a combination of structural biology techniques (crystallography and cryo-EM) and computational approaches, Weng aims to achieve atomistic understanding of the roles played by viral glycoproteins during the host cell entry process and provide a molecular-level blueprint for key vaccine targets.
2. Virus-host interactions
Often serving as the sole surface antigen, viral glycoproteins are responsible for receptor recognition and membrane fusion, and constitute the primary target of the host immune response. Weng’s research aims to elucidate the specificity underlying the host cell entry process utilised by bornaviruses and lyssaviruses and to develop monoclonal antibody-based prophylaxis.
3. Native conformation and glycan profiling
Vaccine development focusing on using viral surface glycoproteins as immunogens necessitates correct protein presentation to elicit a robust immune response. By utilising mass spectrometry and cryo-electron tomography, Weng’s research emphasises the significance of viral glycobiology as a critical parameter during vaccine design and examines the native glycoprotein conformation induced by various vaccine constructs and delivery strategies.
(# indicates first, co-first, or corresponding author)
Ng, W.M.#, Fedosyuk, S., English, S., Augusto, G., Berg, A., Thorley, L., Segireddy, R.R., Bowden, T.A., Douglas, A.D. (2022). Structure of trimeric pre-fusion rabies virus glycoprotein complex with two protective antibodies. Cell Host Microbe. 30(9):1219-1230.e7.
Ng, W.M.#, Sahin, M., Krumm, S.A., Seow, J., Zeltina, A., Harlos, K., Paesen, G., Pinschewer, D.D., Doores, K.J., Bowden, T.A. (2022). Contrasting modes of New World arenavirus neutralization by infection-elicited monoclonal antibodies. mBio. e02650-21.
Graham, C., Seow, J., Huettner, I., Khan, H., Kouphou, N., Acors, S., Winstone, H., Pickering, S., Galao, R.P., Dupont, L., Lista, M.J., Jimenez-Guardeño, J.M., Laing, A.G., Wu, Y., Joseph, M., Muir, L., van Gils, M.J., Ng, W.M., Duyvesteyn, H.M.E., Zhao, Y., Bowden, T.A., Shankar-Hari, M., Rosa, A., Cherepanov, P., McCoy, L.E., Hayday, A.C., Neil, S.J.D., Malim, M.H., Doores, K.J.. Neutralization potency of monoclonal antibodies recognizing dominant and subdominant epitopes on SARS-CoV-2 Spike is impacted by the B.1.1.7 variant. (2021). Immunity. 54(6):1276–1289.e6.
Ng, W.M.#, Stelfox, A. J., & Bowden, T. A. (2020). Unraveling virus relationships by structure-based phylogenetic classification. Virus Evol. 6(1):veaa003.
Stass, R., Ng, W.M.#, Kim, Y.C., Huiskonen, J.T. (2019). Structures of enveloped virions determined by cryogenic electron microscopy and tomography. Adv Virus Res. 105:35-71.
Pryce, R., Ng, W.M.#, Zeltina, A., Watanabe, Y., El Omari, K., Wagner, A., Bowden, T.A. (2018). Structure-based classification defines the discrete conformational classes adopted by the arenaviral GP1. J Virol. 93(1):e01048-18.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.