26 November 2014
I20-1 is now I20-scanning
I20-2 is now I20-EDE
Please reference the beamline and the branchlines with these new names in your publications.
X-ray emission spectroscopy (XES) is one of the so-called photon-in - photon-out spectroscopies in which a core electron is excited by an incident x-ray photon and then this excited state decays by emitting an x-ray photon to fill the core hole. The energy of the emitted photon is the energy difference between the involved electronic levels. The analysis of the energy dependence of the emitted photons is the aim of the X-ray emission spectroscopy.
There is a list of available analyser crystals and what emission lines they can be used for available here.
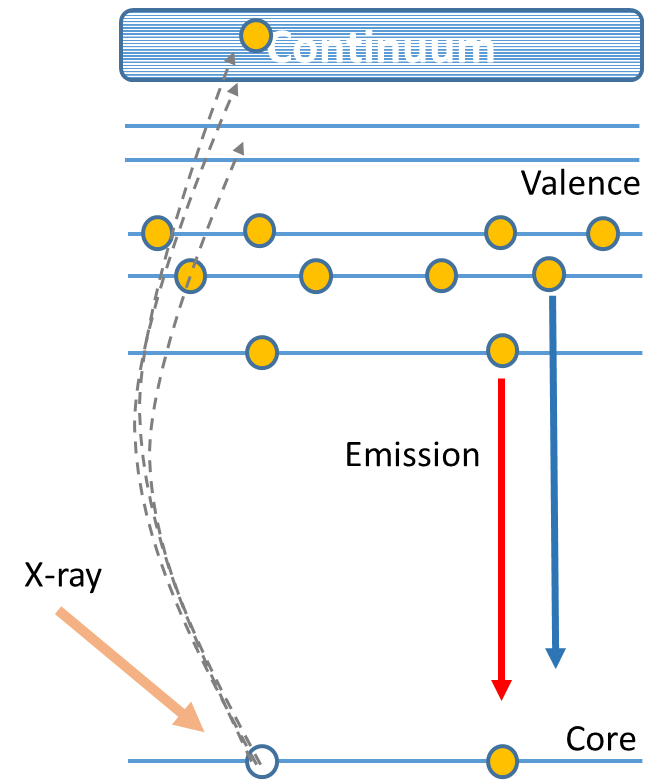

XES is a technique complementary to X-ray absorption spectroscopy (XAS) that provides valuable information with respect to the electronic structure (local charge- and spin-density) as well as the nature of the bound ligands.
In particular, the high-resolution fluorescence detection technique, makes possible to overcome some of the main limitations of conventional XAS. This technique consists of measuring the x-ray absorption spectrum via monitoring the intensity of a fluorescence line corresponding to a specific excited state decay process using a narrow energy resolution. This is generally achieved through the use of a crystal analyzer to select a narrow energy band from the sample’s emission line. By using this technique it is possible to overcome some of the main limitations of conventional X-ray absorption spectroscopy:
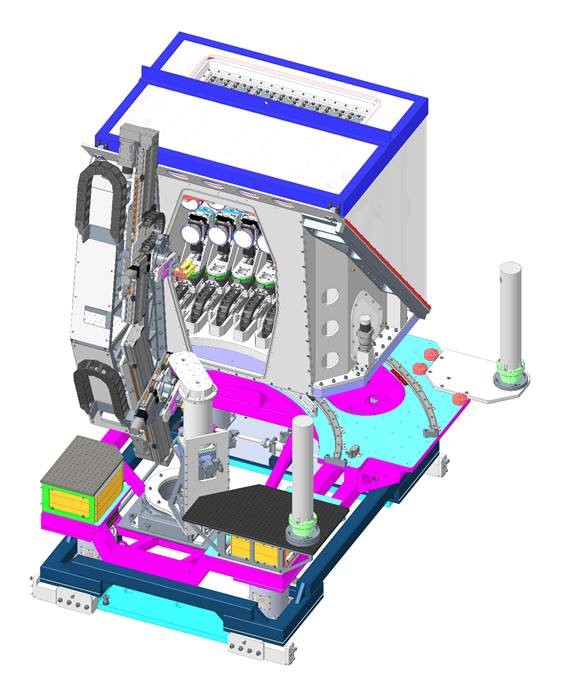
There is now a new 14 crystal spectrometer installed on the beamline. There are two separate rows which can be fitted with the same crystal type to measure one emission line or different crystal types to measure two different lines. However due to the time required to change/align each row it is not possible to change these crystals during an experimental visit
An example of in-situ experiment performed with the old I20 XES spectrometer using the in-house Plug Flow Reactor below. The spectra were measured in a Kapton tube (1.62mmOD; 80mm wall-thickness) from a SiO2 aerogel loaded with 5 wt% CeO2. For this experiment, the XES spectrometer was equipped with three Si(400) analyzers and the Si-drift detector to measure the Ce L-line, 4839eV.

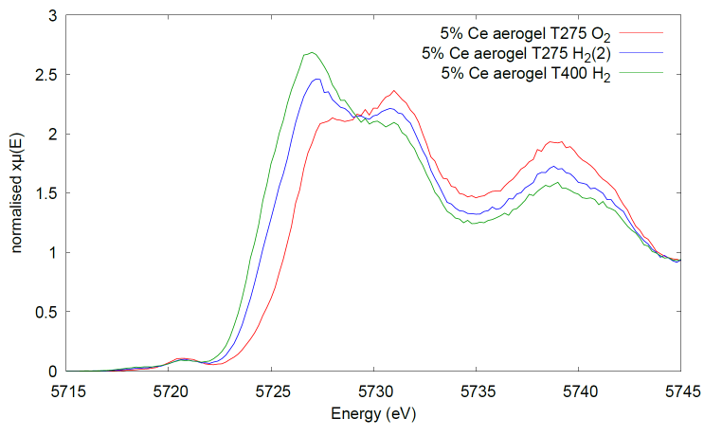
Figure showing the HR-XANES spectra collected at 275C and then at 400C after the reduction treatment (Anna Corrias et al., University of Kent).
[1] K. Hamalainen, D. P. Siddons, J. B. Hastings, and L. E. Berman, Phys. Rev. Lett., 67, 2850 (1991).
[2] (a) F.M.F de Groot, M. H. Krisch and J. Vogel, Topics in Catalysis, 10, 179 (2000). (b) J. van Bokhoven, C. Louis, J. Miller, M. Tromp, O. Safonova and P. Glatzel, Angewandte Chemie-International Edition, 45, 4651 (2006).
[3] (a) K. Hamalainen, C. C. Kao, B. Hastings, D. P. Siddons, L.E. Berman, V. Stojanoff, S. P. Cramer, Phys. Rev. B, 46, 14274 (1992). (b) Q. Qian, T. A. Tyson, S. Savrassov, C. C. Kao, and M. Croft, Phys. Rev. B, 68, 014429 (2003).
[4] P. Glatzel, L. Jacquamet, U. Bergmann, F.M.F. de Groot, S.P. Cramer, Inorg. Chem., 41, 3121 (2002).
[5] P. Glatzel, F.M.F. de Groot, O. Manoilova, D. Grandjean, B.M. Weckhuysen, U. Bergmann and R. Barrea, Phys. Rev. B, 72, 014117 (2005).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.