
Want to learn more about one of science's heroes from history, Henry Moseley? Moseley solved one of chemistry's greatest puzzles - determining what distinguishes elements from one another and developed a means of identifying elements based on their atomic characteristics. Sadly he lost his life fighting at Gallipoli in WWI.
Learn more about his life and legacy by watching our online film here.
The name "halogen" means "salt-producing", and halogens react with metals to produce a range of salts, including calcium fluoride, sodium chloride, silver bromide and potassium iodide. Elemental halogens are highly reactive and toxic, and chlorine, bromine and iodine, are often used as disinfectants. Organobromides are organic compounds containing carbon bonded to bromine, and are the most important class of flame retardants. Organofluorines are organic compounds that contain the carbon–fluorine bond, and have a wide range of applications, including oil and water repellents to pharmaceuticals, refrigerants, and reagents in catalysis. Some organofluorines (such as CFCs) are environmental pollutants, contributing to ozone depletion, global warming, and toxicity.
 Chlorine is a yellow-green gas at room temperature, and is named from the Ancient Greek khlôros, meaning 'pale green'. It is a highly reactive and oxidising element, and all of the chlorine in the Earth's crust is in the form of ionic chloride compounds. The most common of these, sodium chloride (common salt), has been known since ancient times. First synthesised in 1630, chlorine wasn't recognised as an element until 1810.
Chlorine is a yellow-green gas at room temperature, and is named from the Ancient Greek khlôros, meaning 'pale green'. It is a highly reactive and oxidising element, and all of the chlorine in the Earth's crust is in the form of ionic chloride compounds. The most common of these, sodium chloride (common salt), has been known since ancient times. First synthesised in 1630, chlorine wasn't recognised as an element until 1810.
Elemental chlorine is commercially produced from saltwater by electrolysis, and it is used in bleaches and disinfectants and for many processes in the chemical industry. It also has the dubious honour of being the first gaseous chemical warfare agent, used in World War I. Despite its toxicity, chloride ions are necessary for life.

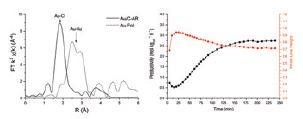
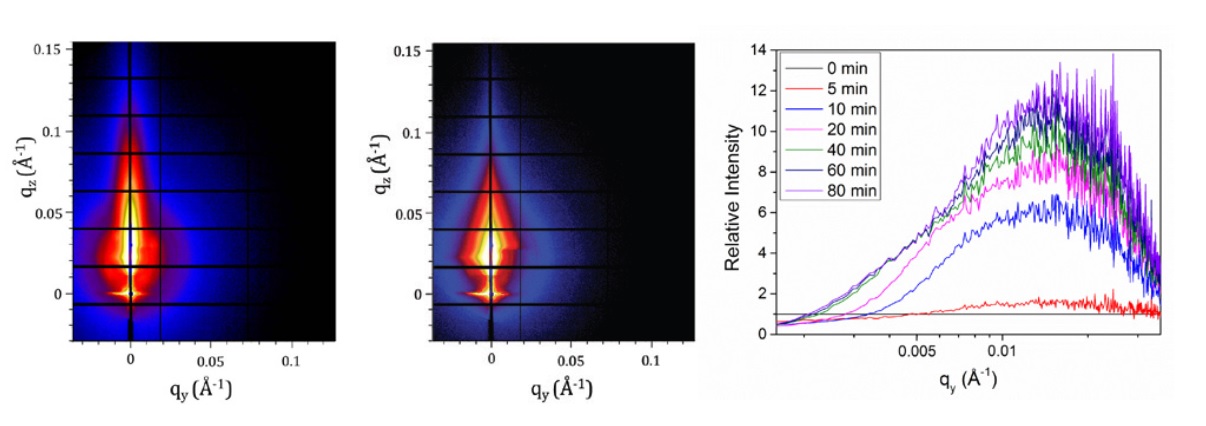
 The same man who added lead to petrol, Thomas Midgley Jr., also pioneered the use of dichlorodifluoromethane, a chlorofluorocarbon (CFC), in residential refrigerators and air conditioners. His company manufactured the gas under the brand name Freon-12. Midgley was fond of demonstrating that his compounds were 'safe', and he inhaled a large amount of Freon-12 and then blew out a candle flame to show the gas was non-toxic and non-flammable.
The same man who added lead to petrol, Thomas Midgley Jr., also pioneered the use of dichlorodifluoromethane, a chlorofluorocarbon (CFC), in residential refrigerators and air conditioners. His company manufactured the gas under the brand name Freon-12. Midgley was fond of demonstrating that his compounds were 'safe', and he inhaled a large amount of Freon-12 and then blew out a candle flame to show the gas was non-toxic and non-flammable.Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.