Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Current production process for vinyl chloride, the monomer used in PVC and everything from piping and tubing to gels and lubricants, relies on a mercury-based catalyst. Mercury is one of the most toxic substances on the planet, and the UN recently ratified the Minamata Convention, meaning that a more environmentally-friendly alternative is required.
The production of vinyl chloride monomer (VCM) as the precursor to polyvinyl chloride (PVC) is a major industrial process. In particular, 42 million tonnes of PVC had being consumed in 2016, constituting 16% of total plastic demand1. The production of VCM through acetylene hydrochlorination (the reaction of acetylene with HCl) using a mercuric chloride based catalyst is the dominant method of VCM production in coal rich areas such as China. However, the catalyst losses significant amounts of mercury, posing a great risk to the environment. The recently ratified Minamata convention has outlined that VCM production must move to mercury free production by 2022, necessitating the development of a new commercial catalyst for VCM production.
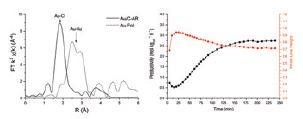
To gain a greater understanding of the active form of the catalyst under operating conditions, a reactor system was designed in consultation with beamline B18. The designed reactor allowed the research team to simultaneously acquire X-ray absorption spectra, at the Au L3 absorption edge, while carrying out an acetylene hydrochlorination reaction at realistic reaction conditions with product analysis by mass spectrometry (Figure 1). This allowed for the correlation of the performance of the catalyst with Au oxidations state and co-ordination environment as determined by the X-ray absorption spectra. Reactions were carried out at 200 °C with 100 mg of 1% Au/C catalyst prepared using the aqua regia impregnation method and 50 ml min-1 of gas flow containing acetylene (C2H2) (2.35 vol%), Hydrogen Chloride (HCl) (2.4vol%) and Argon (Ar) as carrier gas. Using the quick acquisition time available and the x-y movable stage it was possible to record XAS spectra at three points through the catalyst bed, every three minutes during the extended reaction.

This study provided the evidence that the isolated cationic Au species present in the catalyst were in fact the active species and damage and photoreduction of the catalyst materials through ex situ characterisation masked and confused the interpretation of the active form of the catalyst in previous studies. By characterising the catalyst during the reaction, the research team were able to conclusively show that the active catalyst for this reaction is isolated cationic Au sites akin to homogenous catalysts. This provides an opportunity to design the ligand environments around the Au centre to improve catalyst activity and stability which could result in Au becoming the replacement for the mercury based catalysts for VCM manufacture. In fact Au/C catalysts have been successfully operated at a commercial scale and look set to lead the reduction in mercury usage in this large scale industrial process5.
Malta G, Kondrat SA, Freakley SJ, Davies CJ, Lu L, Dawson S, Thetford A, Gibson EK, Morgan DJ, Jones W, Wells PP, Johnston P, Catlow CRA, Kiely CJ, Hutchings GJ. Identification of single-site gold catalysis in acetylene hydrochlorination. Science 355(6332), 1399-1403, doi: 10.1126/science.aal3439 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.