
Want to learn more about one of science's heroes from history, Henry Moseley? Moseley solved one of chemistry's greatest puzzles - determining what distinguishes elements from one another and developed a means of identifying elements based on their atomic characteristics. Sadly he lost his life fighting at Gallipoli in WWI.
Learn more about his life and legacy by watching our online film here.
The alkali metals - lithium, sodium, potassium, rubidium, caesium and francium -- are all shiny, soft and highly reactive. They’re soft enough to cut with a knife, and freshly cut surfaces tarnish rapidly in the air. They have to be stored under soil, and are never found naturally as free elements. They all react with water, with the heavier alkali metals reacting more vigorously.
Atomic clocks using rubidium or caesium are the most accurate way of keeping time, and high pressure sodium lamps are so efficient that they’re used in street lights all over the world. Sodium and potassium are essential elements for health, with major biological roles as electrolytes.
You’ve probably got a device powered by a lithium ion (Li-ion) battery in your pocket, and scientists use Diamond to research new materials to improve Li-ion battery performance, and ways to make sodium ion batteries a viable alternative.
Here are some recent examples of experiments involving Potassium and Lithium at Diamond:
 Soft, silvery-white alkali lithium is the lightest metal and the lightest solid element. Highly reactive and flammable, lithium has to be stored in mineral oil. Industrial applications include heat-resistant glass and ceramics, lithium grease lubricants, flux additives for iron, steel and aluminium production. Mobile devices and electric vehicles depend on lithium-ion batteries.
Soft, silvery-white alkali lithium is the lightest metal and the lightest solid element. Highly reactive and flammable, lithium has to be stored in mineral oil. Industrial applications include heat-resistant glass and ceramics, lithium grease lubricants, flux additives for iron, steel and aluminium production. Mobile devices and electric vehicles depend on lithium-ion batteries.
 Potassium was first isolated from pot ash, the ashes of plants soaked in water in a pot. A soft and silvery-white alkali metal, potassium oxidises rapidly in air, reacts vigorously with water, and burns with a lilac flame. Potassium ions are vital for all living cells, and 95% of global potassium production is used for agricultural fertiliser.
Potassium was first isolated from pot ash, the ashes of plants soaked in water in a pot. A soft and silvery-white alkali metal, potassium oxidises rapidly in air, reacts vigorously with water, and burns with a lilac flame. Potassium ions are vital for all living cells, and 95% of global potassium production is used for agricultural fertiliser.
Treatments for cardiovascular disease, neurological disorders, and pain can be developed by understanding the flow of ions across cell membranes. Potassium 2-pore domain (K2P) channels are important clinical targets, but the mechanisms that control how these channels open and close were not well understood.
The TREK-2 K2P channel is regulated by a range of stimuli, including mechanical changes to the membrane, chemical changes such as pH, biological molecules and many pharmacological agents such as volatile anaesthetics, neuroprotective drugs, and antidepressants such as fluoxetine (Prozac). Researchers used Diamond’s Macromolecular Crystallography (MX) beamlines to elucidate the structure of TREK-2, and their work is a step towards designing molecules that specifically bind to individual K2P channel conformations, opening up new avenues for the design of pain relief treatments and neuropsychiatric treatments. Read more here.
The surface of many Old Master paintings has been affected by the appearance of whitish lead-rich deposits, which are often difficult to fully characterise, thereby hindering conservation. Painted in 1663, Rembrandt’s Homer has a long and eventful past, which has taken its toll on the painting’s chemistry. A barely-visible, whitish crust has formed on the surface of the painting, indicating that chemical reactions are occurring that could cause damage.
A team of conservation scientists from the Mauritshuis in the Hague and the Rijksmuseum in Amsterdam, University of Amsterdam and scientists from Finden Ltd, UCL and Diamond carefully prepared a micro-sample of the damaged paint, and investigated its complex chemistry using X-ray Diffraction Computed Tomography (XRD-CT). They identified a complex mixture of lead sulfates, including lead (potassium) sulfrates, which formed due to the reaction of the chemicals in the surface layers with the toxic gases from harsh environments the painting has experienced in the past.
High-energy density lithium ion batteries are used for a wide range of applications, from portable electronics and medical devices to aerospace and manned spaceflight. Maintaining a high standard of safety for lithium ion cells is more important now than ever before, particularly since high-energy density cells are expected to operate under challenging conditions for applications that are often ‘mission critical’. For example, when a battery pack for the auxiliary power unit caught fire on a Boeing Dreamliner in 2014, the entire fleet of aircraft was grounded for several months due to passenger safety concerns.
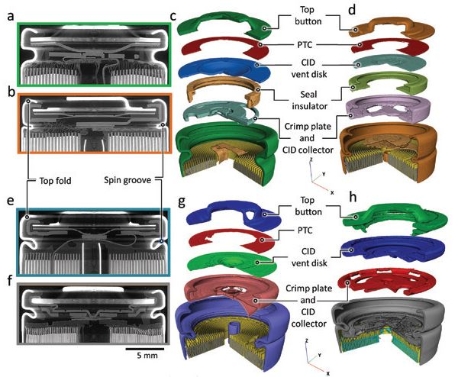
Battery cell failure is extremely rare during normal operation, but under abusive conditions the active materials within Li-ion batteries break down exothermically, generating large amounts of heat that can lead to a positive feedback loop, fire and even explosions – a phenomenon known as thermal runaway. Cell rupture during thermal runaway is one of the most hazardous types of failure, and yet the stages leading to rupture are not well understood. Researchers investigating the causes of rupture use Diamond’s high-speed X-ray imaging capabilities to capture the dynamic sequence of events with enough detail, and their results will guide the development of safe next-generation Li-ion cells. Read more here.
The Li-ion diffusion rate provides a fundamental limitation to the rate at which a battery can be charged and discharged. The conventional approach to improving the Li-ion diffusion rate is to create electrodes with nanoscale structures, which decrease the distance Li-ions must travel, and greatly increase the surface area of the electrode that is in contact with the electrolyte. However, this approach reduces the volumetric energy density (the amount of energy the battery can store), and producing electrodes with nano-architectures is slow and expensive, and can produce large amounts of chemical waste.

Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.