
Want to learn more about one of science's heroes from history, Henry Moseley? Moseley solved one of chemistry's greatest puzzles - determining what distinguishes elements from one another and developed a means of identifying elements based on their atomic characteristics. Sadly he lost his life fighting at Gallipoli in WWI.
Learn more about his life and legacy by watching our online film here.
Non-metals tend to have relatively low melting and boiling points and low densities. They are usually poor conductors of heat and electricity, and the solid elements are mostly brittle. They can be divided into two categories - the noble gases and reactive non-metals. Metalloids are also sometimes counted as non-metals.
There are five times as many metal element as non-metals, but the noble gases hydrogen and helium make up more than 99% of the observable universe, and oxygen makes up nearly half of Earth’s crust, oceans and atmosphere. Non-metals form many more compounds than metals, and living organisms are mostly made of non-metals.
 Carbon is the fourth most abundant element in the universe, and a common element in all known forms of life. Its name come from the Latin word carbo, for charcoal. Carbon has an unusual ability to form polymers at temperatures found on our planet and forms a unique range of organic compounds, and is sometimes referred to as the "King of the Elements".
Carbon is the fourth most abundant element in the universe, and a common element in all known forms of life. Its name come from the Latin word carbo, for charcoal. Carbon has an unusual ability to form polymers at temperatures found on our planet and forms a unique range of organic compounds, and is sometimes referred to as the "King of the Elements".
 The global carbon cycle is key to Earth's habitability, giving our planet a stable and hospitable climate, and an atmosphere relatively low in carbon dioxide. Diagrams of the carbon cycle typically show carbon repositories in the atmosphere, the oceans, and the soil, but not what's going on deeper underground. Earth's mantle potentially holds more carbon than all of the other reservoirs combined. However, investigating the mantle is not easy. It extends from about 35 to 2,890 km below the surface, with temperatures which rise from 200 °C at the upper boundary with the crust to approximately 4,000 °C at the core.
The global carbon cycle is key to Earth's habitability, giving our planet a stable and hospitable climate, and an atmosphere relatively low in carbon dioxide. Diagrams of the carbon cycle typically show carbon repositories in the atmosphere, the oceans, and the soil, but not what's going on deeper underground. Earth's mantle potentially holds more carbon than all of the other reservoirs combined. However, investigating the mantle is not easy. It extends from about 35 to 2,890 km below the surface, with temperatures which rise from 200 °C at the upper boundary with the crust to approximately 4,000 °C at the core.
Using diamond anvil cells (DAC) and laser heating to recreate the extreme conditions in Earth's mantle, a team of experimental geoscientists from the University of Bristol investigated what happens to carbonate minerals subducted from the oceanic crust into the mantle. They found that decarbonation reactions prevent subduction of carbonate deeper than around 1500 km and that the mantle stores carbon as diamonds. Read more here.
 Recent years have seen a global move towards renewable energy, to reduce carbon dioxide emissions from burning fossil fuels. One problem with renewable energy sources is that they tend to produce more energy than we can store during periods of low user consumption (during the day) and not enough during periods of high demand (in the evening). One solution is electrochemical energy conversion technologies, which can use chemical waste products such as carbon dioxide to fuel the chemical reactions. However, converting carbon dioxide directly and selectively to the desired chemicals and fuels is one of the biggest challenges of modern chemistry.
Recent years have seen a global move towards renewable energy, to reduce carbon dioxide emissions from burning fossil fuels. One problem with renewable energy sources is that they tend to produce more energy than we can store during periods of low user consumption (during the day) and not enough during periods of high demand (in the evening). One solution is electrochemical energy conversion technologies, which can use chemical waste products such as carbon dioxide to fuel the chemical reactions. However, converting carbon dioxide directly and selectively to the desired chemicals and fuels is one of the biggest challenges of modern chemistry.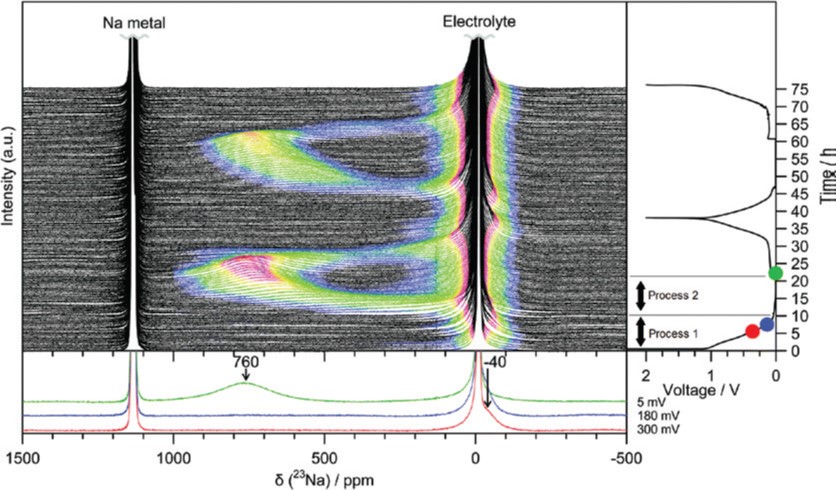 The Nobel Prize in Chemistry 2019 was awarded to three scientists for their contribution to the development of the lithium-ion battery. We now use these lightweight, rechargeable and powerful batteries in everything from mobile phones and laptops to electric vehicles. They can also store significant amounts of energy from solar and wind power, bringing us much closer to a fossil fuel-free society. However, lithium is a limited and expensive resource.
The Nobel Prize in Chemistry 2019 was awarded to three scientists for their contribution to the development of the lithium-ion battery. We now use these lightweight, rechargeable and powerful batteries in everything from mobile phones and laptops to electric vehicles. They can also store significant amounts of energy from solar and wind power, bringing us much closer to a fossil fuel-free society. However, lithium is a limited and expensive resource. 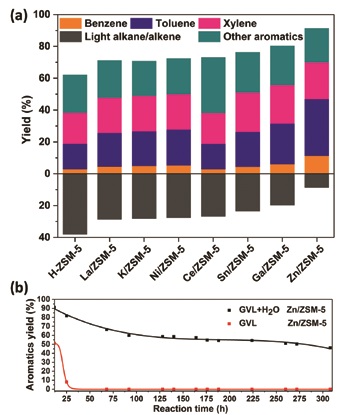 The world currently relies on coal, oil and gas, not only for fuel but also as raw materials for the production of chemicals. With reserves of these fossil fuels running out, and a growing awareness of the carbon dioxide pollution their use causes, it is becoming increasingly important to develop sustainable carbon sources.
The world currently relies on coal, oil and gas, not only for fuel but also as raw materials for the production of chemicals. With reserves of these fossil fuels running out, and a growing awareness of the carbon dioxide pollution their use causes, it is becoming increasingly important to develop sustainable carbon sources.
One option is to use dry plant matter - biomass - and a team of researchers have demonstrated a new method for converting biomass into butene gas, which can be processed into the chemicals used in the production of polymers and resins. Their work focused on Gamma-valerolactone (GVL), a chemical processed from biomass raw material, and the catalyst Zn/ZSM-15. They used high-resolution X-ray powder diffraction (SRXD) on Ill to examine the structure of their samples, which yielded valuable information about the reaction mechanism.
This was the first time that SXRD had been used to investigate the structures of adsorbed structures of the Gamma-valerolactone GVL and immobilised Zn-species used in the research. Their results demonstrated that it is possible to use a renewable source material to produce benzene, toluene and xylene, and are a step towards affordable, sustainable chemical production. Read more here.
 Remote probes tell us that Saturn's moon Enceladus has both water ice and carbon dioxide on its surface, so it is a potential habitat for extraterrestrial microbial life. Conditions on Enceladus - and other icy satellites - may promote the formation of clathrates, cage-like structures of water molecules that house guest gas species. Clathrates form when the gas interacts with ice under high-pressure and low-temperature conditions, and are thought to influence the surface geology and composition of icy bodies in the Solar System. Research suggests that the release of gases (e.g. carbon dioxide) from clathrates is the source of gas plumes emanating from the surface of Enceladus.
Remote probes tell us that Saturn's moon Enceladus has both water ice and carbon dioxide on its surface, so it is a potential habitat for extraterrestrial microbial life. Conditions on Enceladus - and other icy satellites - may promote the formation of clathrates, cage-like structures of water molecules that house guest gas species. Clathrates form when the gas interacts with ice under high-pressure and low-temperature conditions, and are thought to influence the surface geology and composition of icy bodies in the Solar System. Research suggests that the release of gases (e.g. carbon dioxide) from clathrates is the source of gas plumes emanating from the surface of Enceladus.Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.