________________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
Nearly all types of electromagnetic radiation can be used for spectroscopy to study and characterise matter. When using X-rays the photoelectric effect occurs.
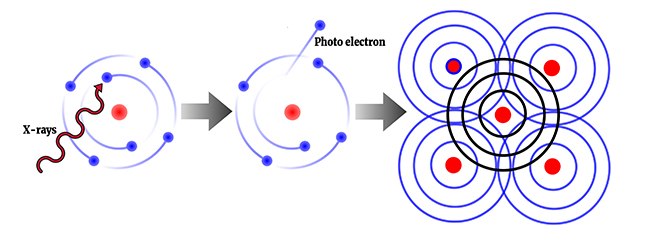
The photoelectric effect refers to the emission or ejection of electrons from the surface of a sample in response to electromagnetic radiation or light. Energy contained within the electromagnetic radiation is absorbed by electrons within the sample, giving the electrons sufficient energy to be 'knocked' out of (emitted from) the sample.
In the X-ray region atoms absorb X-rays sharply at certain wavelengths (called absorption edges) that are characteristic of that particular atomic species. So if you shine a particular frequency or energy of X-rays onto a sample, you can control the type of electron that is emitted.
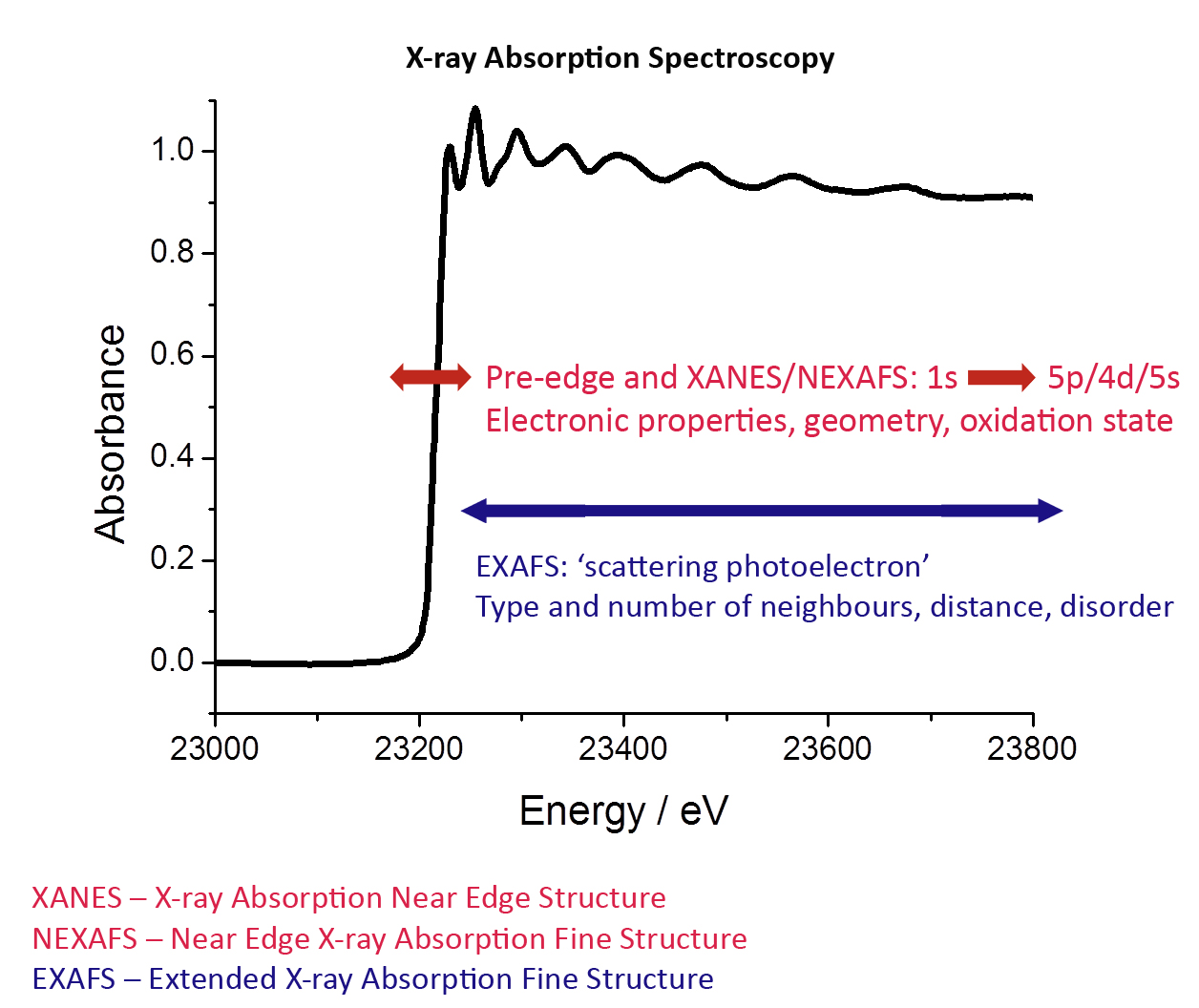
X-ray Absorption Near Edge Strucure (XANES) refers to the absorption fine structure close to an absorption edge, about 10 eV below the absorption edge and 20 eV above the edge. This region usually shows the largest variations in the X-ray absorption coefficient and is often dominated by intense, narrow resonances. This part of the spectrum provides information on the electronic structure of the unoccupied levels and the geometry of the studied element - very few techniques can do this.
XANES is also referred to as Near Edge X-Ray Absorption Fine Structure (NEXAFS). Today, the term NEXAFS is typically used for soft X-ray absorption spectra (surface science, organics) and XANES for hard X-ray spectra (coordination chemistry, metal atoms).
EXAFS is the part of XAS spectrum - typically starting ~ 50 eV above the absorption edge. It originates from the scattering of a photoelectron ejected from the absorbing atom and its neighbours. EXAFS is therefore typically used to determine the local structure of the studied element and its surrounding atoms.
Energy Dispersive EXAFS (EDE) gives a snapshot view of data collected through XAS. The principle of EDE is based on the diffraction of non-monochromatic X-rays by a bent crystal (polychromator).
The polychromatic beam is focused on the sample and then diverges towards a position-sensitive detector where the beam position is correlated to the energy.
The dispersive configuration of XAS has two main advantages that make it scientifically attractive.
First, the whole X-ray absorption spectrum is collected simultaneously which makes the technique especially useful for the study of fast processes.
Second, the size of the focussed beam at the sample position is small and very stable due to the fact that no movement of optical elements is required to collect the spectral data. This feature of the technique is often used in high pressure studies.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.