Recommended viewing

Want to learn more about one of science's heroes from history, Henry Moseley? Moseley solved one of chemistry's greatest puzzles - determining what distinguishes elements from one another and developed a means of identifying elements based on their atomic characteristics. Sadly he lost his life fighting at Gallipoli in WWI.
Learn more about his life and legacy by watching our online film here.
Metalloids
The metalloids have properties that are either a mixture of those common to metals and non-metals, or in-between the two. On the periodic table, they lie on either side of the dividing line between metals and nonmetals, and include boron, silicon, germanium, arsenic, antimony, and tellurium. While metalloids typically have a metallic appearance, they are usually too brittle to have structural uses. They can form alloys with metals, and they are found in biological agents, catalysts, flame retardants, glasses, optical storage and optoelectronics, pyrotechnics, semiconductors, and electronics.
The development of the semiconductor industry in the 1950s and solid-state electronics from the early 1960s both depended on the electrical properties of silicon and germanium.
Featured Element: Arsenic
 The toxic properties of arsenic were known to the Ancient Romans, and it was the poison of choice for the professional poisoners of Renaissance Europe. It wasn't until British chemist James Marsh developed a test to detect minute traces of arsenic in food and human remains - in 1836 - that the epidemic of arsenic poisoning came to an end.
The toxic properties of arsenic were known to the Ancient Romans, and it was the poison of choice for the professional poisoners of Renaissance Europe. It wasn't until British chemist James Marsh developed a test to detect minute traces of arsenic in food and human remains - in 1836 - that the epidemic of arsenic poisoning came to an end.
Industrial uses of arsenic include alloys with lead (for car batteries and bullets) and gallium arsenide, which is the second most commonly used semiconductor after silicon. Use of arsenic in pesticides, herbicides and insecticides is declining due to its toxicity, but millions of people are affected by naturally-occurring arsenic contamination of groundwater.
Iron-Arsenic Superconductors
In 2015, Jenny Rogers from the Centre for Science at Extreme Conditions at the University of Edinburgh talked about her work using high pressures and temperatures to synthesise new materials. The machine she used can produce a force of about 1,000 tons, generating pressures of about 100,000 atmospheres. High pressures allow scientists to create new materials, and Jenny's team were working on iron-arsenic superconductors. With the first one discovered just a couple of years earlier by a group in Japan, the race was on for chemists and physicists around the world to try to make these new iron-arsenic superconductors. Diamond's X-rays allowed the team to see the structure of the materials they made, which kept their remarkable properties when they were returned to normal pressures. Learn more here.
Finding the Weyl Fermion
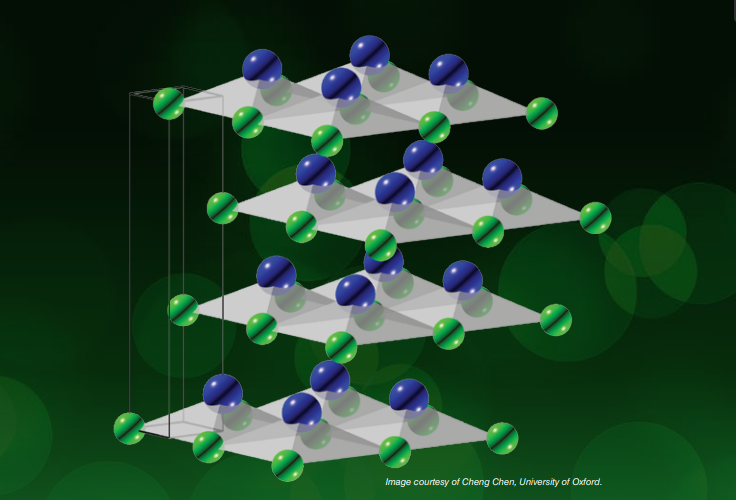 In 1929, German mathematician Hermann Weyl predicted the existence of the Weyl fermion, a new subatomic particle. Researchers kept looking for it, but it consistently failed to show up in experiments, and so gradually slipped into obscurity. When modern researchers investigated the properties of some curious crystals made from a combination of the elements tellurium and arsenic, known as tellurium arsenide, they found that its crystals lack a special point called an inversion centre. A UK group of scientists, led by Yulin Chen, using ARPES on Diamond’s I05 beamline discovered a feature that showed the presence of the elusive Weyl fermion. American and Chinese researchers achieved the same result. It was hailed as the physics breakthrough of the year 2015 by the Institute of Physics and others.
In 1929, German mathematician Hermann Weyl predicted the existence of the Weyl fermion, a new subatomic particle. Researchers kept looking for it, but it consistently failed to show up in experiments, and so gradually slipped into obscurity. When modern researchers investigated the properties of some curious crystals made from a combination of the elements tellurium and arsenic, known as tellurium arsenide, they found that its crystals lack a special point called an inversion centre. A UK group of scientists, led by Yulin Chen, using ARPES on Diamond’s I05 beamline discovered a feature that showed the presence of the elusive Weyl fermion. American and Chinese researchers achieved the same result. It was hailed as the physics breakthrough of the year 2015 by the Institute of Physics and others.
Weyl fermions can carry electrical charge much faster than electrons in ordinary materials, which could make them useful for next-generation computing technologies. Read more on page 16 of Inside Diamond here.
Investigating Toxic Sludge

In October 2010, a dam failure at an aluminium plant in Hungary released around one million cubic metres of toxic red sludge into nearby towns and villages and across agricultural land. The sludge was a mixture of water and mining waste containing heavy metals and was deemed a threat to the environment by Hungarian officials. A team from the University of Leeds used Diamond’s Microfocus Spectroscopy beamline, I18, to determine the chemical form of arsenic, chromium and vanadium found in samples of the red sludge. Knowing the precise chemical form of a toxic metal allows scientists to predict how harmful it is likely to be in the environment.
Their results suggested that the chemical state of the arsenic and the chromium would restrict their environmental mobility, so those elements were unlikely to present a significant danger. However, toxic vanadium from the red sludge was likely to be more mobile. Read more here.
Can earthworms clean toxic soil?
 Mark Hodson from the University of Reading talked to Diamond about his research into earthworms in contaminated soils. Former mining sites from the 19th century have high levels of metals in the soil, and plants don't grow well. They were looking at an old lead mining site in Wales where the primary contaminant is lead, one in the Pennine ore field that is contaminated by lead and zinc, and a third in the South West where there is lots of arsenic and copper.
Mark Hodson from the University of Reading talked to Diamond about his research into earthworms in contaminated soils. Former mining sites from the 19th century have high levels of metals in the soil, and plants don't grow well. They were looking at an old lead mining site in Wales where the primary contaminant is lead, one in the Pennine ore field that is contaminated by lead and zinc, and a third in the South West where there is lots of arsenic and copper.
But if you dig into the soil at those sites, you still find earthworms. How do they cope with the metals? Mark's team used Diamond to investigate the form of the metals in the soil, and after the worms had eaten them. They found that earthworms take the metals in and change the form of the metals to make them less toxic. The research suggested that ingesting the metals triggers the production of a special protein, which essentially wraps around the metal and makes it non-toxic to the earthworm. In the future, we may be able to use these metal-munching earthworms to help us clean up contaminated soils. Learn more here.
Contaminated Well Water in Bangladesh
In the 1970s, between 7-11 million deep wells were dug throughout Bangladesh. The intention was to give the Bangladeshi people a safe source of drinking water, free from the bacteria that cause diarrhoea and other intestinal diseases that were plaguing the country. It wasn't until 1993 that we discovered this "clean" well water contained dangerous quantities of arsenic, resulting in the largest mass poisoning of a population in history.
Bangladesh isn't the only country with this problem. In 2002, the World Health Organization estimated that about 13 million people in the USA were drinking arsenic-tainted water, as well as people in Argentina, Brazil, Chile, Hungary, Mexico, Taiwan (Province of China), Thailand, Viet Nam, and Bengal.
A group of researchers from Manchester used Diamond to investigate ways of using bacteria to deal with arsenic contamination. The researchers used X-ray microfocus spectroscopy to look at arsenic before and after it has been treated by the bacteria. Bacteria can make arsenic insoluble, so depositing them in the wells could ensure that the arsenic isn't in the water people drink. So even if you had put a well into arsenic-contaminated land, you use bacteria is a bio-filter to change the arsenic so that the well provides clean water. Learn more here.
And did you know?

Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.
