Find out more about our ambitious upgrade project, delivering more brightness, more coherence, and greater speed of analysis to UK science. More about Diamond-II
![]()
Find out more about Diamond's response to virus research.
![]()
Discover more groundbreaking research on cancer at Diamond.Read On Cancer Literature Review
Yasinska IM et al. The Tim-3-Galectin-9 Pathway and Its Regulatory Mechanisms in Human Breast Cancer. Frontiers in Immunology 10, 1594 (2019). DOI: 10.3389/fimmu.2019.01594.
Dr Vadim Sumbayev explains,
The human immune system has cells that can attack invading pathogens, protecting us from bacteria and viruses. These cells are also capable of killing cancer cells, but they don't. Cancer cells have evolved defence mechanisms that protect them from our immune system, allowing them to survive and replicate, growing into tumours that may then spread across the body. Unfortunately, the molecular mechanisms that allow cancer cells to escape host immune surveillance remain poorly understood. So, with a growing body of evidence suggesting that some solid tumours also use proteins called Tim-3 and galectin-9 and to evade host immune attack, we chose to study the activity of this pathway in breast and other solid and liquid tumours.
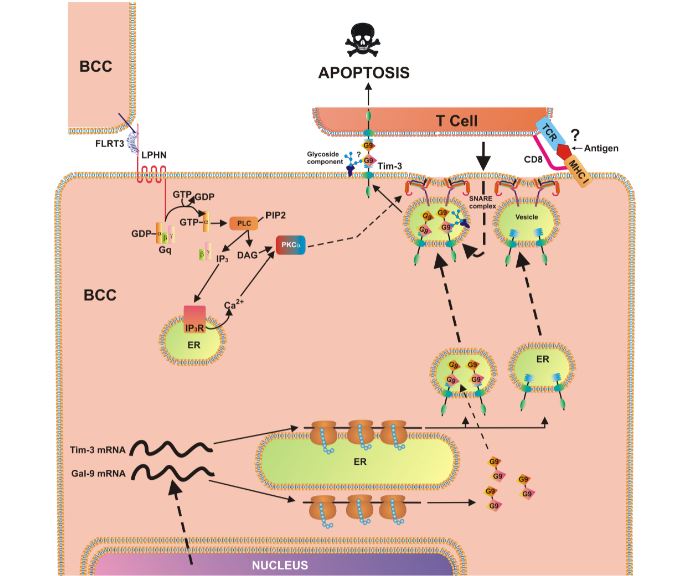
The teams’ previous work at Diamond led them to conclude that the immune receptor Tim-3 and the protein it naturally binds to galectin-9, play a role in allowing acute myeloid leukaemia (AML) cells escape the human immune system. Their new work found that breast tumours express significantly higher levels of Tim-3 and galectin-9 than healthy breast tissues, with the result suggesting that both proteins are expressed by the same cells. Further experiments showed that a wide range of different cancers express detectable amounts of both Tim-3 and galectin-9.
Our findings demonstrate the activity of the Tim-3-galectin-9 biochemical pathway in several types of human cancer cells, as well as its possible role in the suppression of the immune system response. Our ultimate goal is now to find the best way to disable the Tim-3-galectin-9. This could lead to therapies that allow our immune systems to attack cancer, reducing the need for more toxic treatments such as chemotherapy and radiotherapy, which have severe side-effects for patients.
The team are already planning another visit to Diamond, for the next stage in this vital research. "Access to Diamond is crucial to our research. The sensitivity that synchrotron radiation allows cannot be replicated using other techniques, and it allows us to really see what's happening to the proteins, even small details," concludes Dr Sumbayev. At Diamond, the team used Synchrotron Radiation Circular Dichroism (SRCD) spectroscopy on beamline B23 to investigate the point at which the pathway begins.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.