A multi-technique study of polymorphism in NaNb03
Oct 14, 2010
Oct 14, 2010
A polar phase of the perovskite NaNbO3 has been successfully synthesised using sol-gel techniques. Detailed characterisation of this phase has been undertaken using high-resolution powder diffraction (X-ray and neutron) and 23Na multiple-quantum (MQ) MAS NMR, supported by second harmonic generation (SHG) measurements and density functional theory (DFT) calculations. Samples of NaNbO3 were also synthesised using conventional solid-state methods and were observed to routinely comprise a mixture of two different polymorphs of NaNbO3, namely the well-known orthorhombic phase (space group Pbcm) and the current polar phase, the relative quantities of which vary considerably depending upon precise reaction conditions. Our studies show that each of these two polymorphs of NaNbO3 contains two crystallographically distinct Na sites. This is consistent with assignment of the polar phase to the orthorhombic space group P21ma, although peak broadenings in the diffraction data suggest a subtle monoclinic distortion. Using carefully monitored molten salt techniques it was possible to eradicate the polar polymorph and synthesise the pure Pbcm phase.
Sodium niobate, NaNbO3, is a perovskite that possesses an extremely detailed and complex phase diagram containing a series of complicated phase transitions as a function of temperature.[1] In recent years several areas have been heavily re-examined, in particular the low and high temperature regions. One area still widely debated however is the nature of room temperature polymorph(s) of NaNbO3. The aim of this multi-technique study was to understand in detail the structural nature of these polymorphs and their inter-relation, in particular how synthetic methods dictate which polymorphs are formed. The use of high-resolution powder XRD (I11 at Diamond) was pivotal in unravelling some of the complexities in this study. The complementarity of this technique to both neutron diffraction and solid-state NMR (which reveals short-range order rather than long-range crystallographic order) are emphasised.
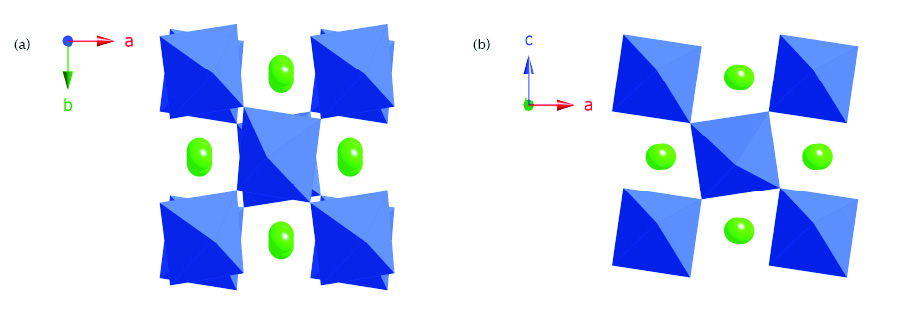
Figure 1: The two polymorphs of NaNbO3, showing the differing ‘tilt’ systems, and cation displacement modes: (a) the well-known Pbcm phase (b) the ‘new’ P21ma phase.
A key success of this study, which exploited the superior resolution characteristics of I11, was the identification of the occurrence of phase mixtures of two very similar perovskite phases in NaNbO3 samples prepared by a variety of methods. Fig. 1 shows the two phases of NaNbO3, designated by their space group symbols; the two structures differ in the nature of their ‘octahedral tilting’ and ensuing cation displacements. The “P21ma” phase [2][3] is characterised in full here, for the first time. This phase has a “polar” structure which is of specific interest due to its potential piezo- and ferro-electric properties [4]. Fig. 2 shows a typical Rietveld plot, which models the structural behaviour of an as-made sample of NaNbO3. As can be seen in the full-range plots, good quality fits can be obtained by fitting a single phase model. However, on closer inspection, fitting a single phase model leaves subtle discrepancies, revealed clearly in the expanded plots, which are only adequately fit by incorporation of a secondary phase. Although the major peaks due to each of the two phases essentially ‘overlap’, the weak superlattice peaks are distinct and characteristic to each phase, due to the differing tilt systems. The nature of this second phase has been confirmed not only from the I11 data but also from complementary and self-consistent neutron, NMR and SHG measurements. The structural similarity of the two phases, and their correspondingly similar diffraction patterns, means that this subtle phase co-existence has probably been missed in many previous works: The highest quality powder diffraction data, together with the additional local-structure information provided by solid-state NMR, is essential in pinning down this extremely subtle phase behaviour.

Figure 2: Rietveld plots of I11 data: (a) and (b) single phase and two-phase refinements, respectively, of the full data set. (c) and (d) corresponding expansion of the key region around 20° showing the significant improvements on using the two-phase model. Note that these peaks correspond to tiny ‘background’ features in the full-scale plots!
In summary, we have confirmed it is possible to synthesise a polar phase (probably monoclinic, but of symmetry very close to orthorhombic, space group P21ma) of NaNbO3 using sol-gel techniques. We have also shown, by means of a systematic study, that synthetic method affects the polymorphic forms of NaNbO3 produced in any one reaction. It is possible to isolate a pure sample of the ‘normal’ Pbcm polymorph, and high-resolution powder diffraction studies confirm that this is indeed a correct space group assignment for this phase: previous confusion regarding the possibility of a symmetry-lowering to monoclinic can be explained by the occurrence of a significant “P21ma” phase impurity in the samples previously studied. Routinely the phases Pbcm and “P21ma” coexist as the two major room temperature polymorphs of NaNbO3. The thermodynamic stabilities of the two phases are believed to be extremely similar, therefore slight variations in the reaction conditions forces one to preferentially form over the other. Consistently the Pbcm polymorph forms more readily, suggesting it to be the more thermodynamically stable of the two. Finally, we emphasise that it is essential to be aware of this subtle polymorphism in NaNbO3, its occurrence as a function of preparative method, and how to identify it from both diffraction and NMR data; much of the earlier work regarding NaNbO3 and its phase diagram should be treated with some degree of caution in light of these findings.
References
[1] C. N. W. Darlington and K. S. Knight, Physica B, 266, 368 (1999).
[2] S. E. Ashbrook et al., Phys. Chem. Chem. Phys., 8, 3423 (2006).
[3] Y. Shiratori et al., J. Phys. Chem. B, 109, 20122 (2005).
[4] Y. Saito et al., Nature, 432, 84 (2004).
Principal Publications and Authors
K. E. Johnston, C.C. Tang, J. E. Parker, K.S. Knight, P. Lightfoot, and S.E. Ashbrook, The polar phase of NaNbO3; a combined study by powder diffraction, solid-state NMR and first-principles calculations, J. Am. Chem. Soc. 132, 8732–8746,(2010).
Funding Acknowledgement
Engineering and Physical Sciences Research Council, UK.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.