One step closer to preventing hip implant replacements
May 11, 2015
May 11, 2015
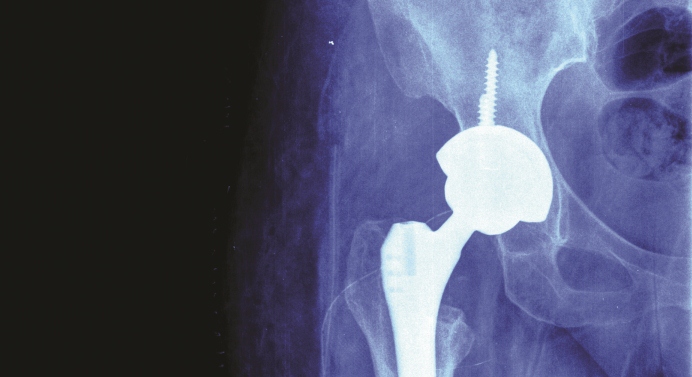 The hip joint is one of the largest joints in the human body and over the last decade, it is estimated that over half a million hips have been replaced in England and Wales as a result of osteoarthritis1. Osteoarthritis is the most common joint disorder in adults, affecting over eight million people in the UK alone2. Surgical replacement of the joint using artificial metal implants is the most effective way to restore activity and reduce pain and disability in osteoarthritis sufferers.
The hip joint is one of the largest joints in the human body and over the last decade, it is estimated that over half a million hips have been replaced in England and Wales as a result of osteoarthritis1. Osteoarthritis is the most common joint disorder in adults, affecting over eight million people in the UK alone2. Surgical replacement of the joint using artificial metal implants is the most effective way to restore activity and reduce pain and disability in osteoarthritis sufferers.
Hip implants rely on the normal functioning of bone cells to achieve fixation of the implant with the bone. However, small metal particles, cobalt (Co) and chromium (Cr), released from hip implants due to friction between the moving surfaces, have been shown to be toxic to the surrounding bone cells. This causes the implant to loosen in the bone and often leads to patients requiring a second surgery to replace the failed implant.
In this study, world-leading researchers from the University of Sheffield used the Microfocus Spectroscopy beamline (I18) and microfocus X-ray spectroscopy, an analytical technique that uses high energy X-ray beams derived by synchrotron radiation, to determine the chemical form and oxidation state of an element in microscopic samples. Specifically, they used Microfocus X-ray Fluorescence (µ-XRF) to measure the elemental distribution and micro-X-ray Absorption Near-Edge Structure (µ-XANES) to characterise the chemical form of the metals in human bone cells.
The overall goal of these studies was to better understand the cellular entry, trafficking, and processing of Co and Cr in bone cells, and identify possible manageable targets to alleviate their adverse effects after total hip replacement. The findings, published in the Journal of Orthopaedic Research, reveal that the location of the metals that are released from implants is different between osteoblasts and osteoclasts, the two types of bone cells responsible for bone maintenance and repair.
Dr Alison Gartland, Senior Lecturer from the University’s Academic Unit of Bone Biology, said: “The fact that we found metal ions in different places within the two types of bone cells suggest that they get into the cells by separate ways. When investigating how the metals entered bone cells we found that when they blocked a molecule called the P2X7 receptor using a specific drug, the entry of metals into the bone cells was reduced. “These results are really exciting because, if we can prevent the entry of the metal into these cells, hopefully we can prevent the metal joint from failing.”
The research was funded by the National Institute for Health Research (NIHR) and carried out in collaboration with Diamond. For more information on the I18 beamline or to discuss potential applications, please contact Principal Beamline Scientist Prof Fred Mosselmans: [email protected]
Shah KM. et al. Understanding the tissue effects of tribo-corrosion: uptake, distribution, and speciation of cobalt and chromium in human bone cells. Journal of Orthopaedic Research (2014) DOI:10.1002/jor.22729
1. National Joint Registry for England and Wales. 11th Annual Report 2014. (2014) p.27.
2. Arthritis Research UK. Osteoarthritis in General Practice. (2013) p.9
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.