- Diamond Light Source
- News & Literature
- Print Publications
- Diamond News
- Autumn 2015
- Focus on science
- The evolution of an active catalyst
Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
The majority of commercially produced materials and chemicals are made using manufacturing processes that utilise catalysts, which help to prompt and speed up chemical reactions. Catalysts perform this function by reducing the activation energy of a reaction, which increases the reaction rate compared with uncatalysed reactions in identical conditions.
Catalysts are chosen based on the reaction they enhance and they can be made of a wide range of materials. One such catalyst, gold-palladium (AuPd) formulated as nanoparticles, is used widely in different catalytic applications, including the industrial production of vinyl acetate. Despite being well-studied, the composition and structure of these nanoparticles over the course of a reaction has not yet been tracked; therefore, efforts were undertaken at Diamond to shed some light on the behaviour of these bimetallic nanoparticles.
Two techniques were combined at Diamond’s Core XAFS beamline (B18): XAFS (X-ray Absorption Fine Structure), which in this instance provided information on the interior of the nanoparticles, and DRIFTS (Diffuse Reflectance Infrared Fourier Transform Spectroscopy) to observe the surface of the nanoparticles during a reaction. Together, they showed that the AuPd nanoparticles underwent significant changes during an oxidation reaction.
Initially, the nanoparticles have a surface consisting of a mixture of oxidic and metallic components, whereas the interior comprised of an Au core surrounded by a mixture of Au and Pd. At the end of the reaction the exterior of the particle restructured to solely metallic Pd and the interior of the particle remained mostly unchanged (Fig. 1). This approach demonstrated how the two techniques can be used to provide valuable insights into the composition of the nanoparticles directly under reaction conditions, which could be applied to many other types of catalysts.
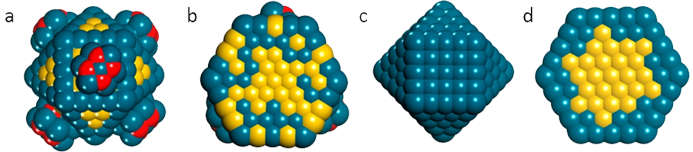
Fig. 1: Schematic representation of (a) the fresh catalyst, (b) the cross-section of the fresh catalyst, (c) the final catalyst state after reaction, and (d) the crosssection of the final catalyst, where the red, blue, and yellow balls indicate O, Pd, and Au atoms, respectively. Reprinted with permission from Gibson et al. Chem Mater. 37, 3714–3720 (2015). ©2015 American Chemical Society.
What was done?
The structure of an AuPd nanoparticle is dynamic over the course of a reaction. However, efforts in the past to explore the composition of the nanoparticles have focused on their arrangement at the start and end of a reaction, but not during. By monitoring the behaviour of the catalyst over the course of a reaction, it is hoped that valuable information could be gathered to design better catalysts.
The study was conducted by scientists from the UK Catalysis Hub, which is in the Research Complex at Harwell, adjacent to Diamond Light Source. Together with scientists from University College London, they designed a system to carry out two characterisation techniques simultaneously during carbon monoxide oxidation.
 Figure 2: Combined XAFS/DRIFTS set-up installed on B18.
Figure 2: Combined XAFS/DRIFTS set-up installed on B18.
At beamline B18, XAFS was used to look at the composition of the interior of the nanoparticles and DRIFTS was used to characterise their surfaces. The team, along with Diamond scientists, developed a system that utilised a rapid scanning infrared spectrometer, a mechanical arm which extends the optical path of the spectrometer, and a bespoke sample environment which allowed both infrared and X-ray measurements to be performed simultaneously.
The samples were assessed whilst performing a carbon monoxide oxidation experiment. This involved slowly increasing the temperature of the reaction whilst simultaneously flowing carbon monoxide and oxygen. The desired product in this process is carbon dioxide and the temperature at which this is formed is very dependent on the catalyst used.
What progress was made?
The team found that the AuPd nanoparticles underwent a significant amount of restructuring over the course of the oxidation reaction. Dr Peter Wells of the UK Catalysis Hub explained: "The study has allowed us to see what parts of the nanoparticles are actually doing the work during catalysis, and this information is of great importance in this field."
Whilst the AuPd catalyst is of huge interest as it could be used in many catalytic processes such as the upgrading of bio-derived chemicals to more valuable products, this combined setup could also be applied to many other types of catalysts. The wealth of information offered by such a technique under reaction conditions and in real time can help advance our understanding of how catalysts work.
In the future, the team plan to improve their methodology and they will use this novel approach with industrial collaborators to understand how the catalysts they make work under real conditions.
To find out more about using the B18 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Dr Giannantonio Cibin: [email protected]
Gibson EK. et al. Restructuring of AuPd Nanoparticles Studied by a Combined XAFS/DRIFTS Approach. Chem Mater. 37 3714–3720 (2015). DOI: 10.1021/acs.chemmater.5b00866
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.