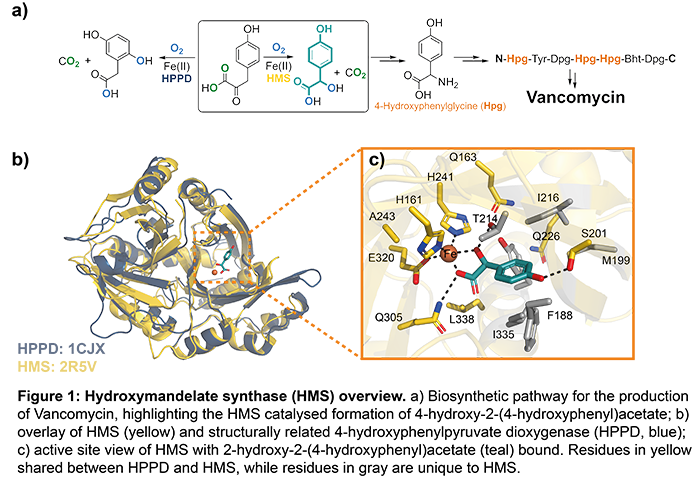
 Antibiotic resistance has become a considerable human health issue in recent decades (Bush et al., Nat Rev Microbiol, 2011, DOI: doi:10.1038/nrmicro2693). Vancomycin, a glycopeptide antibiotic targeting bacterial cell wall biosynthesis, is considered an antibiotic of last resort due to its efficiency in treatment of multidrug resistant bacterial infections, including methicillin-resistant Staphylococcus aureus (MRSA). Production of vancomycin for medical purposes relies on fermentation using Amycolatopsis orientalis, in which hydroxymandelate synthase (HMS), in presence of molecular oxygen, catalyses the formation of 4‑hydroxy‑2‑(4‑hydroxyphenyl)acetate (Figure 1), a recurring skeletal component of vancomycin. HMS is homologous to 4-hydroxyphenylpyruvate dioxygenase (HPPD), closely resembling the α‑carbon backbone and sharing considerable primary sequence identity (Brownlee et al., Biochem, 2008, doi.org/10.1021/bi701438r). Both enzymes bind and react with the same 4‑hydroxyphenylpyruvate substrate, however, forming different products (Figure 1). Limited mechanistic studies on HMS suggest the formation of Fe(III) superoxide and Fe(IV) oxo species as intermediate species during catalysis. So far, no studies have attempted to investigate the active site dynamics of HMS during catalysis in comparison with HPPD. Understanding how HMS and HPPD differentiate their product formation during catalysis using time‑resolved crystallography will improve our understanding of dynamic structural biology and antibiotic resistance.
Antibiotic resistance has become a considerable human health issue in recent decades (Bush et al., Nat Rev Microbiol, 2011, DOI: doi:10.1038/nrmicro2693). Vancomycin, a glycopeptide antibiotic targeting bacterial cell wall biosynthesis, is considered an antibiotic of last resort due to its efficiency in treatment of multidrug resistant bacterial infections, including methicillin-resistant Staphylococcus aureus (MRSA). Production of vancomycin for medical purposes relies on fermentation using Amycolatopsis orientalis, in which hydroxymandelate synthase (HMS), in presence of molecular oxygen, catalyses the formation of 4‑hydroxy‑2‑(4‑hydroxyphenyl)acetate (Figure 1), a recurring skeletal component of vancomycin. HMS is homologous to 4-hydroxyphenylpyruvate dioxygenase (HPPD), closely resembling the α‑carbon backbone and sharing considerable primary sequence identity (Brownlee et al., Biochem, 2008, doi.org/10.1021/bi701438r). Both enzymes bind and react with the same 4‑hydroxyphenylpyruvate substrate, however, forming different products (Figure 1). Limited mechanistic studies on HMS suggest the formation of Fe(III) superoxide and Fe(IV) oxo species as intermediate species during catalysis. So far, no studies have attempted to investigate the active site dynamics of HMS during catalysis in comparison with HPPD. Understanding how HMS and HPPD differentiate their product formation during catalysis using time‑resolved crystallography will improve our understanding of dynamic structural biology and antibiotic resistance.
For the time‑resolved crystallography experiments, a serial crystallography approach is chosen, relying on synchrotron and/or X‑ray free electron laser (XFEL) radiation. An advantage of the serial approach over single crystal rotation approach is that radiation damage can be mitigated, allowing for room temperature data collection, and the observation of photolabile intermediates, such as the Fe(III) superoxide and Fe(IV) oxo species proposed in the HMS mechanism (Rabe et al., Sci Adv, 2021, doi.org/10.1126/sciadv.abh0250; Bergmann et al., Nat Rev Phys, 2021, doi.org/10.1038/s42254-021-00289-3). For this serial crystallography approach, slurries of microcrystals are required. You will be working on the expression, purification, and crystallisation of HMS and when time permits HPPD. An enzyme‑substrate complex will be obtained by crystallising HMS in presence of the 4‑hydroxyphenylpyruvate substrate, under anaerobic conditions. You will be able to participate in beamtime at different beamlines at Diamond Light Source and XFELs abroad. Finally, you will be expected to present your results to researches with different scientific backgrounds.
Essential criteria for applications:
Desirable criteria for applicants:
This project is suitable for students following degrees in the following broad subject areas: Biochemistry
Please note that this role does not meet the required skill level for a Skilled Worker visa and therefore we would be unable to sponsor individuals due to the current UK Home Office immigration rules. To be appointed to the role, candidates will need to have the right to work in the UK without sponsorship from us.
Interviews will take place via ZOOM week commencing 22nd January 2024.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.