Our cells use an ensemble of histone proteins to fold and package the DNA genome into the nucleus. Histones also determine whether to expose DNA to enzymes to allow processes like gene expression, replication, and repair to occur. Although many in vitro studies have explored the mechanism histones use to fold and package DNA into higher-order structures called chromatin, less is known about chromatin organisation inside the nucleus of intact cells, and understanding this phenomenon could be key to understanding multiple DNA-associated processes. Recent advances in cryo-electron tomography have enabled scientists to observe these structures within the nucleus of rapidly cryopreserved cells. Reporting in Nature Communications, scientists at the University of Oxford collaborated with the electron Bio-Imaging Centre (eBIC) at the Diamond Light Source to capture chromatin in the nucleus of immune T cells, revealing that DNA is folded into more flexible and heterogenous fibres than previously modelled. Their experiments lay the groundwork for future studies into the roles of chromatin in health and disease.
Have you ever rushed to pack clothes into a suitcase and skipped the folding step only to find the suitcase wouldn’t close? Though it may have been a struggle, it doesn’t compare to the challenge our cells face when they pack 2 metres of DNA into a nucleus 200,000 times smaller in width. Here an efficient folding mechanism is key, and histone proteins direct the operation.
A complex of histone proteins act as a spool around which 147 base pairs of DNA can wind like thread. Multiple histone spools called nucleosomes can be found along the length of a DNA molecule and coil its strands into so-called chromatin. When chromatin is purified and observed using electron microscopy, scientists have observed that nucleosomes are spaced apart at regular intervals like beads on a string. These beads can then cluster together to form thicker chromatin fibres that pack the DNA into an even smaller volume.
Beyond efficiently folding DNA to fit inside the nucleus, histones play vital roles in regulating gene expression, DNA replication, and repair by loosening or tightening their grip on DNA and controlling its exposure to enzymes. An in-depth understanding of the folding mechanism could help researchers understand how chromatin affects multiple processes within the nucleus.
Scientists have struggled to study the higher order structures in chromatin fibres, partly because chromatin doesn’t retain its shape when extracted from cells during experiments. Currently there are two competing models for chromatin folding: in the solenoid model, nucleosomes wrap around each other like a spring or coil, and in the zigzag model, nucleosomes organise themselves into alternating zigzag patterns connected by straightened DNA links. The jury’s out on how strictly chromatin adheres to either model, and it may only be possible to explore this by observing chromatin within the nucleus.
Cryo-electron tomography (cryoET) has become a prominent tool for imaging cells in a state that resembles physiological conditions. Unlike other electron microscopy techniques that rely on harsh sample processing steps involving chemical fixation, dehydration, and resin-embedding, cryoET isn’t as artefact-prone because it uses cryopreserved samples that retain their native structures. In a process called focused ion beam milling (cryoFIB milling) the samples are sliced into thin sections called lamellae that enable electrons to pass through the sample during imaging. However, lamellae are often too thick (approximately 150 to 200 nm) to detect individual nucleosomes within chromatin.
When scientists manually trim down lamellae too much they risk losing their samples, so a psychological barrier impedes them from trimming them down to an ideal depth, Peijun Zhang, coauthor of the study noted. But eBIC recently acquired an automated cryoFIB instrument to overcome this issue. Zhang noted;
“The newest generation of cryoFIB/SEM Aquilos 2 with automated lamellar processing allows you to generate very thin 80 or 90 nm cellular lamellae.”
Zhang and her colleagues were studying T cells for an unrelated project but were taken by surprise when they could see detailed chromatin in their data. Continuing, Zhang said;
We said ‘wow, this is amazing! People have been hunting for this for ages and we can suddenly see it’
The thin lamellae created by the automated cryoFIB allowed the team to study native chromatin organization in fine detail.
Cryo-electron tomogram showing the entire depth of a lamella from a T cell. Multiple features were segmented, including nucleosomes (beige), the nuclear membrane (grey), and ribosomes (blue). Image taken from Hou et al. Nat Commun (2023) under a CC BY 4.0 license.
The researchers observed that chromatin was more flexible and heterogenous in shape than the models suggested. Both models proposed chromatin fibers should be 30-nm thick, but the researchers observed a range from 20 to 50 nm; the DNA wasn’t all folded and packed in the same fashion.
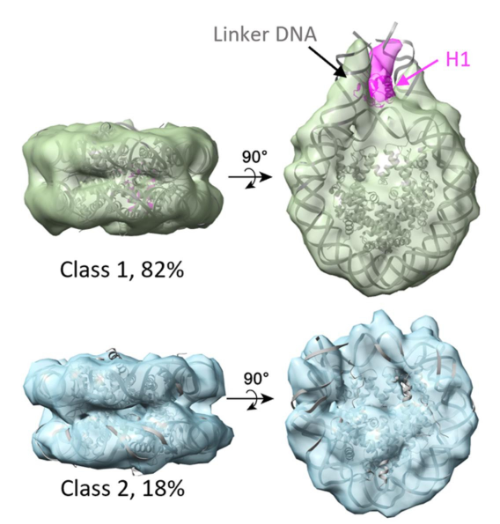
Next, they used sub-tomogram averaging to improve the resolution of the nucleosomes. They aligned together over 6000 nucleosomes and averaged them to resolve their structure, which allowed them to distinguish between histone proteins and DNA in the complex. At that point they realised they were working with two populations of nucleosomes, one of which contained an extra histone called linker histone 1 (H1) on the side of the spool that was connected to a linker DNA.
82 percent of the nucleosomes carried H1, and they reran the averaging on this subset to resolve the structure. They realised they could superimpose the averaged structure onto the original nucleosomes in the tomogram and determine the orientations of neighbouring nucleosomes connected by DNA linkers. By measuring the angles and distances between the nucleosomes, the team found that they adopted the zigzag model but didn’t adhere to it strictly. The researchers didn’t observe any evidence for the solenoid model, but Zhang couldn’t rule out its existence, suggesting that it might exist under other conditions or in other cell types.
This was the first study to demonstrate that cryoET of thin lamellae could be used to observe chromatin organisation under physiologically relevant conditions. Next, the researchers aim to use this technique to study the structure of chromatin at the ends of chromosomes called telomeres, which shorten with age. They would also like to explore how the structure of chromatin changes in different diseases like cancer and how this affects processes like gene expression or the integration of virus DNA into the genome.
So far, the team showed that chromatin adopts a more variable structure than the models suggest, leaving much to unravel about the nucleus’ space-saving strategy. Zhang concluded;
This study not only changes the textbook picture but opens a new avenue for investigating all the DNA associated processes.
To find out more about eBIC or discuss potential applications, please contact eBIC director Peijun Zhang: [email protected].
Hou Z, Nightingale F, Zhu Y, MacGregor-Chatwin C and Zhang P. Structure of native chromatin fibres revealed by Cryo-ET in situ. Nat Commun 14, 6324 (2023). doi: 10.1038/s41467-023-42072-1
Access to the cryo-EM facilities at eBIC was funded by the Wellcome Trust, MRC, and BBSRC. This research was supported by the UK Wellcome Trust Investigator Award (206422/Z/17/Z), the National Institutes of Health grants AI150481, the ERC AdG grant (101021133), and the Wellcome Trust Core Award Grant Number 203141/Z/16/Z with additional support from the NIHR Oxford BRC.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.