Understanding how strontium-90 interacts with uranium in spent nuclear fuel storage
Nov 30, 2022
Nov 30, 2022
Humanity has been using nuclear materials for research and power generation for 60 years, creating a considerable amount of radioactive waste that has to be safely stored for generations to come. While spent nuclear fuel is predominantly uranium, it contains other elements that can interact physically and chemically. The radioactive isotope strontium-90, produced during nuclear fission, significantly contributes to radioactivity in spent nuclear fuel. Although both uranium and strontium-90 will be present wherever spent nuclear fuel has been processed and stored, the interactions between these two elements are not well understood. In work recently published in Langmuir, a team of researchers investigated how strontium-90 reacted with uranium over a wide pH range. Their results show that the interactions are complex and vary according to pH, pertinent information for the safe long-term storage and management of spent nuclear fuel.
The legacy ponds and silos at the Sellafield nuclear facility are among the most hazardous nuclear facilities in the UK. Although the ponds are maintained at a pH of 11.5 to minimise fuel corrosion, historical issues have allowed extensive corrosion of the nuclear fuel stored there. In addition, silicate in the pond walls has dissolved into the pond water. The result is corroded Magnox sludge (CMS), a complex mix in which uranium can form uranium dioxide. As many other types of nuclear fuel incorporate uranium dioxide, it is abundant in most spent fuel storage scenarios. Previous research has shown that corroding uranium can form mobile particles called colloids, affecting the mobility of radioactive material such as strontium-90 that can bind to these colloids during wastewater treatment. Understanding the interactions between strontium-90 and uranium dioxide and uranium(IV)−silicate colloids is, therefore, extremely important in spent fuel storage ponds and radioactive effluent treatment systems.
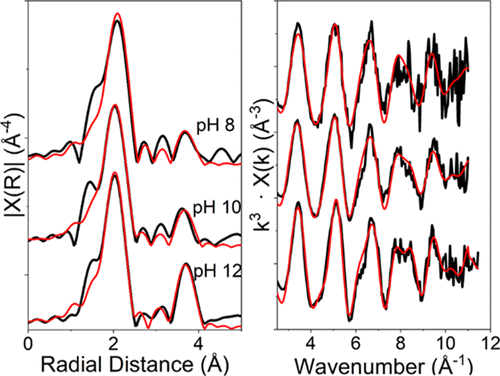
To address the lack of information on how strontium-90 interacts with colloids, researchers from the University of Manchester, the National Nuclear Laboratory, Pacific Northwest National Laboratory and Sellafield Ltd investigated the sorption (absorption and adsorption) of strontium onto uranium nanoparticles across a wide pH range.
They used a combination of investigative techniques, including ultrafiltration to assess the extent of absorption. Combining acid leaching experiments with transmission electron microscopy (TEM) gave them further information on the strontium-uranium interactions. The team then brought some of their samples to Diamond's B18 to perform Extended X-ray Absorption Fine Structure (EXAFS). This synchrotron spectroscopy technique allowed them to probe the mechanisms of strontium sorption.
Their results showed that uranium dioxide and uranium(IV)−silicates demonstrate elevated strontium sorption under conditions relevant to radioactive waste disposal, the storage of spent nuclear fuel and contaminated land scenarios. Furthermore, the effect of uranium(IV)−silicates on strontium mobility is dependent on pH, and understanding these complex strontium-uranium interactions is essential for predicting the behaviour of strontium-90 in nuclear waste storage environments.
To find out more about the B18 beamline or discuss potential applications, please contact Principal Beamline Scientist Diego Gianolio: [email protected].
Neill TS et al. Sorption of Strontium to Uraninite and Uranium (IV)–Silicate Nanoparticles. Langmuir 38.10 (2022): 3090-3097. DOI:10.1021/acs.langmuir.1c02927.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.