Researchers today are striving to make molecular movies, where they can accurately record sequential images of molecules interacting to help us understand their chemistry. This has been a holy grail in the biochemistry field for several decades and advances in facilities like X-ray free electron lasers (XFELs) are making that a reality.
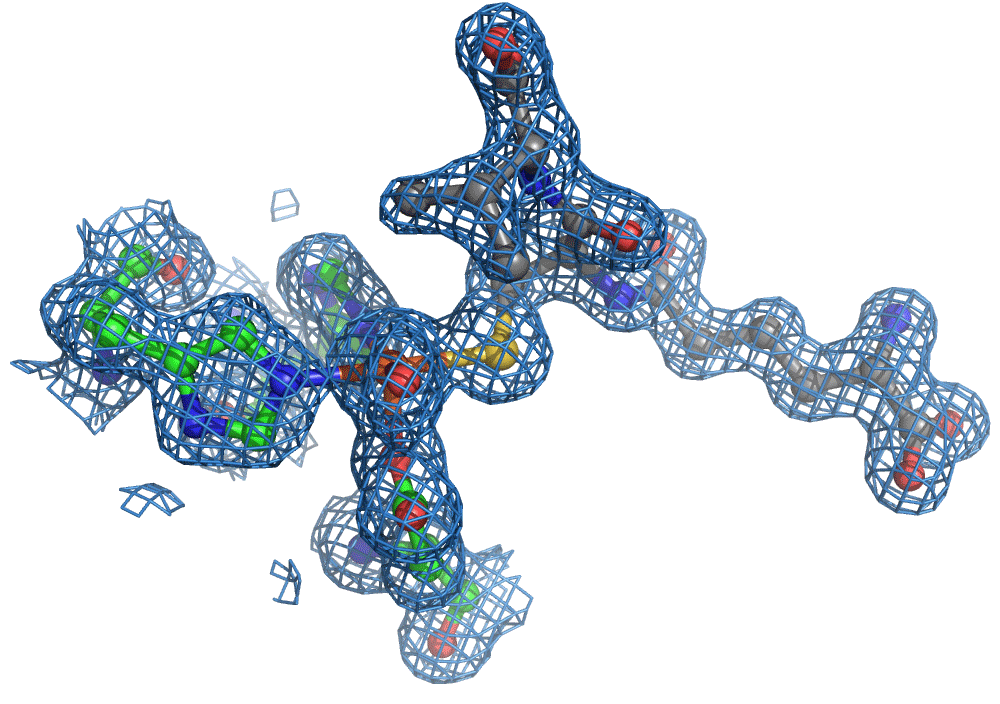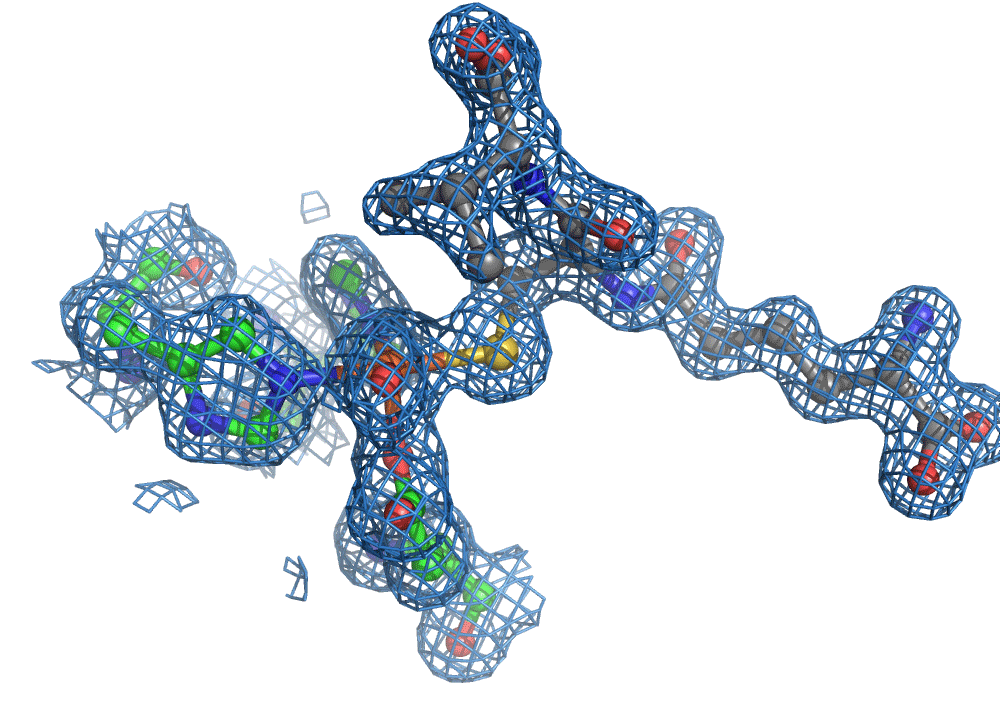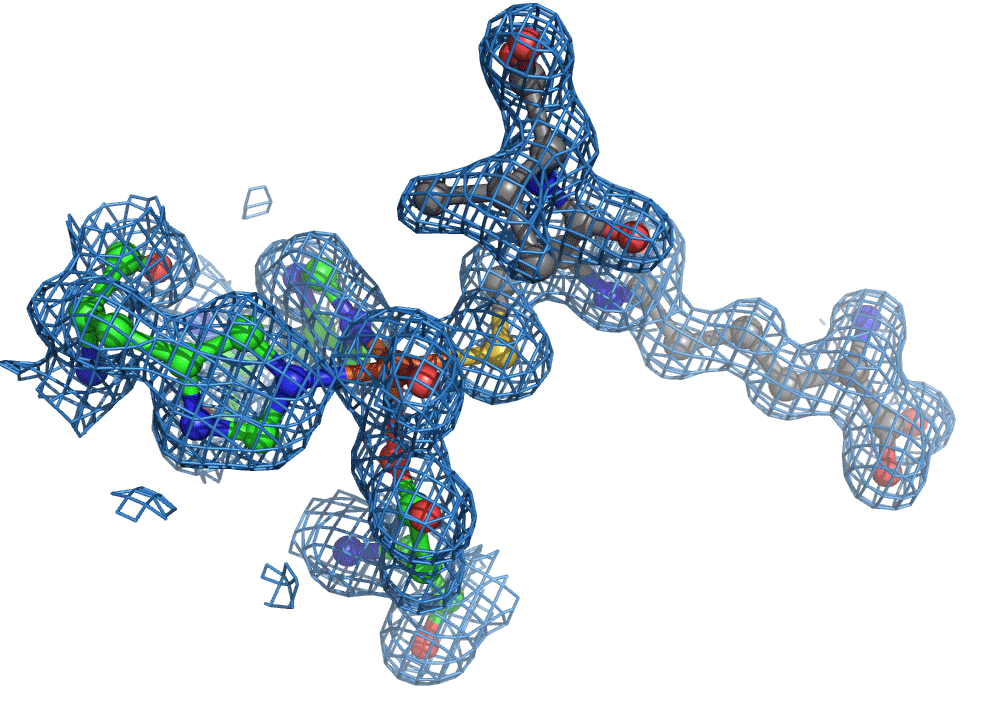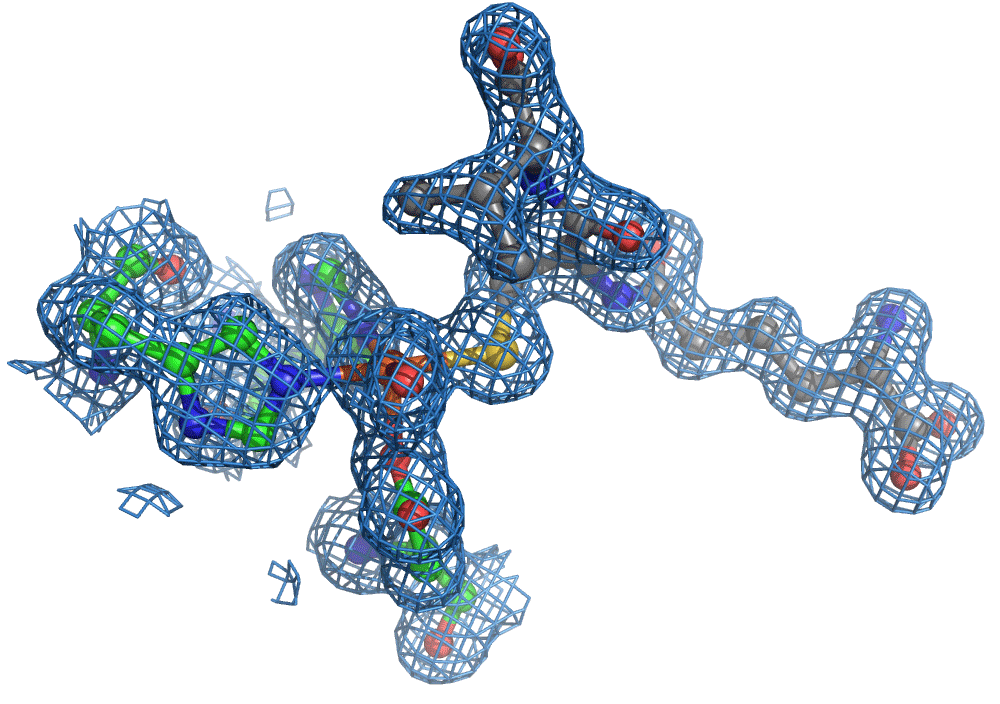

One of the most common methods for visualising molecules in 3D is crystallography. Traditionally, ‘large’ crystals (~ 100s of microns on one or more sides) were produced. These contain millions of copies of the molecule of interest arranged in a regular lattice. They are typically frozen and imaged using traditional X-ray crystallography methods. Now, thanks to the bright and highly focused light from modern XFELs, researchers can look at crystals more than more than one to two orders of magnitude smaller, which is great for making molecular movies of chemical reactions. The reason for this is that to make a movie of a chemical reaction, you need all of the enzymes and reagents (the actors) to be in the right place at the right time when the director shouts action (X-ray pulse), that is in the case of an XFEL, when a very short X-ray pulse (ten of femtoseconds, e.g. 10-15 s) shines on the sample. The problem with large crystals used most commonly is synchronisation … enzyme molecules located in the interior of the crystal will start their reactions later because substrate diffusion from the outside takes longer compared to those enzyme molecules nearer the surface.
The sharpest time-resolved crystallography movies require all of the enzyme molecules to react in unison. The answer to the problem is to use smaller crystals, because indeed, substrates can be more easily mixed with the microcrystal catalyst to ensure the reaction of each enzyme has all started in unison with all the others within the lattice, and aged the same amount of time, before the crystal is exposed to the X-ray pulse. The ultrashort pulse is so intense that each sample is destroyed, but only after the data is safely recorded. As a consequence, the whole dataset is built-up serially from many thousands of exposures. A time-resolved movie is created by collecting datasets at several timepoints and arranging them like a stop-motion animation. It’s critically important to mix the substrate and the catalyst rapidly and evenly before they are imaged. This will allow the researchers to properly synchronise the reaction and select different delay time points spanning the whole reaction cycle. But to collect this much data also requires very sample efficient methods, which has been a challenge. Indeed, enzymes and substrates can be very scarce and/or expensive, which has very often limited potential applications, especially if sample delivery is not as efficient as possible.
High magnification and fast camera video of the drop-on-drop ligand addition strategy prior to the X-ray interaction region enables tr-SFX. The 37500 frames per second (FPS) video on the left shows ~ 3 nL Lysozyme microcrystal slurry on the tape to which 7 approximately 60 pL drops of N-acetylglucosamine ligand are added at 350 Hz. The 45000 FPS video on the right shows ~ 3 nL AmpC (a serine-Β-lactamase) microcrystal slurry to which 50 approximately 60 pL drops of the antimicrobial compound ertapenem are added at 4500 Hz.
An international research collaboration led by the XFEL Hub at Diamond Light Source, Lawrence Berkeley National Lab in the USA, the University of Oxford, and the new Ineos Oxford institute for Antimicrobial Research, developed a novel system for delivering their crystals on-demand into the XFEL beams and tested it at XFELs in Japan and the USA. They used an acoustic droplet injector to deliver nanolitre droplets of a microcrystal enzyme slurry onto a small tape that transported the crystals to the X-ray beam. In order to add the substrate and start the reaction, they then “blanket bombed” their nanolitre slurry droplets with much smaller, picolitre droplets of the substrate.
The benefit of this inventive approach was that the turbulence caused by many smaller drops landing on the larger droplet caused a lot of turbulence in the liquid, meaning that the enzyme and the substrate rapidly mixed. This in turn led to reactions that were well synchronised. The drop-on-drop methods are highly efficient, completely general, and potentially applicable to nearly all known enzyme reactions.
Due to COVID restrictions, the most recent set of these experiments had to be carried out remotely. The Berkeley team was responsible for releasing the larger droplets, that were timed to arrive with the XFEL X-ray pulse delivered by LCLS in Menlo Park CA, and the team releasing the smaller, reagent-containing droplets was based in Oxfordshire. The timing of the drops was so important that the three teams had to account for the speed at which the trigger signals passed from the UK to the US over the internet. This general and sample efficient method can rapidly mix and initiate reactions of almost any substrate and any concentration with any microcrystal slurry to study catalytic reactions ranging from tens of milliseconds to several seconds. This experimental envelope covers nearly all enzyme reactions.
The first portion of this study was published in Nature Communications 2021, 12 (1), 4461. doi: 10.1038/s41467-021-24757-7.
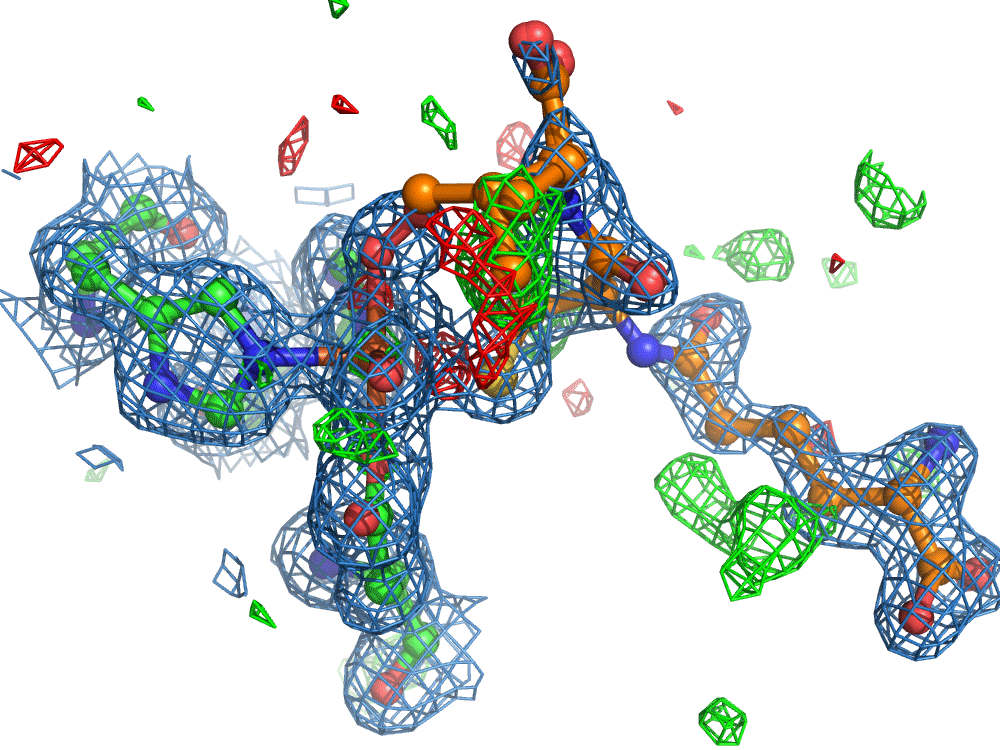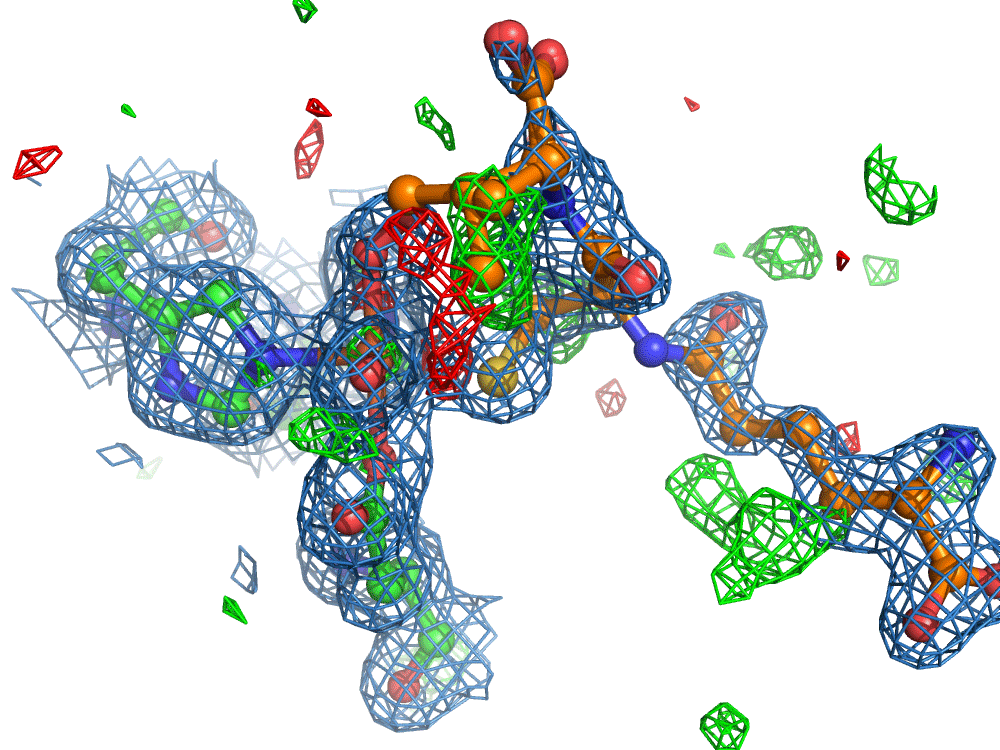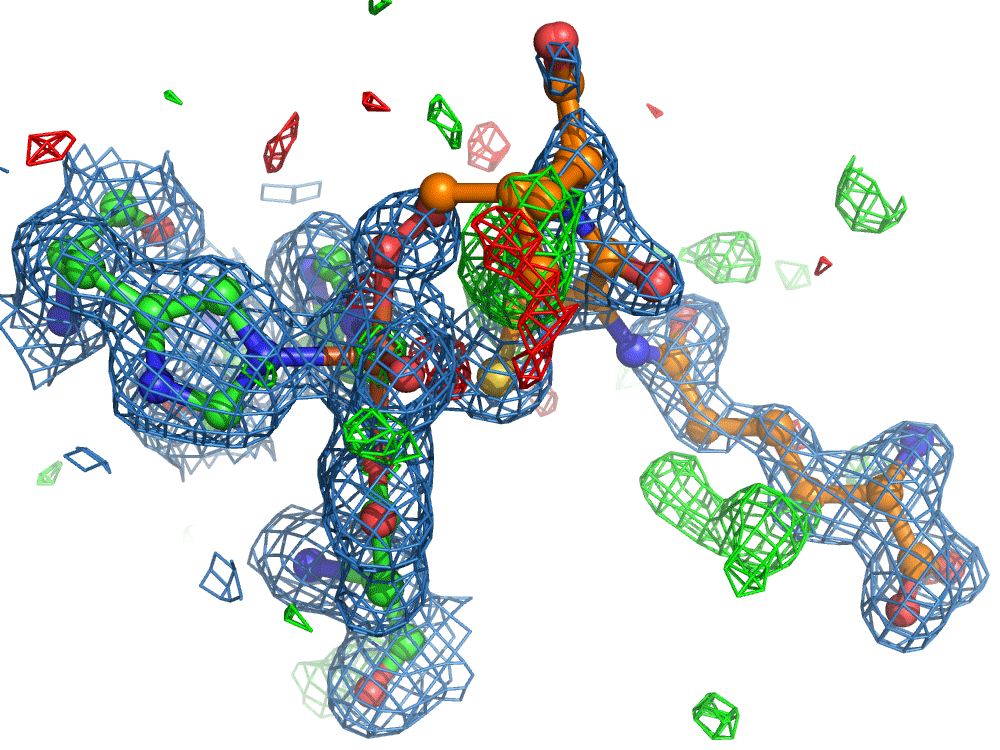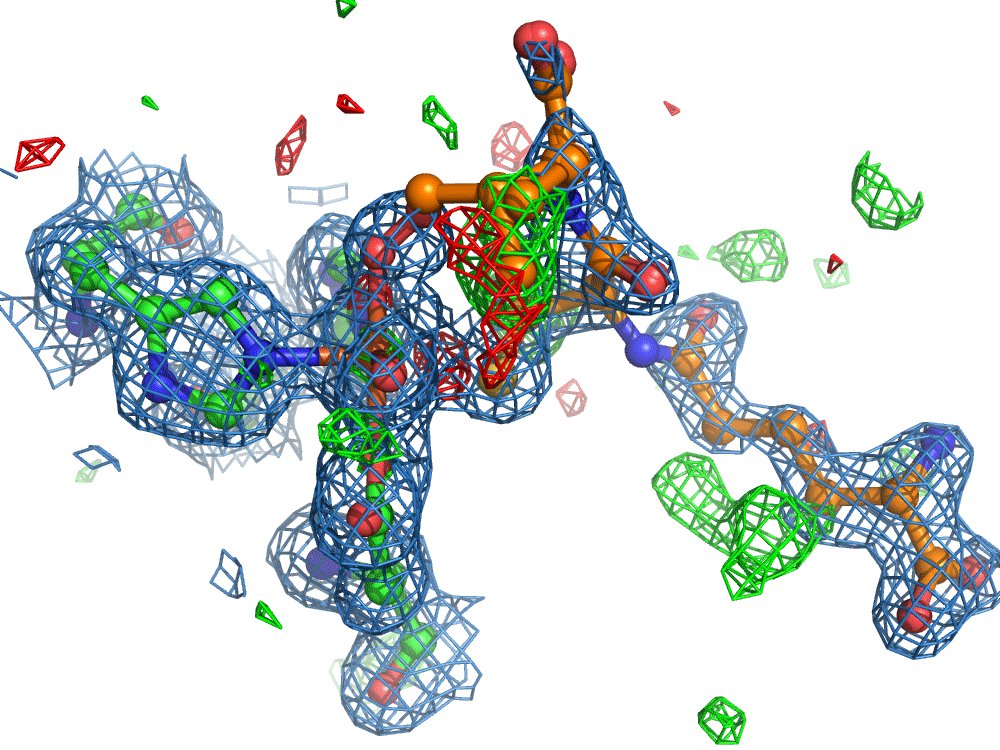
However, the researchers were not finished. They set out to use the on-demand droplet delivery system to study a natural enzyme that produces antibiotics. The catalytic events in the iron-dependent enzyme, isopenicillin N synthase, are initiated by the binding of dioxygen, i.e. O2, the oxygen we breath in the air. For this set of experiments, the researchers took out the second picoliter droplet delivery device and replaced it with an oxygen reaction chamber through which the slurry droplets passed.
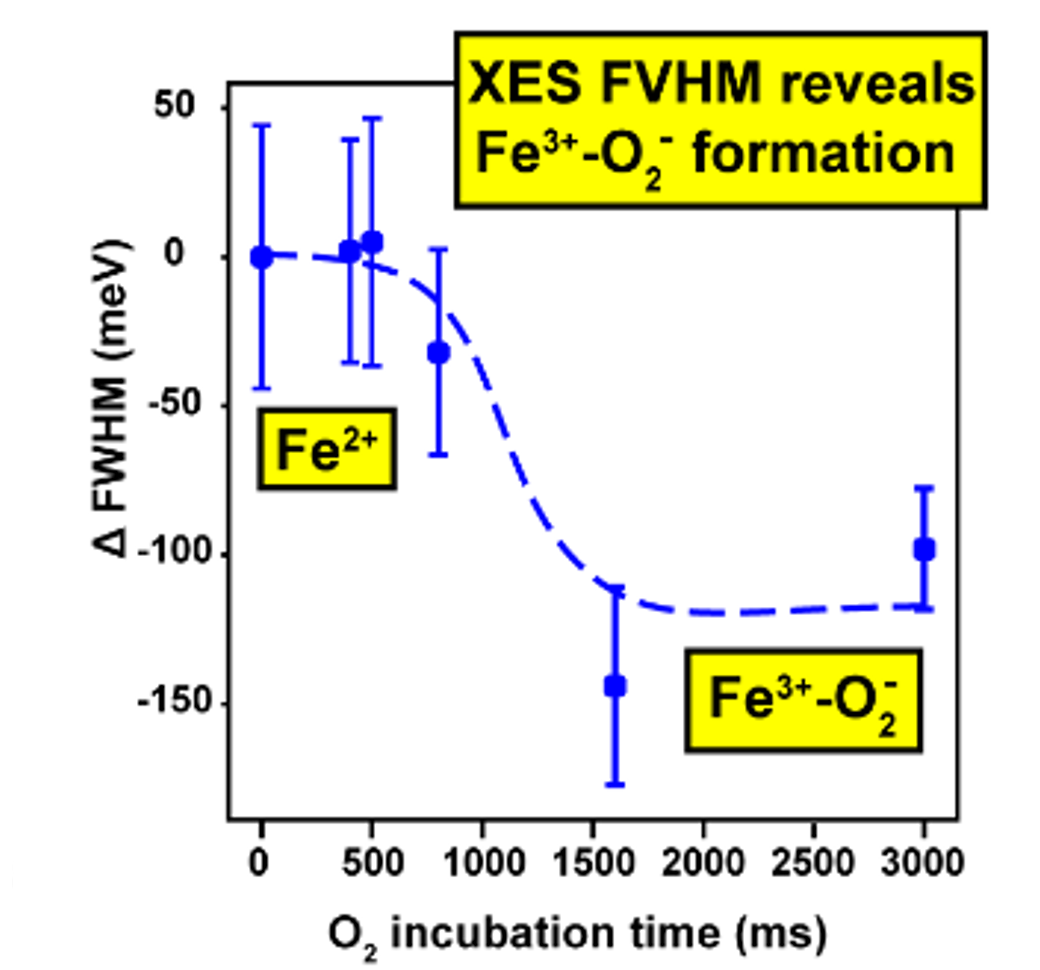
Again, there was a problem when studying oxygen reactions in biological systems using crystallography that the researchers had to overcome. Biology samples exist in water, and X-ray crystallography has a really hard time differentiating between dioxygen (O2) and water (H2O). Indeed, because X-rays detect electrons - which means that hydrogen atoms with one electron are all but invisible in crystallography - they only differ by one O atom. This means that in an X-ray crystallography experiment, the electron density map for an H2O water molecule often looks very much like an O2 dioxygen molecule and vice versa. How does one prove an interpretation of the electron density maps - especially without bias?
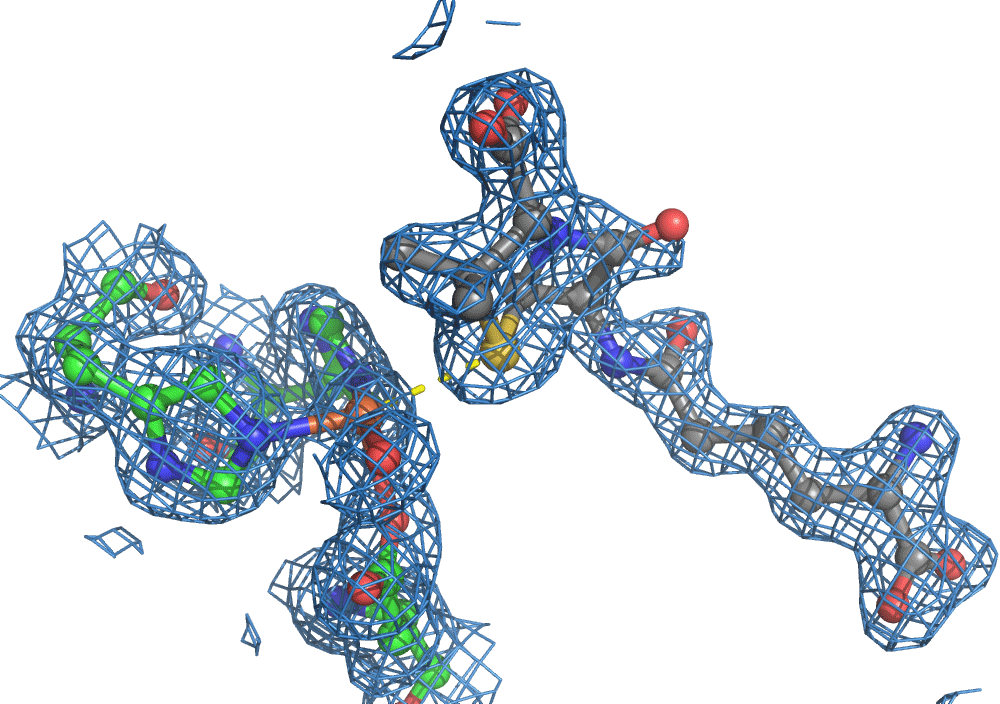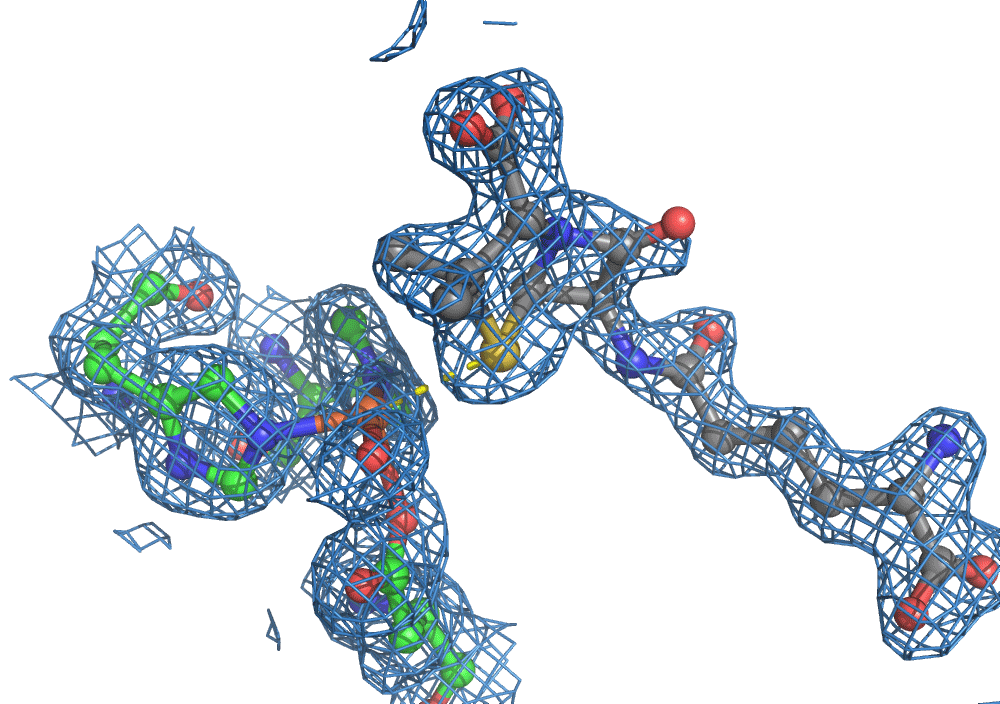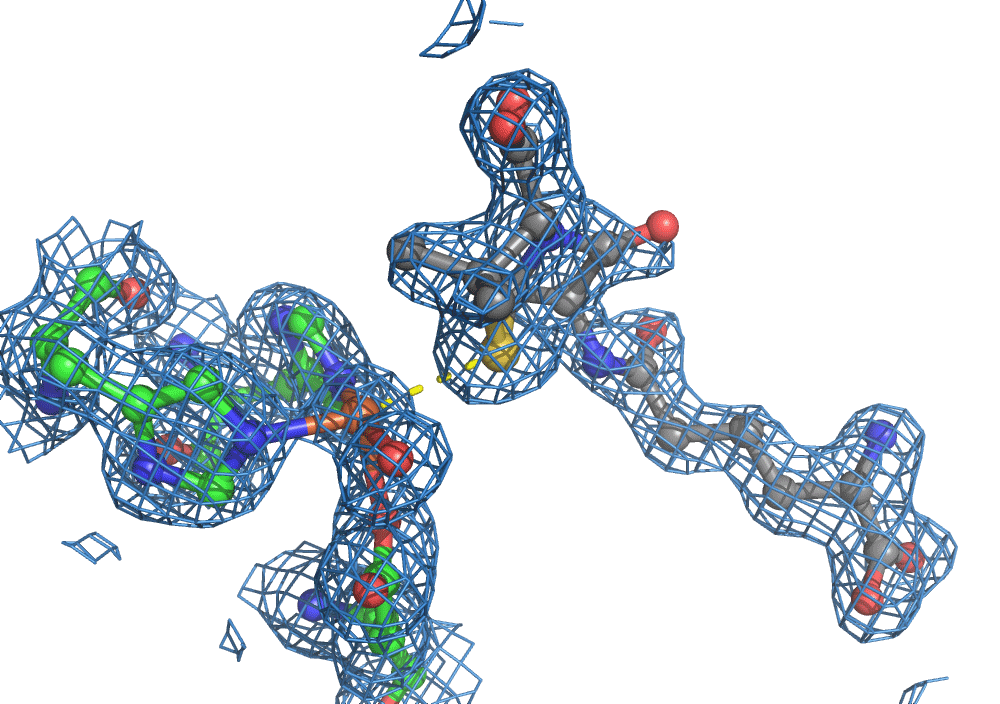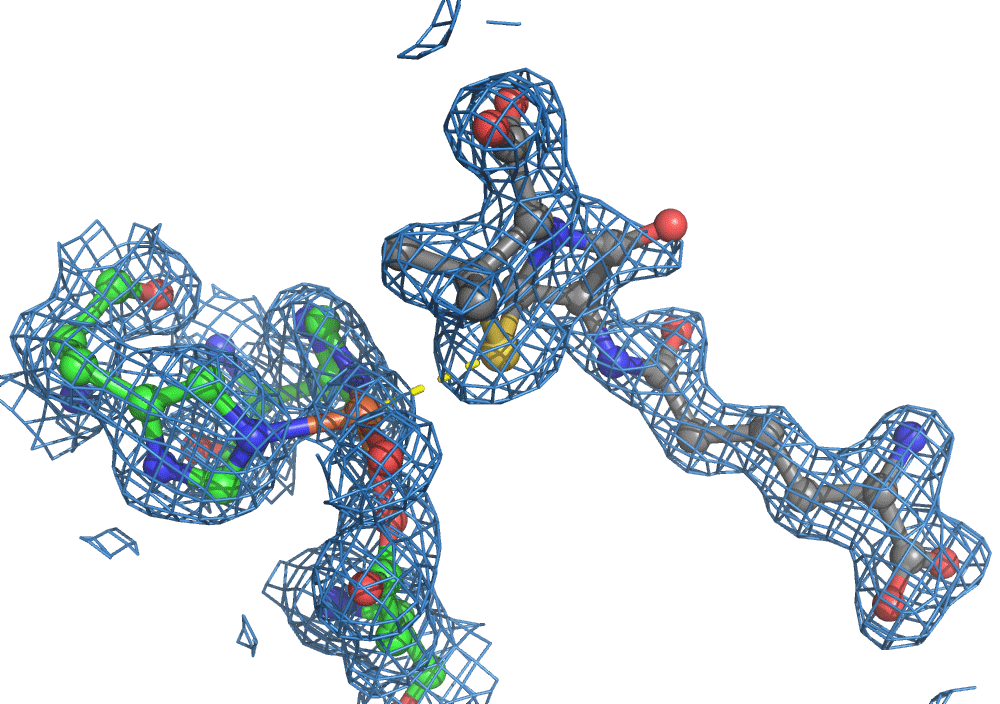
So, when you are looking at a time-resolved reaction catalysed by oxygen that is taking place in a sea of water using X-ray diffraction, you have a tough job ahead. The research team overcame this challenge by building a spectroscopy sensor at a 90° angle to their sample in the X-ray beam. The research team knew that only a small fraction of the X-ray photons that hit each crystal will produce diffraction data collected in the forward direction that can be used for building atomic resolution models of all the ordered atoms within the crystal lattice. In addition, some of the X-ray photons will produce a spectroscopic signal originating from the iron atoms within the enzyme active site and responsible for catalysis. Furthermore, the spectroscopic signal changes as the reaction with O2 progresses, but importantly does not change if H2O binds to the iron. This sensor allowed them to simultaneously collect spectra and diffraction from each microcrystal sample, which meant they could differentiate between oxygen and water by observing changes in the redox state of iron atoms. This allowed the team to propose a spectroscopically verified Fe(III)-O2 intermediate for the first time in the IPNS reaction cycle. The authors note that these results have important implications for all metalloenzymes that react with O2 to generate intermediates that ultimately catalyze some of the most chemically challenging reactions in the biosphere.
The second portion of this work was published in the journal Science Advances 2021, 7 (34), eabh0250. doi: 10.1126/sciadv.abh0250.
The two studies together present a unique and powerful set of methods for studying time-resolved chemical reactions at extremely high resolution, with complementary spectroscopic analysis, and using very efficient sample delivery strategies.
X-ray sources used in both publications include Diamond Light Source in the United Kingdom, the SPring-8 Angstrom Compact free electron LAser in Japan and the Linac Coherent Light Source at SLAC National Accelerator Laboratory in California.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.