The rapid transmission of COVID-19 has shown how important it is to be able to develop new vaccines to counter its impact in a timely manner. Currently there are many other viruses for which vaccines are urgently needed. Active Virosomes (AVs) are a promising proprietary vaccine technology for delivering drugs, metabolites and vaccines. AVs are virus derivatives, that carry selected viral components on their surface and within their cavity which can elicit an immune response.
In work recently published in the Journal of Microscopy, a company working with beamline B24 and the industrial liaison office at Diamond used a combination of X-ray and laser light imaging to investigate whether potential AV vaccines were suitably homogeneous and carried an effective load and to further test their effect on human tissue. The results demonstrate that this novel combination of microscopies can provide rapid assessment of candidate vaccine formulations.
Chikungunya and Zika are both category B priority pathogens, endemic to tropical areas of the world, but with recorded instances in temperate regions also. Spread by mosquitos, they result in infections that can be life threatening or persist and have severe long-term implications to the quality of life for patients. With no current drug treatments or approved vaccines available, the development of a relevant vaccination strategy is an urgent world health requirement.
There are many stages in the development of a new vaccine, including initial R&D, pre-clinical studies, clinical trials, regulatory review and approval, pilot and scale-up manufacturing, quality control and comprehensive register of adverse effects.
Developing and characterising vaccines in the pre-clinical stages involves using primarily biochemical techniques that seek to understand the molecular interactions involved.
What's missing from these traditional profiling approaches is the ability to provide high-resolution quantitative imaging on the native-state of vaccines in their primary formulation and their potential effect on the ultrastructure of human cells. To investigate these aspects, high-throughput correlative 3D imaging of candidate vaccines such as AVs in host cell populations under near-physiological conditions is essential.
Researchers brought samples of AV vaccine formulations for Chikungunya and Zika viruses to Diamond's correlative cryo-imaging beamline B24 to take advantage of a novel combination of 3D imaging techniques which lead to the assessment of AV batch homogeneity, antigen load and their effect on living human cells.
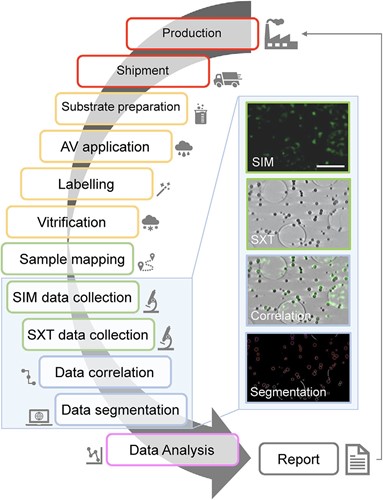
Beamline B24 is a unique correlative cryo-imaging beamline offering a 3D fluorescence microscope that delivers imaging beyond the diffraction limit of light (cryo-Structured Illumination or cryo-SIM) alongside 3D X-ray imaging for cells using a technique much like computerised tomography (cryo Soft X-ray Tomography or cryo-SXT). The B24's cryo-SIM microscope is a working prototype and the only such facility currently accessible on-demand to users internationally. The B24 X-ray microscope on the other hand offers near-physiological 3D cell imaging using a technique that is currently available at only a handful of facilities around the world.
All samples at B24 are cryogenically prepared. The advantage of bringing samples to cryogenic temperatures through flash-freezing is that we capture cells in an instant in their life much like taking a snapshot. This allows us to preserve their true physiological state at that moment and see how they have responded to environmental stressors such as chemicals and vaccines. It also makes samples robust enough to be transferred across to different microscopes allowing us to image them with different light (laser or X-rays) and therefore explore more aspects of their physiology.
For this research, the team used 3D fluorescence imaging to localise viral components decorating the surface of AVs and quantify the viral protein load within different vaccine batches. 3D X-ray imaging then supplied complementary information on the size and shape of each AV formulation and individual AVs within those formulations. Beyond sample characterisation these methods were used to look into human tissue cultures that were exposed to AVs and assess their responses as well as their uptake and secondary expression of antigens (AV technology is used to deliver partial viral genomes which then turn the recipient cells into antigen factories without the adverse effects of an actual infection).
New vaccines must be safe, specific, and reproducible. This work provides evidence for a new robust way of quickly and efficiently assessing and validating potential vaccine formulations during both R&D and production. This reliable and robust high-resolution 3D imaging regime evaluates the microstructure and biochemistry of novel vaccine formulations such as AVs and their effect on host cells.
To find out more about the B24 beamline or discuss potential applications, please contact Principal Beamline Scientist Maria Harkiolaki: maria.harkiolaki@diamond.ac.uk.
Okolo C et al. Correlative imaging using super‐resolution fluorescence microscopy and soft X‐ray tomography at cryogenic temperatures provides a new way to assess virosome solutions for vaccine development. Journal of Microscopy (2021). DOI:10.1111/jmi.13054.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.