Modern life relies on magnetism, from electric motors and generators to digital storage methods. The best magnets include rare earth elements, which are uncommon, costly and toxic. It would be beneficial to just use cheap and abundant transition metals, but their magnetism is not as strong without the presence of rare earth elements. In work recently published in Chemical Science, an international team of researchers used a variety of synchrotron X-ray techniques to characterise iron-doped lithium nitride, a novel material in which iron atoms exhibit magnetism as if they were a rare earth element. A full understanding of its structure could lead to the development of transition metal magnets, high-density digital storage, and advances in quantum computing.
With applications including electric motors and generators, strong magnets are extremely useful. However, they usually require the inclusion of neodymium or one of the other rare earth elements to exhibit permanence, the ability to keep its magnetic moment fixed (also called magnetic anisotropy). As those elements are uncommon and found only in specific geographic locations, they are expensive. They are also toxic.
Transition metals are far more abundant and less toxic. Although they carry spin (due to unpaired electrons in their outer shell), their magnetic moments and magnetic anisotropy are not usually as strong as those of rare earth elements. Would it be possible to create a strong magnet using only transition metals?
Nine years ago, Dr Anton Jesche happened upon a novel material that formed large single crystals: lithium nitride doped with iron. Unexpectedly the iron atoms within lithium nitride seemed to behave like rare-earth ions with large magnetic anisotropy. Although this effect does not occur at room temperature, it works below 50 K and very well below 10 K, a temperature that can be reached in the lab using liquid helium.
However, this unexpected magnetic behaviour could not be explained, so to understand the material better, Dr Jesche collaborated with Dr Michael Baker at the University of Manchester. Dr Baker is part of The University of Manchester at Harwell, with extensive experience in using synchrotron techniques to analyse materials.
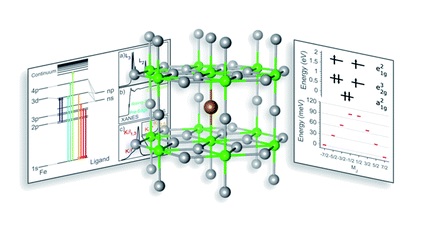
Because the iron atoms in this material are dopants, they are randomly distributed throughout the material. They cannot be examined using X-ray diffraction techniques (which require a regular structure). However, other synchrotron X-ray techniques are element specific and can be tuned to the iron atoms in the sample.
During a series of experiments at two Diamond beamlines (I10 BLADE and I20 Scanning), Dr Baker and PhD student Myron Huzan analysed the novel material using extended X-ray absorption fine structure (EXAFS), X-ray absorption near edge spectroscopy (XANES), Kβ X-ray emission spectroscopy (XES) and L2,3-edge X-ray absorption spectroscopy (XAS) single-crystal measurements.
Each technique tells us something different about the sample. EXAFS is used to examine the geometric structure of non-crystalline materials. In this case, it showed a very unusual bond length. XANES added the coordination geometry and bond angles, and XAS gave a detailed understanding of 3d electronic structure, which is directly related to the magnetic properties.
The team used XES to answer concerns from a theory study whose results showed that the structure changed depending on the iron concentration. XES is a very sensitive probe of unpaired electrons in the 3d shell and showed that the oxidation state does not change with iron concentration.
The results from these experiments have answered a lot of questions about the material. We now know how its short linear N-Fe-N bonding relates to its electronic structure and resultant large magnetic anisotropy.
By performing temperature dependent L-edge XAS measurements between 4.5 and 400 K (-269° to 127° C) it was possible to characterise the magnetic anisotropy barrier to understand the mechanism by which the magnetic poles flip from one state to another.
It is understood that applying a small magnetic field to iron doped lithium nitride causes dramatic changes of/to the anisotropy barrier. Depending on the orientation of the field, it can make it easier, or much harder, to flip the magnetic poles. As this is tuning the quantisation state, it opens up the potential to use this material for quantum computing. There is also the potential for using this material for high-density magnetic data storage.
Dr Baker, concludes:
This material is not suitable for industrial applications, because it requires a low temperature. However, we may be able to use our new understanding of its structure to design transition metal magnets that work at room temperature. And that would allow us to move away from expensive and toxic rare earth elements.
The team have already studied a series of similar materials. Dr Jesche made analogues doped with magnesium, cobalt, nickel and copper rather than iron. Their magnetic properties are very different, but analysing their structures should help explain why iron-doped lithium nitride is so special.
To find out more about the I10 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Paul Steadman: paul.steadman@diamond.ac.uk. The Principal Beamline Scientist for I20 is Fred Mosselmans: fred.mosselmans@diamond.ac.uk.
Find out more about the eBIC: Electron Bio-Imaging Centre at Diamond.
Huzan M et al. Single-Ion Magnetism in the Extended Solid-State: Insights from X-ray Absorption and Emission Spectroscopy. Chemical Science 11, 11801-11810 (2020). DOI:10.1039/D0SC03787G.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.