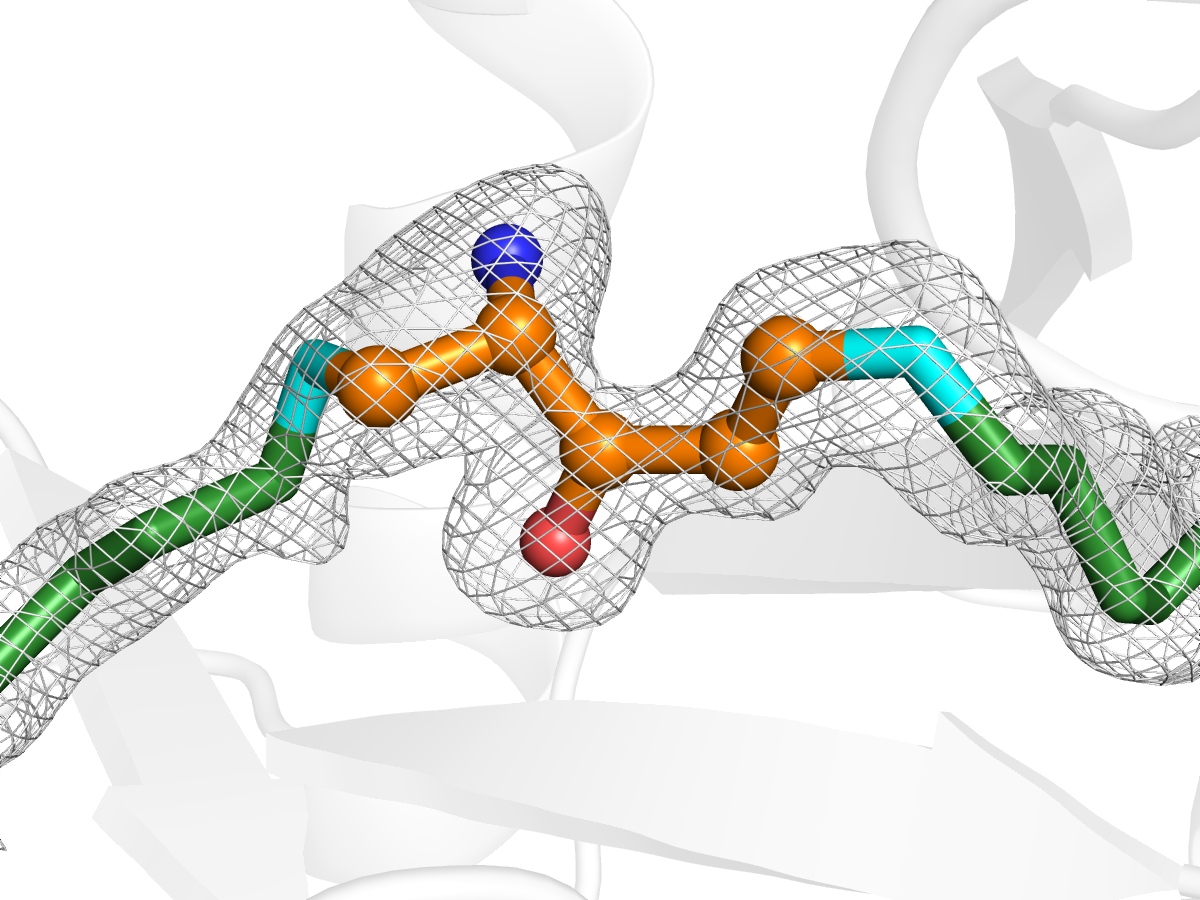
A paper published in Nature Chemical Biology is the culmination of more than 10 years of research, for a team led by Dr Ivo Tews of Southampton University, into the structure of proteins involved in the biosynthesis of vitamin B6 in plants and bacteria. Pyridoxal 5′-phosphate (PLP) is an active form of vitamin B6; the PLP-synthase complex, which consists of the enzymes Pdx1 and Pdx2, is found in all domains of life. The published work focuses on a Pdx1 enzyme from Arabidopsis thaliana, and furthers our understanding of how the protein catalyses the synthesis of vitamin B6. X-ray crystallography and spectroscopy allowed the discovery of previously unknown intermediate stages of the process, which offers up a new target for drugs designed to combat diseases such as malaria and TB by affecting the synthesis of a vitamin that is essential for all organisms.
To find out more about the I24 beamline, or to discuss potential experimental applications, please contact Principal Beamline Scientist Dr Robin Owen: [email protected]. In order to discover more about the PhD programme at Diamond, please see here or contact [email protected].
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.