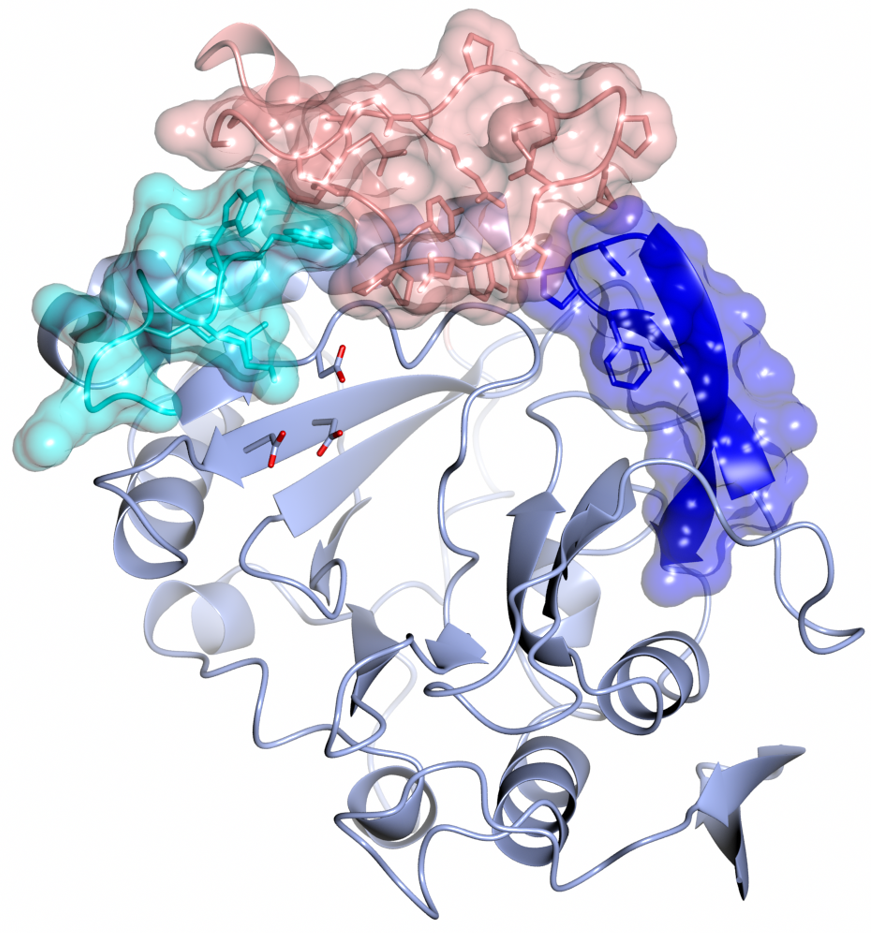
The mechanistic details of a mysterious mycobacterial DNA repair apparatus that had long eluded the scientific community have been revealed with the aid of the Macromolecular Crystallography beamline (I02) at Diamond Light Source. As recently reported in Nature Communciations, the prokaryotic Ligase C (LigC) complex was found to be involved in base excision repair pathways that fix damaged DNA.
In 2016, 1.7 million people died from tuberculosis, a notoriously difficult disease to treat, which is caused by Mycobacterium tuberculosis. These mycobacteria can lie dormant in the human body for decades, and in fact a quarter of the world’s population is believed to carry this latent form of the disease. Tackling this elusive mycobacterium is a global health goal and this might be partially assisted by targeting the machinery involved in protecting its genome from DNA damage to eliminate these pathogens.
A collaboration between the University of Sussex and the Polish Academy of Sciences set out to explore the fundamental components of the DNA repair machinery of Mycobacterium tuberculosis. The group focused their efforts on an ambiguous DNA repair enzyme called LigC and its polymerase component (Prim-PolC) to understand their cellular roles.
Using macromolecular X-ray crystallography at the I02 beamline, along with a series of in vitro and in vivo experiments, the team were able to characterise LigC and its binding partners. They discovered that LigC acts as a key coordinator of DNA excision repair in mycobacteria. Importantly, the scientists were able to knock out LigC and Prim-PolC and observed that these mycobacterial strains were more sensitive to DNA damage, which suggests that targeting such repair pathways could be an exciting new avenue for future antibiotic development.
The I02 beamline has since closed and been replaced by the fully-automated VMXi beamline, which will welcome users later this year. To find out more about the original I02 beamline or the VMXi beamline, please contact Principal Beamline Scientist Prof Thomas Sorensen: thomas.sorensen@diamond.ac.uk
Płociński P et al. DNA Ligase C and Prim-PolC participate in base excision repair in mycobacteria. Nature Comms 2017; 8: 1251.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.