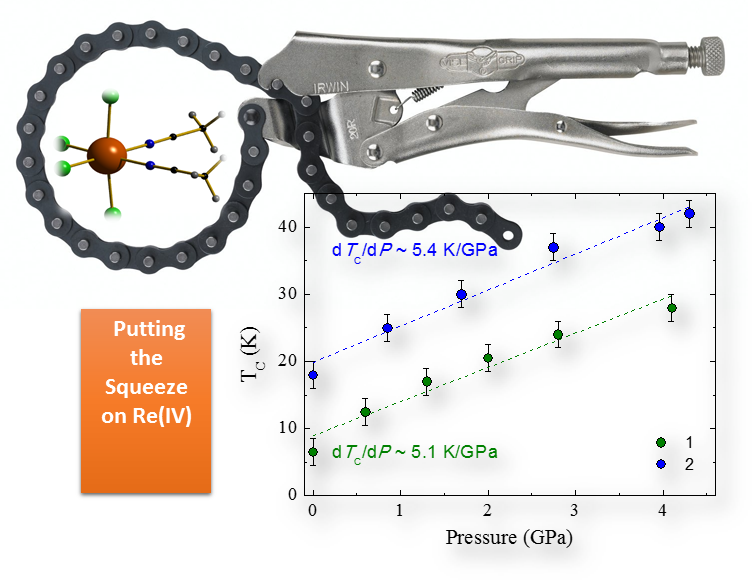
 Figure 1: The ordering temperature Tc that characterises Re(IV) compounds increases with applied pressure.
Figure 1: The ordering temperature Tc that characterises Re(IV) compounds increases with applied pressure. Long-range magnetic order under high pressure
Materials characterised by LRMO are of fundamental importance to many modern technologies for which size reduction is a prominent goal of academic and industrial research. In this context, the possibility of designing molecules that behave in the same way as bulk magnets would represent a major step towards device miniaturisation. This capability requires a thorough understanding of the connection between molecular structure, magnetic anisotropy of the molecule and observable physical properties. A collaboration between three British universities (the University of Edinburgh, the University of Glasgow and Newcastle University) and the University of Valencia in Spain has now investigated the relationship between changing molecular structure and modifications to the magnetic behaviour in mononuclear compounds of Re(IV) under increasing hydrostatic pressure. Professor Euan Brechin, at University of Edinburgh and co-author of the paper, explained: “Mononuclear compounds of Re(IV) are unusual species because they display LRMO despite being mononuclear. This is due to the presence of important intermolecular interactions. Since the strength of these interactions is determined by intermolecular distances, we predicted that squeezing the molecules closer together would increase the magnitude of the interaction and thus the temperature at which LRMO is observed.”
To find out more about the I19 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Dr Dave Allan: [email protected].
Woodall CH et al. Pressure induced enhancement of the magnetic ordering temperature in rhenium(IV) monomers. Nature Communications 7, 13870 (2016). DOI:10.1038/ncomms13870.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.