Recycling has become commonplace in our everyday life, and is evident in reduction in the consumption of resources, saving energy and minimising waste. Similarly, cells constantly recycle proteins and lipids, with a direct impact on nutrient uptake, re-sensitisation to environmental signals, immune surveillance and waste management. Retromer is a multiprotein complex that recycles cargo proteins from endosomes to the trans-Golgi network and the plasma membrane. Defects in retromer impair various cellular processes and underlie some forms of Alzheimer’s and Parkinson’s diseases. The mechanisms for the recruitment of retromer to endosomal membranes and cargo recognition have remained elusive. Diamond Light Source’s Macromolecular Crystallography (MX) beamlines I02, I03, and the solution state High Throughput Small-Angle X-ray Scattering beamline (B21) were employed in this research. They revealed the process of recruitment of retromer complex to endosomal membranes and selection of cargo proteins into transport carriers. The results demonstrate how signal-recognition is mechanistically coupled to membrane recruitment. Thus, retromer is simultaneously both the adaptor and the scaffolding complex for sorting cargo into tubular carriers.
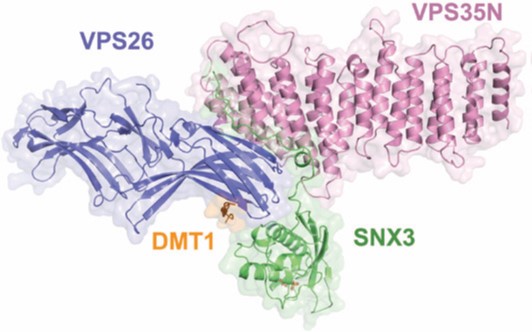
Endosomes are key intracellular recycling compartments where the biosynthetic and endocytic pathways intersect. Here, the fate of sorting transmembrane proteins, such as ion channels, transporters, enzymes and receptors, often referred to as cargo, is directly linked to their selective recruitment into tubular-vesicular carriers. The carriers are formed by unique protein coat complexes and regulatory factors that recognise signals in the cytosolic domains of the cargo proteins in order to ensure their packaging while contributing to local deformation of the membrane. One of the key components of this endosomal protein sorting machinery is the retromer complex. Retromer is composed of a highly conserved core of the Vacuolar Protein Sorting (VPS) proteins VPS26, VPS29 and VPS35, and a membrane targeting subcomplex of mono or hetero dimeric sorting nexins (SNXs) that binds to phosphoinositide lipids. While the initial model for cargo recognition pointed towards a direct interaction of the canonical ØX(L/M) recycling motif (where Ø is an aromatic amino acid), with the VPS subcomplex1, recent data have shown additional retromer dependent cargos that associate with distinct domains of the SNX component2. Taking into account diverse functions ascribed to retromer subunits and the precedents set by other coat complexes, the existence of multiple retromer complexes has been hypothesised. These might be formed by the VPS complex as a central core and distinct adaptors, such as SNXs, to allow the recycling of specific cargo through different trafficking pathways3.
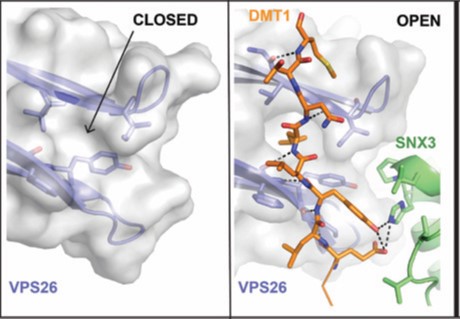
For better understanding of how cargo is recognized by retromer, a four-subunit complex comprising VPS26, the N-terminal portion of VPS35 (VPS35N), SNX3 and a canonical recycling signal from the divalent metal transporter 1 isoform II (DMT1-II) was crystallized. The structure was solved by molecular replacement using individual structures as separate searching models (Fig. 1). Structure-based single and double methionine mutants used as selenomethionine markers were particularly useful to unambiguously confirm the binding mode of the DMT1-II recycling signal. The binding site of DMT1-II is located at the interface between VPS26 and SNX3 (Fig. 2). The crystal structure shows the involvement of an extensive network of hydrogen bonds and hydrophobic interactions, engaging both SNX3 and VPS26 in the recognition of canonical recycling signals (Fig. 2). Isothermal titration calorimetry experiments confirmed that DMT1-II recognition involves binding to both retromer and SNX3. Besides, the crystal structure reveals that association of SNX3 is coupled with a closed-to-open conformational change in the cargo binding pocket of VPS26 that favors the interaction with the canonical recycling motif. These findings are in support of the fact that cargo recognition is concomitant with SNX3 binding to retromer.
Even though the crystallographic structure of VPS29-VPS35C was known4 and the VPS26-VPS35N structure was determined in this work, the picture of full-length retromer was missing. In order to address this question, inline Size-Exclusion Chromatography (SEC) coupled to Small Angle X-ray Scattering (SAXS) experiments were performed on the High Throughput Small-Angle X-ray Scattering beamline (B21). The model obtained for retromer in solution has an elongated and slightly curved shape (Fig. 3a). Rigid body refinement on the bead model, using the crystal structures of VPS26-VPS35N and VPS29-VPS35C, define the structure of retromer with VPS26 and VPS29 bound to opposite ends of VPS35. These results provide a picture of the entire retromer architecture at molecular resolution. Further analysis of retromer at different salt concentrations performed with size-exclusion chromatography, coupled to multiangle light scattering (SEC-MALS) at B21, revealed that retromer forms dimers at physiological ionic strength. SAXS analysis of the dimer revealed an inverted U-shaped architecture (Figure 3b,c). However, modelling a dimeric retromer was ambiguous considering the fact that the monomer could be fitted in two different orientations within the SAXS envelope. To solve this puzzle, maltose-binding protein (MBP)-tagged versions of the retromer complex in which MBP was fused to VPS26 or VPS29, were produced and measured by SAXS. The MBP tags were located using the multiphase modelling program MONSA5. The position of MBP clearly indicated that dimerisation occurs through the VPS29 side of retromer (Fig. 3b-e). In addition, the dimeric structure of retromer with the VPS26 subunits at the distal ends is compatible with SNX3 binding in an orientation parallel to the membrane plane, and is suitable for SNX3 binding to phosphatidylinositol 3-phosphate (Figure 3f). In this regard, it is worth noting that the formation of a coated carrier for cargo-sorting from endosomes would require retromer oligomerization on the membrane, where self-dimerization might represent the minimal repetitive assembly.
Together, these results suggest a model in which retromer recruitment to the endosomal membranes is coupled with cargo selection. This mechanism reveals a basic concept; the recycling machinery is brought to work by the cargo. Nonetheless, an exciting ‘‘Cryptex code’’ emerges from the structural analyses, where combinatorial retromer-SNXs interactions might reshape the cargo binding pocket to discriminate between different cargos. The challenge resides now in the structural analysis of other SNXs-retromer combinations with different cargos.
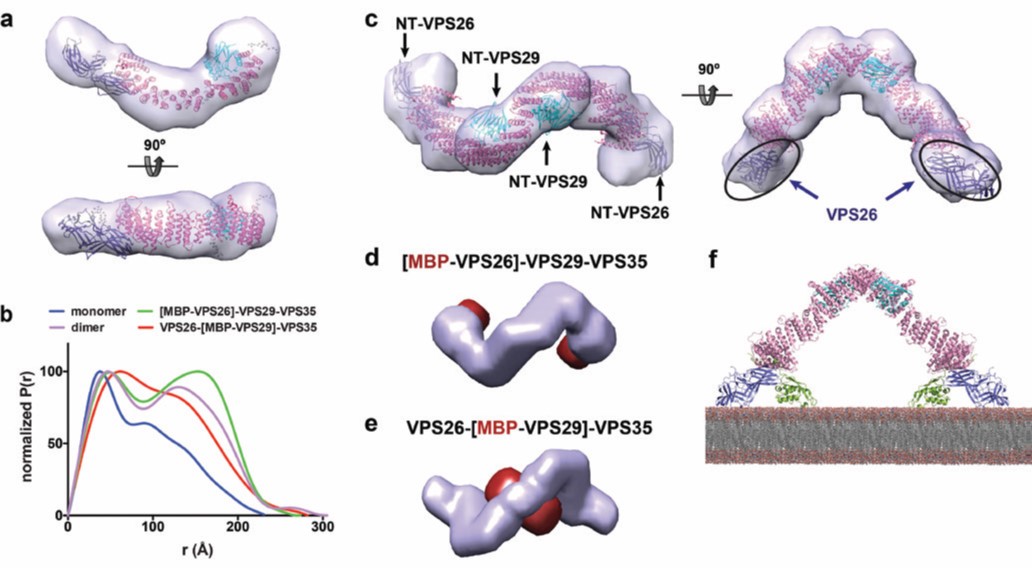
Figure 3: Structure of the retromer complex in solution; (a) ab initio shape reconstruction of monomeric retromer by DAMMIN; (b) Normalized P(r) distance distribution function of the full retromer complex VPS26-VPS29-VPS35 (monomer and dimer), the N-terminal MBPtagged constructs [MBP-VPS26]-VPS29-VPS35 and VPS26-[MBP-VPS29]-VPS35; (c) ab initio shape reconstruction of dimeric retromer by DAMMIN, showing the fit with the crystallographic structures of VPS26-VPS35N and VPS29-VPS35C (PDB codes: 5F0L and 2R17); (d-e) Multiphase envelopes calculated by MONSA in surface representation indicating the calculated phase for (d) MBP(red)-tagged VPS26, and (e) MBP(red)-tagged VPS29; (f) Retromer dimer bound to two SNX3 molecules sits parallel to the membrane plane.
References:
Funding acknowledgement:
This work was supported by the Carlos III Health Institute grant PI11/00121, the Basque Government grant PI2011-26, the Spanish Ministry of Economy and Competitiveness Grant BFU2014-59759-R, the European Community’s Seventh Framework Programme (FP7/2007-2013) under BioStruct-X (#283570), and the Horizon 2020 programme iNEXT (H2020 project #653706).
Corresponding authors:
Dr María Lucas, Dr Adriana L. Rojas and Dr Aitor Hierro, CIC bioGUNE, ahierro@cicbiogune
Related publication:
Lucas M, Gershlick DC, Vidaurrazaga A, Rojas AL, Bonifacino JS, Hierro A. Structural Mechanism for Cargo Recognition by the Retromer Complex. Cell 167(6), 1623-1635, doi:10.1016/j.cell.2016.10.056 (2016).
Publication keywords:
Retromer; Sorting nexins; Endosomes; Retrograde transport; Endocytic recycling; Cargo recognition; Sorting signals; Protein coats; Membrane recruitment; Membrane tubules
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.