In Brief
Chalk and other sediments found at the bottom of the ocean are largely made up of microscopic shells. These shells contain important chemical clues to the environment in which the animal lived, such as temperature, ocean pH, ocean productivity, ocean circulation, global ice volumes and much more. But there are questions as to how to interpret these clues, particularly in understanding how the shell chemistry changed after the animal dies, over the millions of years it lies in the sediments. A group of scientists from the University of Cambridge used the imaging branch of beamline I13 to study a range of shells, some “pristine” specimens from shallow water to much more heavily modified specimens found in deep water to try and understand how the chemistry is affected.
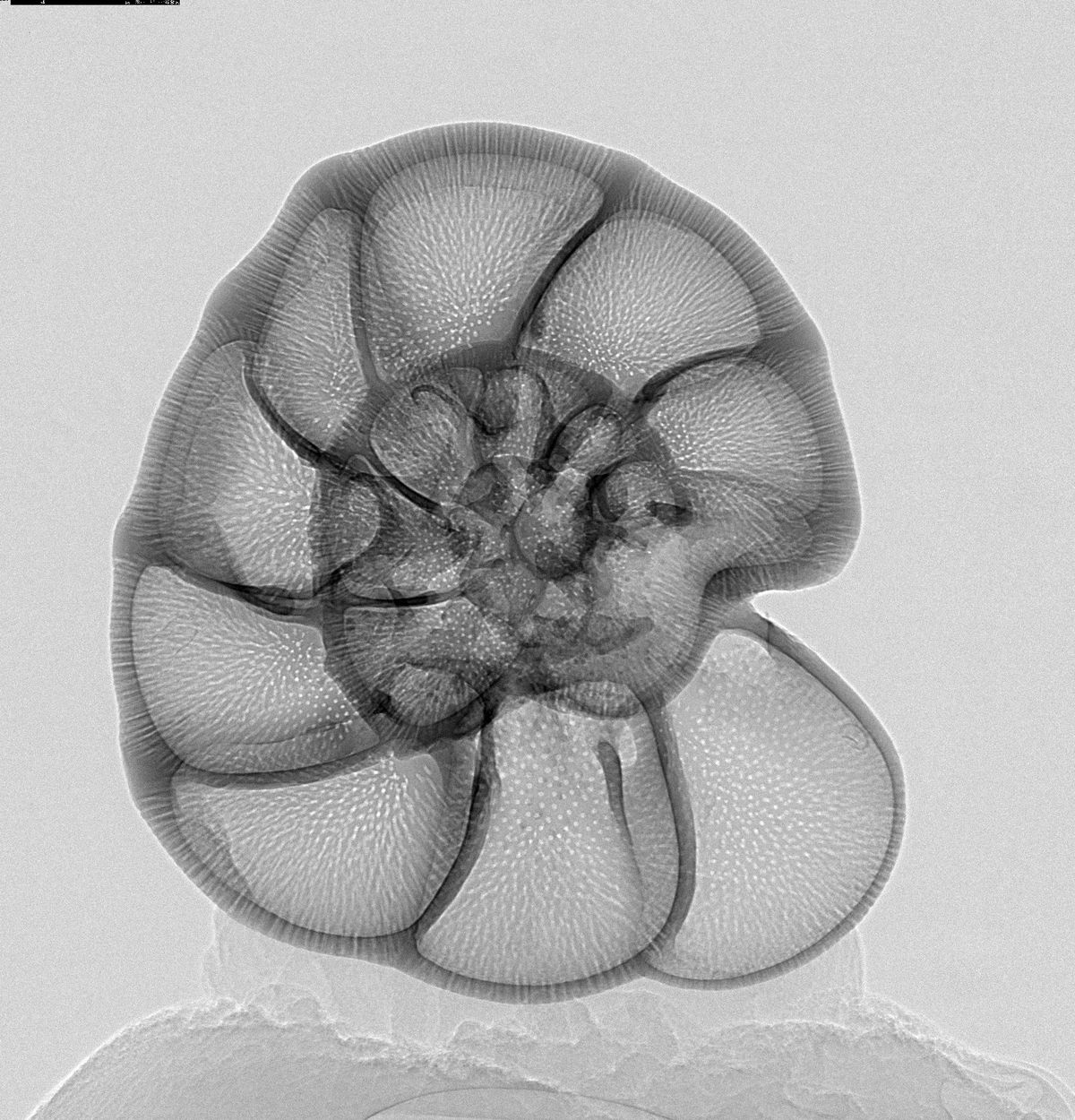
The trace chemistry of shells changes depending on the environment the animals live in. Ocean carbonate sediments (e.g. chalk) build up over millions of years, and are composed of billions of microscopic shells. Analysing the chemistry of these shells provides 'snapshots' of the environmental conditions when those organisms were alive, and allow us to reconstruct unparalleled long-term records of global climate change.
An important group of organisms in these reconstructions are the ubiquitous foraminifera, single-celled organisms that produce beautifully intricate calcite shells. Different species of foraminifera are found in the surface waters (planktic) and on the seafloor (benthic), and the chemistry of their shells provides a wealth of information, from which people have derived climate records stretching back tens of millions of years, covering temperature, ocean pH, ocean productivity, ocean circulation, global ice volumes and much more. This data is central to understanding the past and future Earth climate, and its sensitivity to perturbations like the anthropogenic production of CO2.
However, there are numerous uncertainties and questions surrounding the links between foraminifera chemistry and their environment. These uncertainties lie in two main areas: the role of biology in modulating shell chemistry, and the potential modification of the recorded chemical signals after deposition in the sediments through deposition/re-precipitation reactions (diagenesis). Describing and understanding these uncertainties is essential in interpreting and using environmental data derived from shell chemistry. The scientists used the micro-tomography capabilities of the new I13 beamline to try and address some of the issues surrounding post-depositional modification of the shells, with the aim of directly observing some of the diagenetic changes occurring in the sediments.
They generated 3D tomography scans of several relatively young (less than 10,000 years) foraminifera found on the upper surface of the sediment at different water depths on the Ontong-Java plateau. Seawater chemistry changes with depth (the saturation of CaCO3 reduces with depth), and the deeper specimens were expected to be more heavily modified, although the form this modification would take is unclear.
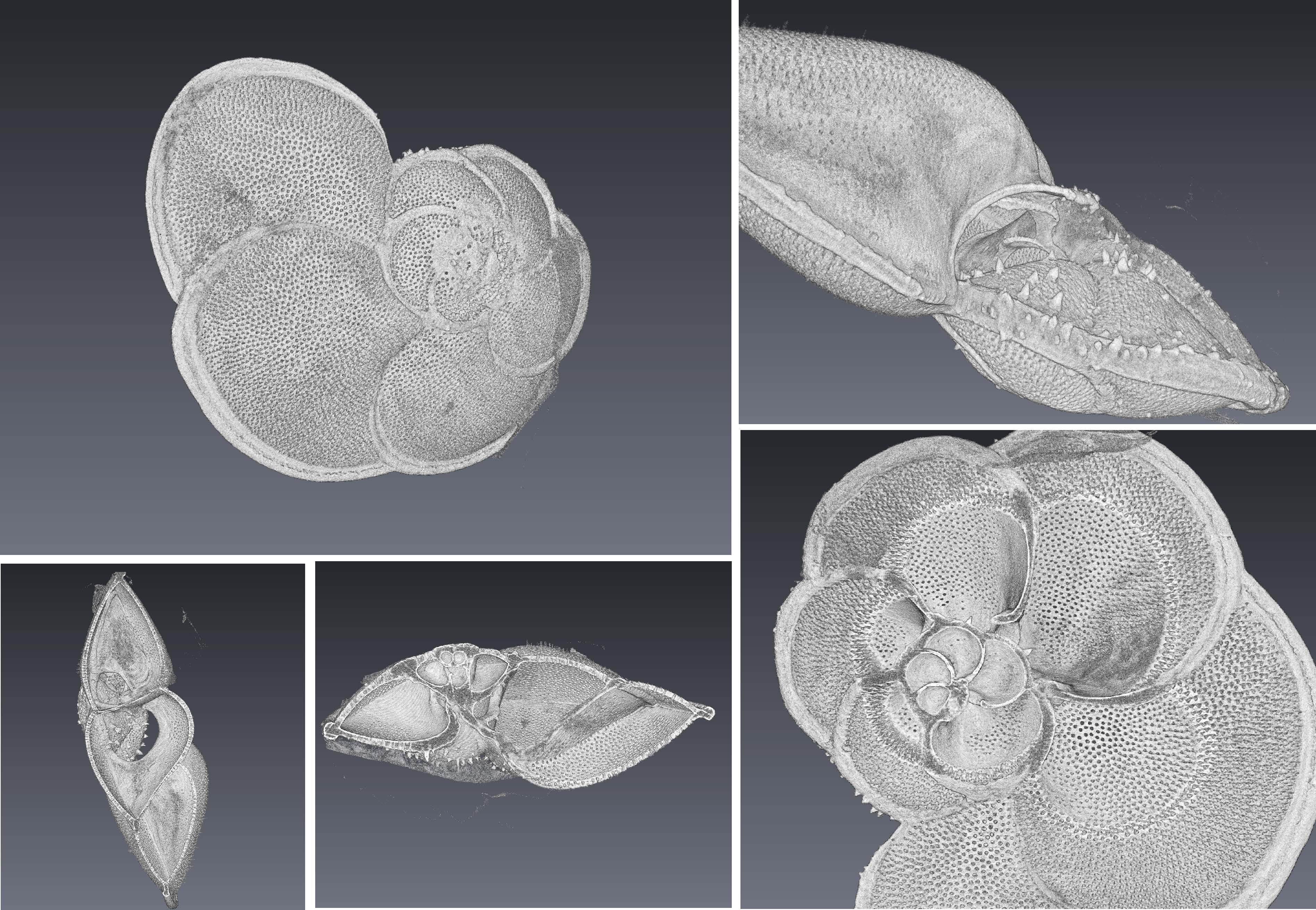
They observed a range of preservations, from 'pristine' specimens in the shallow samples, to heavily-modified specimens that were barely recognisable as foraminifera in the deepest samples. The modification was manifest in the dissolution of the interior chamber walls, and the deposition of a thick 'blocky' calcite fringe on the outer wall of the foraminifera. This blocky fringe was almost certainly not deposited by the organism, as all the 'biogenic' calcite has clear signs of internal structure (esp. laminations and pore structures), which was absent in the fringes. Some of the pore structures of the original shell continue into the fringe material, but this is probably the result of the orientated nucleation sites available to the growing crystals (i.e. the original shell structure), and/or variations in the concentration of organic molecules on the original structure.
The percentage of original shell material (relative to the volume of the encrusting fringe) decreased rapidly with depth, with as little as 10-20% original shell material remaining in the deepest samples. This dissolution/precipitation appeared to be active well above the depth at which CaCO3 becomes undersaturated in seawater.
“Following this research we think that understanding the preservation of specimens should be paramount in their chemical analysis. If 80% of the calcite being analysed was precipitated in the sediments, rather than while the foraminifera was still alive, the measured signal will not necessarily reflect the conditions that the organism lived in. This will be a major source of data scatter and error in palaeo-oceanography climate reconstructions.”Oscar Branson, University of Cambridge
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.