Our indigenous gut microbiota play an important role in maintaining normal health and nutrition. They provide us with traits that the human genome does not encode, such as the degradation of otherwise indigestible dietary polysaccharides. Survival in the gastrointestinal (GI) tract depends on the ability of these microorganisms to rapidly respond to changes in their dynamic nutrient environment. Here we show that a dominant member of the normal gut microbiota, Bacteroides thetaiotaomicron, utilises a membrane associated hybrid two-component system (HTCS), BT1754, to sense and respond to the presence of extracellular fructans, ß2,1 or ß2,6 linked homopolymers of fructose. HTCS appear to be a common adaptation to life in the gut and all currently sequenced Bacteroidetes sequenced contain large numbers of genes encoding these novel sensor-regulators, closely linked to genes encoding proteins involved in complex carbohydrate acquisition and degradation. The periplasmic sensor domain of BT1754 recognises monomeric fructose, despite the preference of the bacterium for ß2,6 linked fructans. Specificity for the ß2,6 linkage is driven instead by the outer membrane components of the fructan utilisation system. The structure of the periplasmic domain of BT1754 bound to fructose reveals the protein adopts a classical two-domain TypeI periplasmic-binding protein fold and provides significant insight into the mechanism of signal transduction in this ubiquitous class of bacterial sensor-regulators.
Our indigenous gut microbiota, composed of ~100 trillion mainly bacterial cells, play a significant role in maintaining normal health and nutrition. Microbial survival in the distal intestine is dependent on the ability of members of the community to harvest energy from the wide range of dietary polysaccharides (mainly plant derived) that the host cannot access. In the dynamic nutrient environment of the gut, survival depends on the ability of these microbes to respond rapidly to changes in glycan availability. In bacteria, responses to changes in the extracellular milieu are commonly mediated by two-component systems (TCS), which are composed of a membrane-bound sensor histidine kinase (HK) which recognises a specific environmental signal and a cytoplasmic response regulator that is activated by the HK and is responsible for the required output, often direct induction of gene expression.
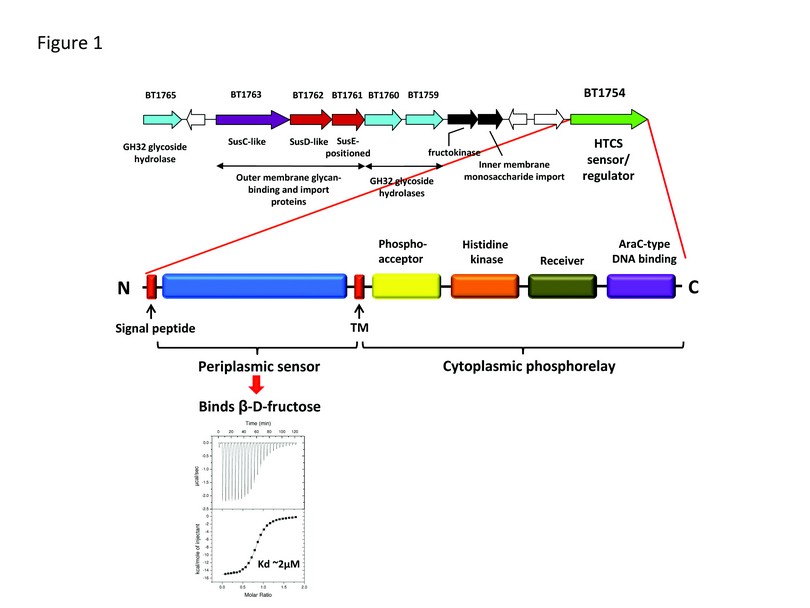
Figure 1: Bacteroides thetaiotaomicron fructan utilisation locus and domain architecture of the hybrid two-component system BT1754. The upper panel shows the gene content of the Bt fructan locus including the main structural genes and the regulator of the locus BT1754. Genes that encode proteins of related function are colour coded, intervening unrelated genes are coloured white. The lower panel shows the domain composition of BT1754 HTCS and the isothermal titration calorimetry (ITC) data revealing the periplasmic domain binds to ß-D-fructose. The upper part of the ITC insert displays the raw heats of binding and the lower part shows the integrated binding data fit to a single site binding model.
The genomes of all sequenced gut Bacteroidetes, one of the two dominant phyla in the gut, contain large expansions in gene families whose products are predicted to be involved in the acquisition and degradation of complex glycans, including outer membrane associated polysaccharide binding and transport proteins (SusC and SusD homologues) and carbohydrate-active enzymes (e.g. glycoside hydrolases).1 Many of these genes are closely linked on the genome in discrete co-regulated clusters known as polysaccharide utilisation loci (PULs), each of which is physically linked to a gene encoding a novel class of membrane associated sensor/regulator known as a hybrid two-component system (HTCS), which contains all of the domains of a classical TCS but in a single polypeptide.
Here we show that Bacteroides thetaiotaomicron (Bt), a dominant member of the normal gut microbiota, utilises a HTCS to mediate its response to fructan, a plant-derived homopolymer of fructose found in many foods. The HTCS, BT1754, controls expression of the Bt fructan PUL that encodes multiple family 32 glycoside hydrolases (GH32) as well as SusC-like and SusD-like outer membrane polysaccharide binding and transport proteins (Fig. 1). Although Bt displays a strong preference for ß2,6- over ß2,1-linked fructan, binding studies with the periplasmic domain of BT1754 (BT1754pd) reveal the sensor recognises monomeric fructose and cannot therefore distinguish between the two forms of fructan (Fig. 1). Specificity for ß2,6-fructan is instead driven by the surface located components of the PUL; the polysaccharide binding proteins BT1761 and BT1762 and the endo-acting GH32, BT1760 (Fig. 1).
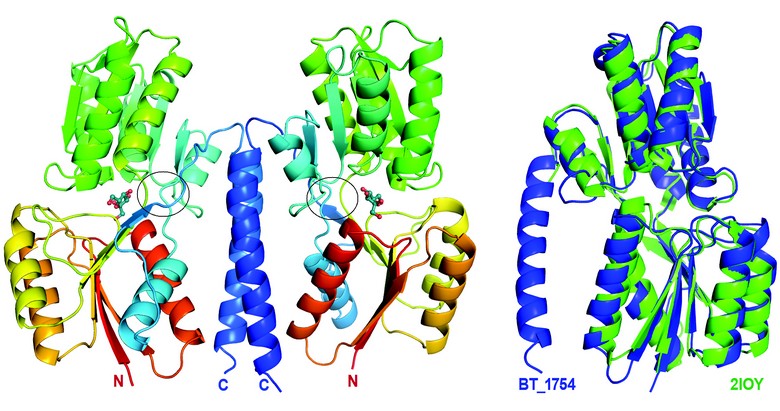
Figure 2: Structure of the periplasmic sensory domain of BT1754 (BT1754pd). The left hand panel shows the crystal structure of the homodimeric form of BT1754pd, with each monomer bound to a single molecule of ß-D-fructose (shown as ball and stick) sandwiched between the upper and lower subdomains. The flexible hinge region between the two subdomains of each monomer is circled. The right hand panel shows a superimposition of 1754pd (blue) with its closest structural homologue Thermoanaerobacter tengcongensis ribose binding protein (RBP; green), revealing the major difference between the two proteins is the presence of an extended C-terminal helix in BT1754pd.
To provide insight into the mechanism of signal perception and transduction in HTCS we determined the structure of the periplasmic domain of BT1754 bound to fructose. The structure revealed that the sensor adopts a Type 1 bacterial periplasmic binding protein (PBP) fold which consists of two subdomains, each consisting of a core of 6 ß-strands with each side of the sheet flanked by a-helices (Fig. 2). The polypeptide chain forms a hinge by crossing between the two subdomains 3 times along one side (Fig. 2), the last of these exiting the PBP-fold and then forming a long a helix which extends back along the length of the protein to the N-terminal region (Fig. 2). This C-terminal helix provides the predominant interface for homodimerisation with the equivalent helix in the 2nd molecule of the asymmetric unit and is the main structural difference between classical soluble PBPs such as RBP and BT1754pd (Fig. 2). Although there are several hydrogen bonds to retain the turn between the PBP-fold and the helix, once the polypeptide has progressed beyond the first residue of the helix, the remainder of the contacts, both inter- and intra-molecularly, are nonpolar. The dimer, generating a buried surface area of 2640 Å,2 appears to be biologically relevant (see below) as both the N- and C-termini of each molecule are orientated such that they face in the same direction and therefore both molecules are positioned correctly for insertion into the membrane (Fig. 2). A single fructose molecule is bound between the two subdomains, making numerous polar interactions via its hydroxyls and ring oxygen with the side chains of mainly Arg and Asp residues (Fig. 3). In addition, both faces of the furanose ring make hydrophobic contacts with the planar surface of a tryptophan residue, one from each subdomain such that the sugar is ‘sandwiched’ between the two aromatics (Fig. 3).
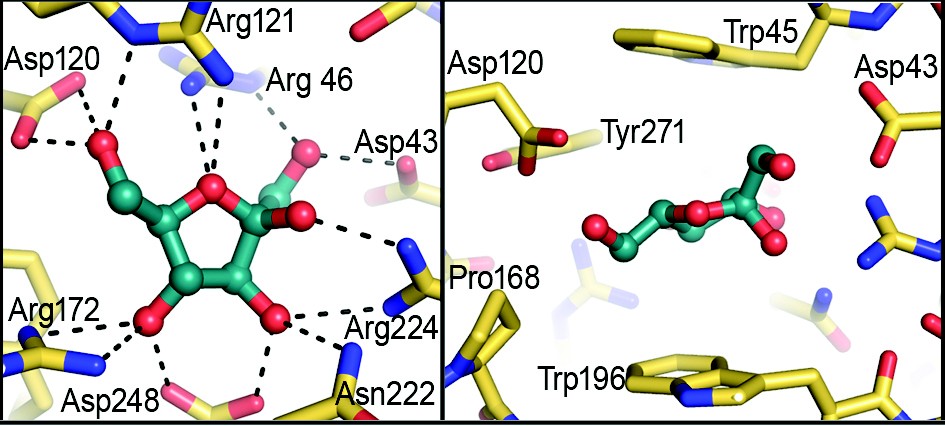
Figure 3: Binding site of BT1754pd. The left hand panel shows a top-down view of the bound fructose displaying main polar contacts between the sugar molecule and protein. The right hand panel is a side-on view of the fructose molecule showing the two tryptophan residues (Trp45 and Trp196) that sandwich the sugar ring.
A number of PBP structures have been determined in both apo and liganded forms revealing that the hinge allows a significant degree of flexibility enabling the subdomains to close around the ligand.2 The precise mechanism by which signal is transduced across the membrane to the cytoplasmic HK domains of classical two-component systems is not yet fully elucidated. It is known that many membrane associated sensor HK proteins dimerise to form a ‘four helix bundle’ in the membrane comprising the N- and C-terminal transmembrane domains.3 Current models for signal transduction indicate that on ligand binding a conformational change occurs in the extracellular sensor domain that is transmitted via movement of the transmembrane helices relative to one another to activate the associated cytoplasmic HK.4 Hybrid two-component systems are as yet not characterised, but data presented here supports a similar model for signal transduction in BT1754. Full length BT1754 is predicted to have two transmembrane helices – the N-terminal uncleaved signal sequence and a C-terminal helix extending from the end of the periplasmic sensor domain to the start of the cytoplasmic kinase domain and the crystal structure reveals the protein homodimerises (Fig. 2). In common with other members of the PBP-family the binding of ligand to BT1754 likely causes a conformational change in the protein as the two subdomains of the sensor close around the sugar molecule - indeed this movement of the subdomains must occur to allow access of the fructose to the binding site as in the ligand bound structure the sugar is excluded from bulk solvent. This contraction would be transmitted across the membrane, possibly as a vectorial ‘piston-like’ movement of the transmembrane helices relative to one another, resulting in activation of the cytoplasmic phosphorelay and upregulation of the fructan utilisation locus.
Sonnenburg, E.D., Zheng, H., Joglekar, P., Higginbottom, S.K., Firbank, S.J., Bolam, D.N., Sonnenburg, J.L. Specificity of polysaccharide use in intestinal Bacteroides species determines diet-induced microbiota alterations. Cell. 141, 1241-52 (2010)
References
- Martens, E.C. et al. Complex glycan catabolism by the human gut microbiota: the Bacteroidetes Sus-like paradigm. J. Biol. Chem. 284, 24673-7 (2009).
- Cuneo, M.J. et al, Ligand-induced conformational changes in a thermophilic ribose-binding protein. BMC. Struct. Biol. 8, 50 (2008).
- Szurmant, H. et al, a Sensor complexes regulating two-component signal transduction. Current Opinion in Structural Biology. 17, 706-15 (2007).
- Sevvana, M. et al, A ligand-induced switch in the periplasmic domain of sensor histidine kinase CitA. J. Mol. Biol. 377, 512-23 (2008).
Funding acknowledgment
Biotechnology and Biological Sciences Research Council, UK. National Institutes of Health, USA.
Research carried out at Diamond IO4.


