The rich and diverse biochemistry of proteins exemplifies the key importance of nanoscale phenomena on the mechanism and function of living organisms, and offers a wealth of opportunities for the development of new types of hybrid nanomaterials with tailored structure, function and form 1. The lipid bound protein bacteriorhodopsin is used by the organism Halobacterium Salinarum for light driven proton transport under anaerobic conditions. 2D arrays of this protein arranged as hexagonal trimers within the lipid bilayer may be isolated from the bacterium to form micron sized sheets 4.7 nm thick known as purple membrane (PM). Purple membrane can undergo a photochromic transition during the proton pumping cycle which has led to its employment as a holographic data storage medium2. Previously it was shown that ordered stacks of PM could be embedded periodically within a bioinorganic nanocomposite by intercalation of a 3-aminopropylsilicic acid precursor into the interlayer spaces of a pre-formed cast film. Upon drying, the organosilane underwent hydrolysis and condensation to produce an intercalated mesolamellar composite comprising nanometre-thin layers of hydrated silica located specifically between the PM sheets3, 4. Given the potential simplicity of this approach, it seems feasible that similar strategies could be developed based on the in situ synthesis of an optically transparent organic polymer specifically within the interlayer spaces of pre-formed PM lamellar films (Fig. 1). More recently, we demonstrated that self-supporting films of polymer/purple membrane layered nanocomposites could be prepared by in situ polymerization and cross-linking of pre-organized methacrylic acid monomers intercalated specifically within the interlayer spaces of an expanded methacrylic acid/purple membrane mesolamellar film. Significantly, the films showed enhanced structural and chemical stability and retained their photochromic functionality.
Structural studies on PM-containing films were undertaken using X-ray reflectometry (XRR). Control films of purple membrane on mica displayed two Bragg peaks at incident angles ? = 0.526° and 1.050° corresponding to the (001) and (002) reflections of a well-ordered lamellar mesostructure with an interlayer spacing d = 4.83 ± 0.01 nm (Fig. 2a), consistent with previous results for purple membrane multilayers. The lamellar structure was retained after swelling and reconstitution of the film in the presence of aqueous monomer at a methacrylic acid: purple membrane mass ratio = 1.2 : 1, although the (001) and (002) reflections were shifted to q = 0.348° and 0.763°, respectively, corresponding to an expanded interlayer spacing of 6.98 ± 0.33 nm (Fig. 2b). The ca. 2.2 ± 0.4 nm increase in layer spacing was commensurate with the intercalation of a multilayer of monomer and water molecules between the stacked PM sheets in the bionanocomposite film.
By inducing a free radical polymerisation reaction within the interstitial monomer it was possible to form free standing films comprising of poly(acrylate) and purple membrane. These films displayed a greatly enhanced resistance of the biological bacteriorhodopsin component to denaturation by ethanol compared to polymer free controls. XRR studies on the poly(acrylate)/purple membrane films showed broad Bragg peaks at ? = 0.465°, 0.290° and 0.200° that were assigned to multiple (001) reflections having an interlayer spacing of 5.5 ± 0.05 nm, 8.8 ± 0.15 nm and 13 ± 0.5 nm respectively (Fig. 2c). Although absence of the (002) reflection and detection of the 5.5 nm reflection indicated a loss of long range order and presence of the native purple membrane structure, respectively, the expanded interlayer spacings at 8.8 and 13 nm were consistent with partial retention of an intercalated lamellar mesostructure within the polymerized films. The exact magnitude of these spacings varied according to the grazing angle used, but in each case remained significantly larger than the interlayer distance measured for the monomeric methacrylic acid/purple membrane films, indicating that in situ polymerization was accompanied by a considerable volume expansion. We attribute this to the presence of different arrangements of cross-linked poly(acrylate) macromolecules within the inter-membrane regions of the layered bionanocomposite films.
The results represent the successful integration of self assembly and polymer chemistry to form nanostructured bionanocomposites comprising ordered arrangements of functionally intact cell membranes for use for example in data storage. Moreover, it should be possible to apply a similar approach to other systems based on functional trans-membrane proteins provided that they can be extracted or reconstituted in the form of structurally robust lipid/protein supramolecular complexes.
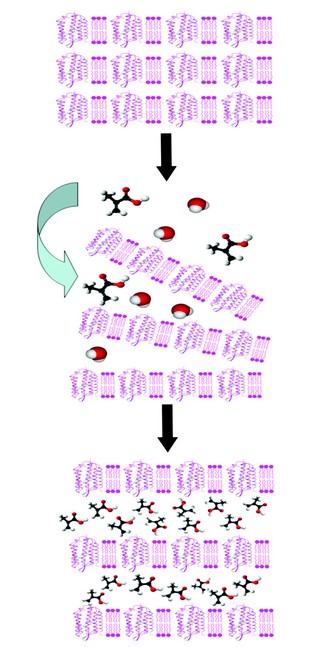
Figure 1: General method for preparation of polymer/purple membrane layered nanocomposites. (a) A thin film consisting of an ordered lamellar stack of PM sheets containing transmembrane bacteriorhodopsin protein molecules is produced by electrodeposition or controlled air drying. (b) Addition of an aqueous solution of an organic monomer (eg. methacrylic acid) onto the surface of the film induces swelling and partial exfoliation allowing the monomer to penetrate into the interlayer spaces. (c) Evaporation of water induces re-alignment of the stacked assembly along with intercalation of the monomer. Subsequent photolytic or free radical polymerisation in combination with chemical crosslinking results in a polymer-intercalated layered nanocomposite with bio-functional properties.
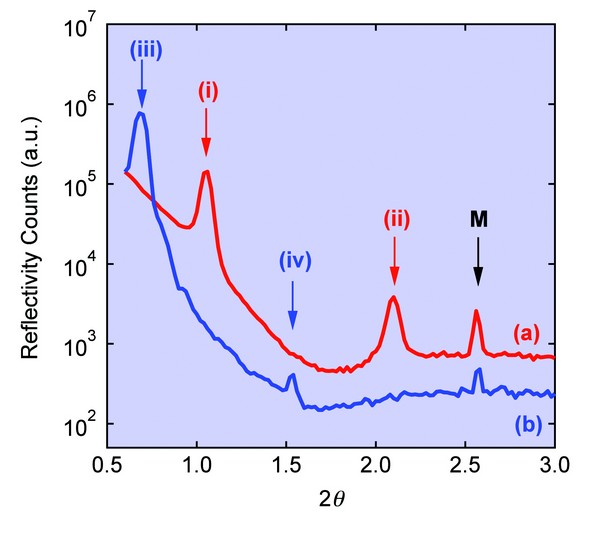
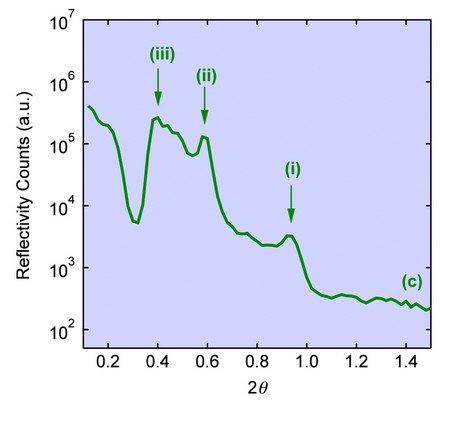
Figure 2: XRR scans for (a) Purple membrane control film displaying (001) and (002) Bragg reflections (peaks (i) and (ii), respectively) corresponding to a lamellar mesostructure with an interlayer spacing of 4.8 nm. (b) Intercalated methacrylic acid/purple membrane composite film showing retention of the layered structure but with expanded (001) and (002) spacings (peaks (iii) and (iv), respectively) corresponding to an interlayer distance of 6.2 nm. (c) poly(acrylate)/purple membrane film showing multiple (001) reflections (peaks (i)-(iii) having interlayer spacings of 5.5, 8.8 and 13 nm respectively). Peaks marked M represent a forbidden Bragg reflection from the mica substrate.
Collins, A.N., Kaus, N.H.M., Speranza, F., Briscoe, W.H., Rhinow, D., Hampp, N., Mann, S., Assembly of poly(methacrylate)/purple membrane lamellar nanocomposite films by intercalation and in-situ polymerisation. J. Mater. Chem.20, 9037-904 (2010)
References
- Mann, S. Angew. Chem.-Int. Edit. 47, 5306-5320 (2008).
- Hampp, N., Brauchle, C., and Oesterhelt, D. Biophysical Journal. 58, 83-93 (1990).
- Bromley, K.M. et al. Adv. Mater. 19, 2433-2438 (2007).
- Collins, A.M. et al. Biomacromolecules.10, 2767-2771(2009).
Acknowledgements
The authors would like to thank EPSRC for funding, and Oier Bikondoa (ESRF BM28) and Thomas Arnold (Diamond Light Source I07) for their technical expertise with XRR measurements.


