In metallo-supramolecular chemistry small molecules are organised into larger, more complex assemblies through the use of reversible metal-ligand coordination interactions. These assemblies are highly complicated and are often polyhedral or prismatic in shape and of nanometre scale. Given the right conditions they effectively make themselves in a process called self-assembly. They may have significant internal space where other molecules can be trapped or even react, acting like nano-sized reaction vessels. Our research utilises ligands based on the host molecule cyclotriveratrylene (CTV) that have a rigid pyramidal shape, with a distinct molecular cavity. These self-assemble with appropriate metal cations to produce large metal-ligand cage-like assemblies that we dub the “star-burst” prisms.1,2 As the ligands also serve as molecular hosts in their own right, they have molecular recognition sites on the inside. The smallest such cage is a metallo-cryptophane where two CTV-type ligands are joined together via metal centres to create a capsule.3,4 These are analogous to the purely organic cryptophanes which have applications in host-guest chemistry, including binding of gases such as hydrocarbons and Xe4. Structural characterisation of such metallo-supramolecular assemblies is dependent on single crystal X-ray techniques, however this often presents difficulties as the crystal obtained are often small and are highly solvated, hence disordered, by design. The use of synchrotron radiation can therefore be critical in being able to obtain crystal structures, as we have used synchrotron data to structurally characterise a highly novel dimer of a new type of metallo-cryptophane using a carboxylic acid derived CTV-ligand.
Tris[3,5-bis(methyl)benzoic acid]cyclotricatechylene (H3L) is a novel cavitand with carboxylic acid functionality on its upper rim groups (or “side-arms”) (Fig. 1). Carboxylic acids can be easily de-protonated to the corresponding carboxylate. These are excellent ligand groups, able to bind metal centres through a number of modes, including a bridging mode which encourages formation of high nuclearity metal clusters (Fig. 1). In this study, we investigated the formation of new self-assembled metallo-supramolecular species using this new cavitand-type ligand.
A mixture of copper(II) acetate, Cu(OAc)2, and H3L in dimethylformamide (DMF) gave greenish blue crystals of composition [Cu3L2(DMF)3].2(DMF) 1 after heating at 90°C for 48 hours, followed by slow cooling. The structure was determined by single crystal X-ray techniques, with a structure solution obtained in the triclinic space group P1. The complex is a novel type of metallo-cryptophane where a trimer of Cu(II) centres is capped by two L3- ligands arranged in a head-to-head fashion (Fig. 2a). The carboxylate groups of L3- bridge between Cu(II) centres, to form the trinuclear Cu(II) cluster. The metal clustering means the ligand side-arms are directed inwards above the molecular bowl of the cavitand, rather than outwards as occurs with polymeric or larger cage-like assemblies. This gives the metallo-cryptophane a pinched in and distinctive “bow-tie” appearance which is quite distinct from other examples of [M3L2] metallo-cryptophanes,3 or organic cryptophanes4 which have a larger guest-accessible cavity in their central region. Each Cu(II) centre in 1 is coordinated by four carboxylate donors arranged in an approximate square plane and one DMF ligand occupying an axial position to give a square pyramidal geometry. An analogous [Cu3L2(DEF)3] metallo-cryptophane can be isolated from diethylformamide (DEF) solution, where the axial ligands are the larger DEF solvent molecule.
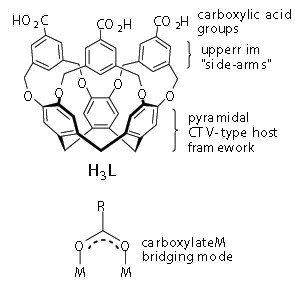
Figure 1: Ligand design and typical carboxylate binding mode.
Similar treatment of a mixture of cobalt(II) acetate, Co(OAc)2, and H3L in dimethylformamide (DMF) resulted in formation of the dark purple complex [Co7(µ3-L)2(µ3-OAc)4(µ4-O)2(DMF)2].(DMF).2(H2O) 2. The structure was determined by single crystal X-ray techniques, with a structure solution obtained in the monoclinic space group P21/c. The molecular structure of [Co7(µ3-L)2(µ3-OAc)4(µ4-O)2(DMF)2] consists of a cluster of seven Co(II) centres capped by two L3- ligands arranged in a head-to-head fashion, in another example of a “bow-tie” metallo-cryptophane (Fig. 2b). This, however, is a much more complicated metal cluster than was seen for [Cu3L2(DMF)3]. The two L3- ligands cap a central centrosymmetric Co7(µ-OAc)4(µ-O)2(DMF)2 cluster where bridging between metal centres occurs via oxide (O2-) and acetate (OAc-) ligands. The cluster contains a central octahedrally coordinated Co(II) that is connected to the other six Co(II) centres via acetate ligands and the two oxides. Above and below this Co(II) are two symmetry-related Co3 layers which are arranged staggered with respect to each other. One Co(II) in these layers is octahedrally coordinated by two acetate ligands, two carboxylates groups from one L3- ligand and a terminal DMF ligand. The remaining Co(II) centres are both tetrahedrally coordinated, being bound by the oxide, acetate and L3- ligands. While Co7 clusters supported by organic ligands are known, this particular polyhedral motif is very rare with only one other example having been reported, namely complex [Co7(µ4-O)2(µ3-OAc)4(µ2-OAc)6(OPEt3)2] which also features the same pattern of bridging oxide, carboxylate and terminal ligands.5
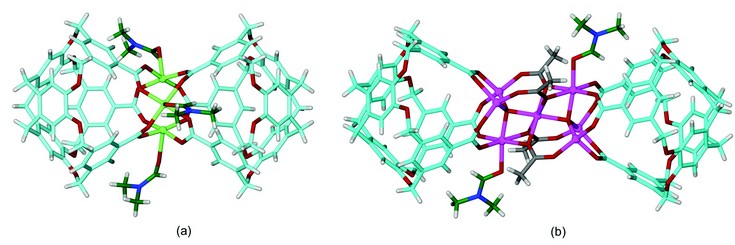
Figure 2: Bow-tie metallo-cryptophanes from the X-ray structures of complexes 1 and 2. (a) Complex [Cu3L2(DMF)3]. (b) The [Co7(µ3-L)2(µ3-OAc)4(µ4-O)2(DMF)2] assembly with unusual Co7 cluster. O = red; N = dark blue; H = white; Cu = light green; Co = pink; C = light blue (in L3-), dark green (in DMF) or grey (in OAc).
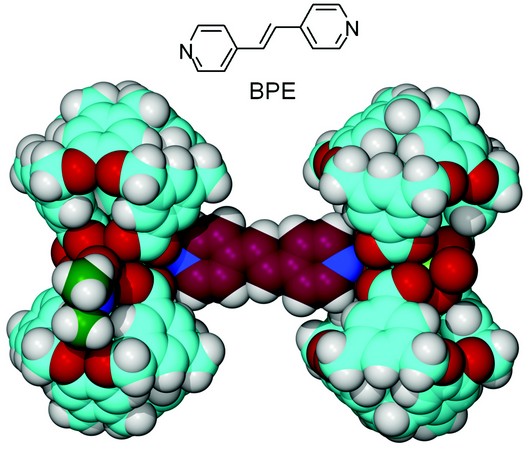
Solvent molecules such as DMF are relatively weak ligands, and we were interested in establishing whether or not the terminal solvent ligands on a metallo-supramolecular assembly such as the bow-tie metallo-cryptophanes could be displaced by other ligands. Use of a bridging ligand, for example, to replace the axial DMF ligand(s) of [Cu3L2(DMF)3] would result in the formation of larger clusters or networks. Hence, the reaction of H3L with Cu(OAc)2 was carried out in the presence of the potentially bridging ligand 1,2-bis(4-pyridyl)ethylene (BPE) to yield very small blue crystals of complex [{Cu3L2(DMF)(H2O)}2(µ-BPE)].4(DMF) 3. The crystal structure was determined using synchrotron radiation, in the triclinic space group P1.
The [{Cu3L2(DMF)(H2O)}2(µ-BPE)] complex consists of a discrete dimer of [Cu3L2(DMF)(H2O)] units linked together by a bridging 1,2-bis(4-pyridyl)ethylene ligand, Fig. 3. The two [Cu3L2] metallo-cryptophane units in [{Cu3L2(DMF)2}2(µ-BPE)] are very similar to those of [Cu3L2(DMF)3] in complex 1 and are crystallographically equivalent, being related by a centre of inversion. The bridging 1,2-bis(4-pyridyl)ethylene ligand takes up the axial binding position of one of the Cu(II) centres of each [Cu3L2], linking them together in a linear fashion with a Cu•••Cu separation of 13.85 Å. The remaining two Cu(II) centres of each [Cu3L2] unit are still coordinated by a terminal ligand (DMF or water), despite the presence of an excess of 1,2-bis(4-pyridyl)ethylene.
Figure 3: The [{Cu3L2(DMF)(H2O)}2(µ-BPE)] dimeric assembly of BPE-linked metallo-cryptophanes from the crystal structure of complex 3 shown in space-filling mode. Colour scheme as for Figure 2 with dark red = C (in BPE).
In summary, we have found that the carboxylate moiteies of L3- bridge between metal centres and promote the formation of high nuclearity M3 or M7 metal clusters. This generates a highly novel class of metallo-cryptophanes. The “bow-tie” cryptophanes have pinched in, metalled cores, rather than the large guest-accessible void space that is usually seen for (metallo)cryptophanes. Linking the [Cu3L2] complexes together into a dimeric extended assembly via bridging ligands, points to the potential for linking even larger metallo-cages through similar mechanisms. This would create crystalline materials with embedded cages as well as scaffolding-like porous frameworks.
Ronson, T. K., Nowell, H., Westcott, A. & Hardie, M. J. Bow-tie metallo-cryptophanes from a carboxylate derived cavitand. Chem. Commun. 47, 176-178 (2011)
References
- Sumby, C. J. et al. . Tris(pyridylmethylamino)cyclotriguaiacylene cavitands: an investigation of the solution and solid state behaviour of metallo-supramolecular cages and cavitand-based coordination polymers. Chem. Eur. J. 12, 2945-2959 (2006).
- Ronson, T.K. et al.Star-burst prisms with cyclotriveratrylene-type ligands: a [Pd6L8]12+ stella octangula, Angew. Chem. Int. Ed. 46, 9086-9088 (2007).
- Zhong, Z. et al. Creation of novel chiral cryptophanes by a self-assembling method utilizing a pyridyl-Pd(II) interaction. Org. Lett. 3, 1085-1087 (2001).
- Brotin, T. & Dutasta, J.-P. Cryptophanes and their complexes – present and future. Chem. Rev.109, 88-130 (2009)
- Ackermann, H. et al. Crystal structure of the oxo-acetate complex [Co7(m4-O)2(O2CCH3)10(OPEt3)2], Z. Naturforsh. B, 53, 1241-1243 (1998).
Acknowledgements
We thank EPSRC, STFC, EPSRC Mass Spectrometry Service in Swansea and University of Leeds for their support.