Anions are of central importance in natural, medical, and industrial systems: mis-regulation of chloride channels is known to be a cause of cystic fibrosis; use of nitrates and phosphates in fertilisers has caused enormous environmental damage through eutrophication; and pertechnetate is a radioactive bi-product of the nuclear industry. Stimulated by the fundamental roles negatively charged species play in these areas, there has been enormous interest in the field of supramolecular anion chemistry during the past few decades
However, due to their more diffuse charge, variable geometry, pH sensitivity, and high solvation, anions are generally more difficult to coordinate than their cationic counterparts. Despite these difficulties, nature has produced a surfeit of anion binding solutions, and it is from these that synthetic chemists take their inspiration. In particular, the highly selective phosphate and sulphate binding proteins stand out as anion hosts. In both cases the oxoanion is totally encapsulated below the surface of the protein, entirely shielded from the solvent; selectivity arises due to subtle differences in cavity size, the intricate network of hydrogen bonding donor/acceptor groups, hydrogen bonding torsion angles, and electrostatic effects.
Inspired by nature’s successful strategies we have undertaken a research programme with the objective of using anion templation to construct elaborate interlocked host molecules whose unique three dimensional topological cavities are designed to emulate nature’s oxoanion binding site protein network of hydrogen bonds. By using a topologically unique cavity contained within an interlocked molecule, the anion can be shielded from the solvent and the 3D environment of the cavity tailored to produce selectivity for different anions.
Figure 1: Crystal structure of a calixarene [2]rotaxane encapsulating a chloride anion.
The relationship between structure and function is of crucial importance in the design of interlocked host systems for anions. Solution-phase techniques, such as NMR spectroscopy, provide valuable information, but often it is only when the crystal structure of a supramolecular system has been solved that the cooperative balance of non-covalent interactions that control its behaviour is revealed and appreciated. As the field of supramolecular chemistry advances however, the structures become larger and more complex, and crystallography becomes increasingly challenging. Large supramolecular structures are inherently difficult to crystallise. The crystals tend to be small, with large unit cells, displaying high levels of disorder and solvent inclusion. All of these factors result in weak X-ray diffraction and lead to a need for high intensity X-ray radiation, and therefore Diamond Light Source’s beamline I19 is an invaluable resource for conducting research in this area.
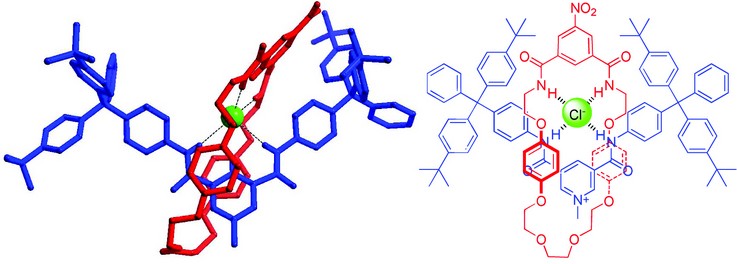
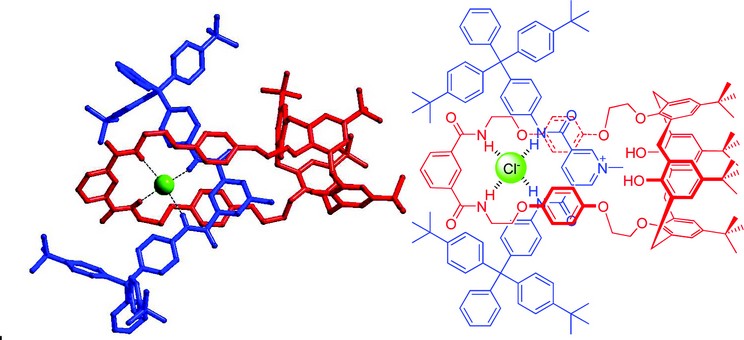
Figure 2: Crystal structure of a [2]rotaxane capable of recognising chloride in aqueous media.
Our interlocked host anion encapsulation strategy can be illustrated by one of the first structures analysed and published from I19.1 We reported the formation of a calix[4]arene-based [2]rotaxane host system, which was found to be selective for chloride and bromide in aqueous solvent mixtures. The crystal structure reveals a chloride anion held entirely within the binding cleft of the macrocycle by a pseudo-tetrahedral array of hydrogen bonds (Fig. 1). We have since elaborated on this binding motif, producing a range of new rotaxanes capable of recognising chloride in aqueous media (Fig. 2).2 Crystal structure elucidation was used to help understand the various steric requirements and thus correlate the trends in binding strength to structural features.
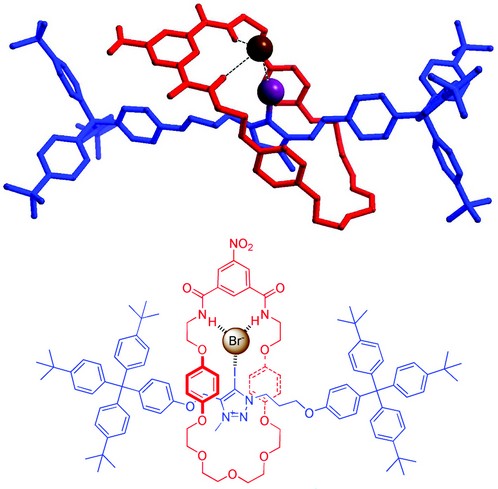
Figure 3: Crystal structure of a halogen bonding rotaxane binding a bromide anion.
In order to further tune the geometrically controlled selectivity of the anion-binding site we have begun investigating the use of halogen bonding in anion binding. Halogen bonds are formally noncovalent interactions formed between polarized halogen atoms functioning as electrophilic centres (Lewis acids) toward neutral or anionic Lewis bases. These attractive interactions arise from the terminal positive polarization of a covalently bonded halogen atom along the direction of the R-X bond, which concomitantly produces a perpendicular “belt” of negative electron density around the halogen, producing a strictly linear geometry to the halogen bond. Halogen bonds are largely underexploited in solution-phase supramolecular chemistry, which is surprising given their analogy to ubiquitous hydrogen bonding. We have sought to exploit the increased geometric and steric requirements of halogen bonding in the construction of novel selective anion hosts.3 This has led to the synthesis of the first interlocked molecule assembled via halogen bonding,4 which was found to display unusual iodide selectivity in aqueous media. X-ray crystal structure analysis at I19 provided vital characterization of the anion binding interaction in the halogen bonding rotaxane (Fig. 3).
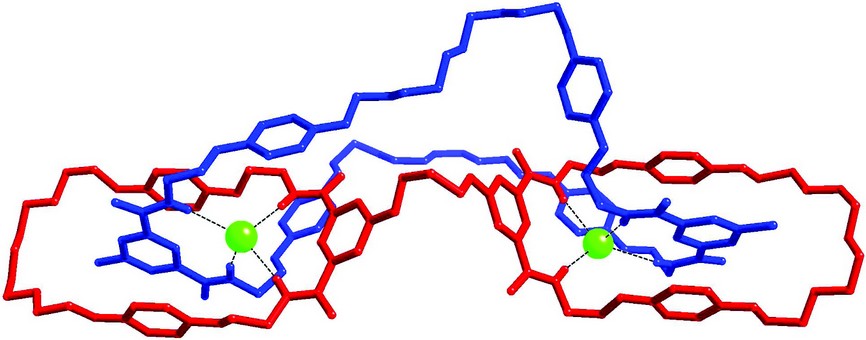
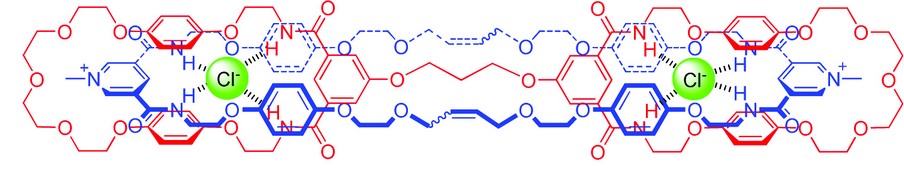
Figure 4: Crystal structure of a chloride anion templated handcuff catenane.
Although in recent years we have established the use of anion templation to generate singly interlocked structures, the production of higher-order structures is in its infancy. A “handcuff” catenane is an example of one such system in which two covalently linked macrocycles have a single macrocycle passing through both rings. To the best of our knowledge, only two examples of such species – constructed using cationic templation – have been described in the chemical literature to date. We have recently prepared and characterized a novel chloride anion templated handcuff catenane, and obtained its crystal structure at I19 – this being the first handcuff catenane crystal structure to be determined (Fig. 4).5
In summary, interlocked molecules are showing immense promise as potent and selective hosts for anions. The further elaboration and design of these structures has been helped immeasurably by crystallographic analysis, without which it would be impossible to fully understand the cooperative non-covalent interactions between interlocked host and anion guest involved in these systems.
Hancock, L.M., Gilday, L.C., Carvalho, S., Costa, P.J., Felix, V., Serpell, C.J., Kilah, N.L. and Beer, P.D. Rotaxanes Capable of Recognising Chloride in Aqueous Media. Chemistry - A European Journal. Vol.16, 44, 13082–13094, (2010).
References
- McConnell, A.J. et al. Chem. Eur. J.16, 1256-1264 (2010).
- Hancock, L.M. et al. Chem. Eur. J.16, 13082-13094 (2010).
- Serpell, C.J. et al. Angew. Chem. Int. Ed. 49, 5322-5326 (2010).
- Kilah, N.L. et al. J. Am. Chem. Soc.132, 11893-11895 (2010).
- Evans, N.H., Serpell, C.J. and Beer,P.D. Angew. Chem. Int. Ed., 50, 2507-2510 (2011).
Acknowledgements
We thank the EPSRC and the Royal Commission for the Exhibition of 1851 for post-doctoral funding and the EPSRC, Johnson Matthey and Woolf Fisher Trust for studentships, and Diamond Light Source for the award of beamtime.