Dr Steve Smerdon, National Institute for Heath Research
A protein complex called MRE11-RAD50-NBS1 (MRN) coordinates the detection and repair of one of the most hazardous forms of DNA damage, the double strand break (DSB), in which both strands of a DNA helix are cut. The nuclease and scaffolding components of MRN, MRE11 and RAD50 respectively, have been widely studied at the molecular level, however the molecular basis of NBS1 function remains undefined. Using beamline I04 at Diamond we have been able to solve a 2.3 Å crystal structure of the N-terminal region of Schizosaccharomyces pombe NBS1 (spNBS1), revealing an unusual, supra-modular fusion of FHA and BRCT-repeat domains that is currently unique to this protein. Genetic and biochemical analysis of human NBS1 (hNBS1) showed that the FHA domain binds to Ser-Asp-Thr-Asp- motifs recently identified in the DNAdamage scaffold protein Mdc1, when phosphorylated by casein kinase 2 isoforms. Furthermore, we howed that these motifs are evolutionary conserved and occur within fission yeast CTP1 where they promote interactions with the spNBS1 FHA domain necessary for its recruitment to DSBs. Finally, we have shown that human NBS1, but not its fission yeast orthologue, contains a second phosphobinding site, and can also bind to MDC1 motifs through interactions with the BRCT-repeat domain. This, together with the unusual structural arrangement of the NBS1 FHA- and BRCT-domains, suggests an important and unsuspected interplay between the two regions for NBS1 function in the DNA-damage response.
Double strand breaks are one of the most cytotoxic forms of DNA damage because of their potential to cause genomic abnormalities often linked to cell death and cancer. In response, cells launch a highly complex set of signalling and repair processes that involves chromatin modifications, checkpoint activation and the recruitment of numerous repair factors and adaptor/scaffold proteins to the damage site [1]. In eukaryotic cells, the error-free repair process of homologous recombination is initiated by the MRE11-RAD50-NBS1 (MRN) complex. Together these proteins execute early events such as DNA-end bridging via RAD50 coiled coils, nucleolytic processing of DNA ends through MRE11 and NBS1 recruitment/activation of the kinase ATM [2]. NBS1 was originally identified as the gene mutated in Nijmegen breakage syndrome, a rare autosomal genetic disorder characterised by growth retardation, microcephaly, immunodeficiency, and a predisposition to cancer [3,4]. NBS1 also acts downstream of ATM and forms phospho-dependent protein-protein interactions with molecules such as mediator of the DNA-damage checkpoint 1 (MDC1) [5-8]. This interaction is dependent on the N-terminal end of NBS1 that contains forkhead associated FHA) and BRCA1 C-terminal (BRCT) motifs, and a phosphorylated acidic repeat region in MDC1, the SDTD repeat. The subunits MRE11 and RAD50 of the complex have previously been well characterised, however, our understanding of the downstream role of NBS1 in diverse DSB signalling and repair pathways is limited. So by using a combined approach of structural, biochemical and genetic analysis on human and fission yeast NBS1, we have been able to uncover how NBS1 plays crucial roles in mediating NBS1 adaptor functions.
To define functional NBS1 regions, we expressed a series of truncated NBS1 fragments from a variety of species. We were able to purify 382- and 323-residue N-terminal fragments of hNBS1 and spNBS1 respectively. While the human protein has not yielded crystals, we successfully solved the X-ray structure of spNBS1 by single wavelength anomalous diffraction to 2.6 Å (PDB 3I0M). Collection of data from reductively methylated crystals then allowed the resolution to be extended to 2.3 Å (PDB 3I0N). Overall, the spNBS1 N-terminus reveals a surprisingly compact structural FHA-BRCT1-BRCT2 unit; while individually the FHA and BRCT-repeat domains show similar folds to other known domain structures, the configuration of the two regions is unusual in that it generates a substantial interface between the FHA domain and first BRCT repeat (Figure 1). Importantly, our crystal structure has also provided a framework for understanding disease-associated hNBS1 mutations that map to the BRCT1 core or FHA domain.
Using isothermal titration calorimetry (ITC) we found that our hNBS1 fragment bound a synthetic MDC1 pSDpTD motif peptide with an affinity of ~1 µM. No binding was detected to a peptide of the same sequence in which the threonine was not phosphorylated reflecting the pThr selectivity of FHA domains. However, it seems that NBS1 does have the ability to respond to varying phosphorylation levels; a peptide in which the serine was not phosphorylated reduced binding to ~20 µM demonstrating a NBS1 preference for diphosphorylated sequences over singly phosphorylated ones. Despite no clear MDC1 orthologs in yeast, purified spNBS1 fragments bound to the same MDC1 diphosphopeptide, prompting a search for other MDC1-related motifs in S. pombe. As a result, two potential sites were found within the S. pombe repair factor, CTP1, and used to investigate in vitro binding to spNBS1. Again, ITC revealed tight binding to diphosphorylated forms of these peptides in a pThr specific manner, suggesting an evolutionary conserved function for NBS1.
Analysis of the spNBS1 tructure and sequence revealed that the fission yeast BRCT-repeat domain does not contain crucial lysine and serine/threonine residues required for potential phosphate-binding interactions. These residues, which are present in hNBS1, led us to speculate that human NBS1 has a dual phospho-recognition mode. Indeed, and in contrast to spNBS1, ITC titrations of hNBS1 with MDC1 phospho-peptides had a stoichiometry greater than 1 and site-directed mutagenesis of conserved phospho-binding residues within the FHA domain (Arg28 or Ser42) or the BRCT-repeat (Lys160) had only modest affects on reducing binding affinities. To fully abolish phosphopeptide binding to hNBS1, both FHA and BRCT-repeat domains had to be disrupted. Collectively, our structural and biochemical data imply that the direct fusion of two phospho-binding domains in human NBS1 could enable crosstalk between the two binding sites that may be essential for NBS1 function in the response to DNA damage.
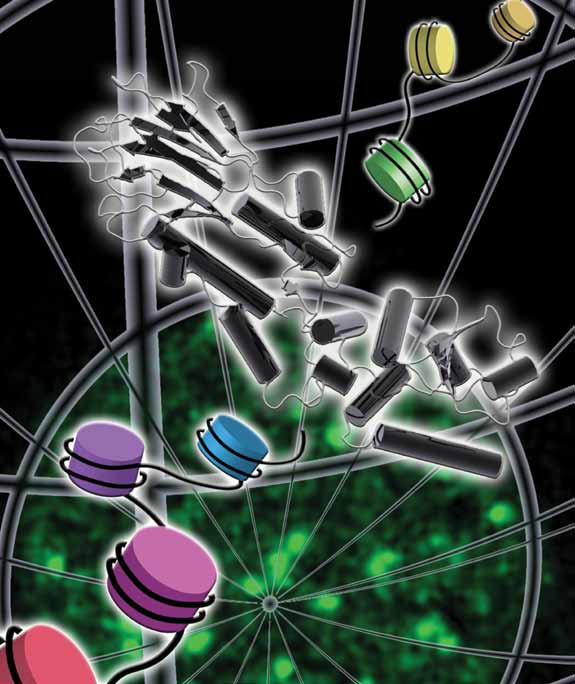 |
| Phosphorylation of the C-terminus of a variant histone, H2AX, is among the earliest events in the human response to double-stranded DNA breaks, showing up as foci of repair activity. Nbs1 – through its N-terminal region - mediates a variety of crucial recruitment and signalling activities in the pathways of DNA repair and cellcycle checkpoint regulation. |
References
[1] Riches LC, Lynch AM, Gooderham NJ. Mutagenesis, 23, 331-339 (2008).
[2] Williams RS, Williams JS, Tainer JA Biochem Cell Biol, 85, 509-520 (2007).
[3] Carney JP, Maser RS, Olivares H, Davis EM, Le Beau M, Yates JR, Hays L, Morgan WF, Petrini JH. Cell, 93, 477-486 (1998).
[4] Varon R, Vissinga C, Platzer M, Cerosaletti KM, Chrzanowska KH, Saar K, Beckmann G, Seemanova E, Cooper PR, Nowak NJ, Stumm M, Weemaes CM, Gatti RA, Wilson RK, Digweed M, Rosenthal A, Sperling K, Concannon P, Reis A. Cell, 93, 467-476 (1998).
[5] Chapman JR, Jackson SP. EMBO Rep, 9, 795-801 (2008).
[6] Melander F, Bekker-Jensen S, Falck J, Bartek J, Mailand N, Lukas J. J Cell Biol, 181, 213-226 (2008).
[7] Spycher C, Miller ES, Townsend K, Pavic L, Morrice NA, Janscak P, Stewart GS, Stucki M (2008). J Cell Biol, 181, 227- 240 (2008).
[8] Wu L, Luo K, Lou Z, Chen J. Proc Natl Acad Sci USA, 105, 11200-11205 (2008).
Principal Publications and Authors
Lloyd J, Chapman JR, Clapperton JA, Haire LF, Hartsuiker E, Li J, Carr AM, Jackson SP, Smerdon SJ. A supramodular FHA/BRCTrepeat architecture mediates Nbs1 adaptor function in response to DNA damage. Cell, 139, 100-1 (2009).
Funding Acknowledgement
Medical Research Council, UK.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.