We have demonstrated the use of a new technique for determining absolute crystal chirality by resonant X-ray diffraction of circularly polarized X-rays from a crystal of tellurium. The new approach is based on studies of ‘forbidden’ reflections, which, as the name implies, are normally considered not to exist. With the high-intensity X-ray beam available on Diamond beamline I16 such signals gain considerable intensity and are exceptionally sensitive to the chirality (handedness) of the crystal structure. We have shown, for the first time, that the new technique can be applied effectively to a crystal containing a single element – something that is not possible with established methods.
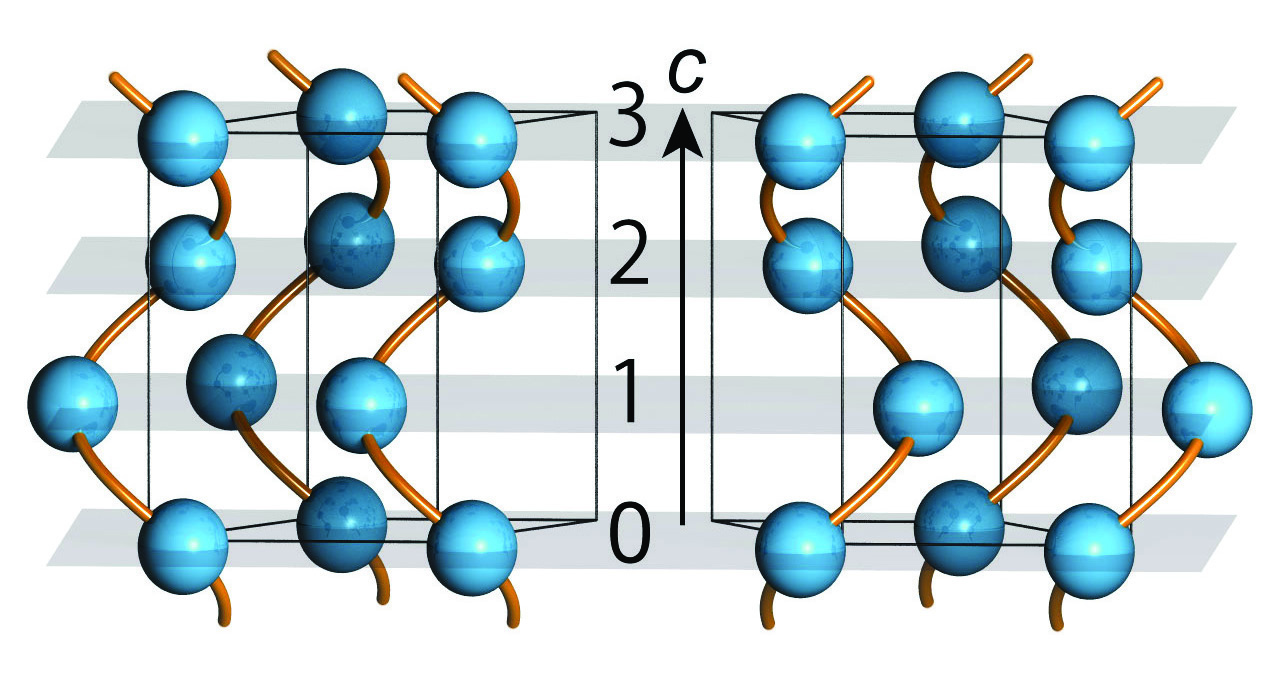
Figure 1: The crystal structure of tellurium for space group P3121 (right panel) and space group P3221 (left panel). Blue spheres represent tellurium atoms and black lines indicate the crystal unit cell, with the c-axis marked by an arrow. Screw lines for each panel are guides to eyes to highlight the two opposite spiral arrangements.
Chirality, or handedness, is ubiquitous in life at all length-scales down to individual molecules. An object is said to be chiral if there is no possible mirror reflection that leaves the object unchanged. Given the close relationship between diffraction and symmetry, it might seem surprising that the most basic and well-understood mechanism responsible for X-ray diffraction – the standard tool for studies of crystal symmetry – cannot distinguish between left- and right-handed structures. However, since the seminal paper by Bijvoet in 1951 [1], researchers have known how to extract chiral information from the small differences in the intensity of sister reflections called ‘Friedel pairs’ by exploiting the proximity of atomic resonances in the materials under investigation. This technique relies on a careful analysis of interference between the waves scattered from various types of atom in the target crystal.
The use of circularly polarised synchrotron X-ray beams provides an obvious probe of handedness, since the spiral form of the wave is manifestly chiral. However, it had been thought that such sensitivity would be weak and require a very exotic scattering process, beyond the scope of the normal ‘electric dipole’ scattering theory. This was recently proved not to be the case, in an important study of quartz [2] and subsequent characterization of its absolute chirality [3,4].
In a recent experiment at Diamond beamline I16, we have applied the same approach to studies of a single crystal consisting only of elemental tellurium [4]. The aim of the exercise was to show that handedness could be established unambiguously, by determining whether the crystal is described by the symmetry group P3121 (a right-handed screw) or P3221 (a left-handed screw).
Atomic structures of tellurium for both handed screws are shown in Figure 1. There are three tellurium atoms in each unit cell. The difference between space group P3121 and P3221 is simply the stacking sequence of atomic planes along the c axis, which is opposite in each case.
The nature of the forbidden reflections with Miller indices 00l can be understood as follows. In case of conventional x-ray diffraction, the scattering from tellurium atoms in plane 0 (Figure 1) is exactly equivalent to those of planes 1 and 2. The minimum repeat unit for reflection 00l is c/3 (the distance between two adjacent planes). In other words, reflection 00l is ‘allowed’ when l=3n (n is an integer) and forbidden when l is not equal to 3n. However, when the X-ray energy is tuned to an absorption edge, the scattering becomes extremely sensitive to the electronic state of the valence electrons and the forbidden reflections become weakly allowed. We call this technique pure resonant X-ray diffraction. The essential point is that, at the resonance energy, the scattering becomes very sensitive to the orientation of the atoms along the spiral, with respect to the X-ray polarization. The scattering from tellurium atoms in plane 0 is then no longer equivalent to those of planes 1 and 2 and the exact cancellation of scattering from these planes is prevented, allowing the 001 and 002 reflections to be observable.

Figure 2: The energy spectrum of the forbidden reflection 001 near to the L1 absorption edge for various azimuth angles. A consistent, single sharp resonance, lies beneath a rapidly fluctuating background of multiple scattering events. The inset shows a schematic view of Bragg diffraction with a right-handed coordinates x, y, and z.
Measurements were carried out by choosing the X-ray energy to be close to three atomic resonances (absorption edges), L1, L2 and L3, each of which gives sensitivity to specific symmetries of the valence electrons. We found the L1 resonance to give particularly spectacular results, showing a very strong sensitivity to the sample orientation. Figure 2 shows the intensity of forbidden 001 reflection as a function of X-ray energy, close to the L1 absorption edge. Each line shows an energy spectrum for a selected azimuth rotation angle Ψ of the sample about an axis perpendicular to the diffracting crystal planes (i.e. the c-axis). We find that reflection 001 is accompanied by strong ‘multiple scattering effects, adding apparently random artefacts to the spectra, which vary rapidly with Ψ. The underlying resonant reflection 001 at the L1 edge is observed as a single peak (the maximum is at E=4.94 keV) with a shape (but not intensity) that is independent of azimuth angle Ψ. Figure 3 shows the integrated intensity of reflections 001 (panel a) and 002 (panel b) as a function of azimuth angle Ψ for circularly polarised incident beams, and the difference (panel c) between opposite states of circular polarization which are represented by P2=+1 (right) and P2=-1 (left). As we observed, the intensity of these forbidden reflections clearly changes with the helicity of the circular polarization. It is interesting to note that the signs of these intensity differences are opposite for the two reflections, mimicking the effect of reversing the handedness of the crystal. This arises because the 001 and 002 reflections effectively sample the structure with different frequencies, producing an effect that is analogous to the apparent change in direction of a helicopter propeller in a film clip as its speed changes.
 Figure 3: The integrated intensity of two forbidden reflections as a function of azimuth angle Ψ, observed with the incident beam at E=4.94 keV and a circularly polarized beam. Closed and open circles (triangles) represent the diffraction intensity with the positive and the negative circularly polarized beams for reflection 001 (002), respectively. The difference intensity, (+)-(-), between two polarizations of the incident beam for reflection 001 and 002 is shown in panel c and appears to be less noisy, due to the cancellation of multiple scattering by subtracting intensities from opposite circular polarization states.
Figure 3: The integrated intensity of two forbidden reflections as a function of azimuth angle Ψ, observed with the incident beam at E=4.94 keV and a circularly polarized beam. Closed and open circles (triangles) represent the diffraction intensity with the positive and the negative circularly polarized beams for reflection 001 (002), respectively. The difference intensity, (+)-(-), between two polarizations of the incident beam for reflection 001 and 002 is shown in panel c and appears to be less noisy, due to the cancellation of multiple scattering by subtracting intensities from opposite circular polarization states.
After data analysis using the mathematical framework of atomic resonant scattering tensors, derived from the crystal symmetry, we conclude that the tellurium crystal has symmetry P3121 and is therefore right handed. It is hoped that this approach can eventually be applied to much more complex crystal structures that are of interest in the life sciences.
References
[1] J.M. Bijvoet, A.F. Peerdeman, and A.J. van Bommel, Nature, 168, 271–272 (1951).
[2] Y. Tanaka et al. Phys. Rev. Lett. 100,145502 (2008).
[3] S .W. Lovesey, E. Balcar, and Y. Tanaka J.Phys.:Condens. Matter, 20, 272201 (2008).
[4] Y. Tanaka et al. Phys. Rev. B, 81, 144104, (2010)
[5] Y. Tanaka, S. P. Collins, S. W. Lovesey, et al., J. Phys.: Condens. Matter, 22, 122201 (2010).
Principal Publications and Authors
Y. Tanaka, S. P. Collins, S. W. Lovesey, T. Moriwaki, M. Matsumami, and S. Shin, J. Phys.: Condens. Matter, 22, 122201 (2010).
Funding Acknowledgement
Grants-in-Aid for Scientific Research (A) No. 21244049 from the Japan Society for the Promotion of Science.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.