2017 Interfaces in Heterogeneous Catalytic Reactions: Ambient Pressure XPS as a Tool to Unravel Surface Chemistry R. Palomino, R. Hamlyn, Z. Liu, D. C. Grinter et al. J. Electron Spectr. Rel. Phenom. Accepted 2017 (link)
Ceria-based model catalysts: Fundamental studies on the importance of the metal-ceria interface in CO oxidation, the water-gas shift, CO2 hydrogenation, and methane and alcohol reforming J.A. Rodriguez, D.C. Grinter et al. Chem. Soc. Rev. Accepted 2017 (link)
Geometric Structure of Anatase TiO2(101) J. Treacy, H. Hussain. X. Torrelles, G. Cabailh, D.C. Grinter et al. Phys. Rev. B 95, 075416, 2017 (link)
2016 Room Temperature Activation of Methane and Dry Reforming with CO2 on Ni-CeO2(111) Surfaces: Effect of Ce3+ Sites and Metal-Support Interactions on C-H bond Cleavage P.Lustemberg, P. Ramirez, Z. Liu, D.C. Grinter et al. ACS Catalysis 6, 8184-8191, 2016 (link)
Structure of a TiO2 Photocatalytic Interface H. Hussain, G. Tocci, T. Woolcot, X. Torrelles, C. L. Pang, D. S. Humphrey, C. M. Yim, D.C. Grinter et al. Nature Materials, 2016 (link)
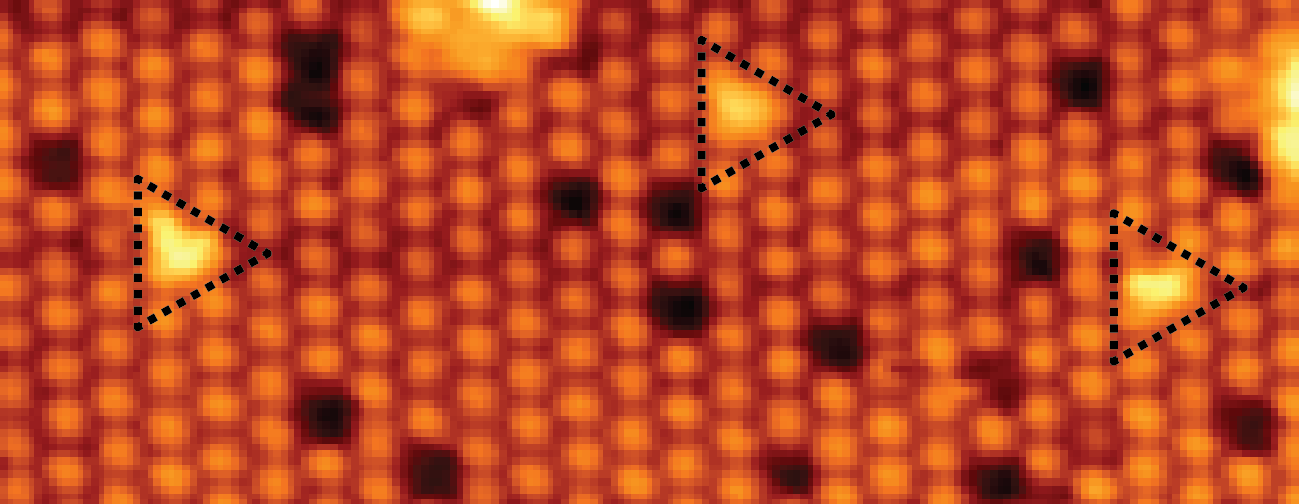
Potassium and Water Co-adsorption on TiO2(110): OH-induced Anchoring of Potassium and the Generation of Single-Site Catalysts D.C. Grinter et al. J. Phys. Chem. Lett. 7, 3866-3872, 2016 (link)
Inverse Oxide/Metal Catalysts in Fundamental Studies and Practical Applications: A Perspective of Recent Developments J. Rodriguez, P. Liu, J. Graciani, S.D. Senanayake, D.C. Grinter et al. J. Phys. Chem. Lett. 7, 2627-2639, 2016 (link)
Diffusion barriers block defect occupation on reduced CeO2(111) P.G. Lustemberg, Y. Pan, B-.J. Shaw, D.C. Grinter et al. Phys. Rev. Lett. 116, 236101, 2016 (link)
Spillover Reoxidation of Ceria Nanoparticles D.C. Grinter et al. J. Phys. Chem. C 120, 11037-11044, 2016 (link)
Dry reforming of Methane on a highly-active Ni-CeO2 catalyst: Effects of metal-support interactions on C-H bond breaking Z. Liu, D.C. Grinter et al. Angew. Chemie 55, 7455-7459, 2016 (link)
Ambient Pressure XPS and IRRAS Investigation of Ethanol Steam Reforming on Ni-CeO2(111) Catalysts: An In Situ Study of C-C and O-H Bond Scission Z. Liu, T. Duchon, H. Wang, D.C. Grinter et al. Phys. Chem. Chem. Phys. Accepted 2016 (link)
In Situ Growth, Structure, and Real-Time Chemical Reactivity of Well-Defined CeOx-Ru(0001) Model Surfaces D.C. Grinter, S.D. Senanayake and J.I. Flege J. Catal. B: Environ. Accepted 2016 (link)
2015 Water-Gas-Shift Reaction over Gold Nanoparticles Dispersed on Nanostructured CeOx-TiO2(110) Surfaces D.C. Grinter et al. Surf. Sci. 650, 34-39, 2015 (link)
Lepidocrocite-like TiO2 and TiO2(110)-(1x2) supported on W(100) G.T. Harrison, M.C. Spadaro, C.L. Pang, D.C. Grinter et al. Mat. Sci. & Technol. 32, 203-208, 2015 (link)
2014 Ordered Carboxylates on TiO2(110) Formed at Aqueous Interfaces D.C. Grinter et al. J. Phys. Chem. Lett. 5, 4265-4269, 2014 (link)
Spectro-Microscopy of a Model Water-Gas-Shift Catalyst: Gold Supported on Ultrathin Ceria D.C. Grinter et al. J. Phys. Chem. C 118, 19194–19204, 2014 (link)
Characterising Ultrathin Ceria Films at the Nanoscale: A Combined Microscopic Approach D.C. Grinter et al. J. Electr. Spec. Rel. Phenom. 195, 13-17, 2014 (link)
2013 A Scanning Tunneling Microscopy Study of Ultrathin Film Rutile TiO2(110) Supported on W(100)-O(2 × 1) C.L. Pang, D.C. Grinter et al. J. Phys. Chem. C 117, 25622–25627, 2013 (link)
Oxidation State Imaging of Ultrathin CeO2(111) Films on Re(0001) D.C. Grinter et al. J. Phys. Chem. C 117, 16509–16514, 2013 (link)
Scanning Tunneling Microscopy and Molecular Dynamics Study of the Li2TiO3(001) Surface K. Azuma, C. Dover, D.C. Grinter et al. J. Phys. Chem. C 117, 5126–5131, 2013 (link)
2012 Acetic Acid Adsorption on Anatase TiO2(101) D.C. Grinter et al. J. Phys. Chem. C 116, 11643-11651, 2012 (link)
Binding of a Benzoate Dye-Molecule Analogue to Rutile Titanium Dioxide Surfaces D.C. Grinter et al. J. Phys. Chem. C 116, 1020-1026, 2012 (link)
Characterization Tools of Ultrathin Oxide Films D.C. Grinter and G. Thornton in “Ultrathin Oxide Films: Science and Technology” Wiley-VCH 2012 (link)
2011 Reduction of Thin-film Ceria on Pt(111) by Supported Pd Nanoparticles Probed with Resonant Photoemission J. Matharu, D.C. Grinter et al. Surf. Sci. 605, 1062-1066, 2011 (link)
2010 The Defect Structure of Ultra-Thin Ceria Films on Pt(111): Atomic Views from Scanning Tunnelling Microscopy D.C. Grinter et al. J. Phys. Chem. C 114, 17036-17041, 2010 (link)