Carbon dioxide (CO2) is often in the news these days. As a greenhouse gas, released during the combustion of fossil fuels, it is fuelling climate change, and reducing our CO2 emissions is critical to a sustainable future. CO2 is also a by-product of many industrial processes, including the production of ammonia used for fertilisers. On the other hand, many industries need a regular supply of CO2, and shortages have caused problems in recent years. It makes sense, therefore, to find ways to recycle some of the waste CO2 we produce into useful products. However, CO2 conversion reactions are energy-intensive, and new catalysts are needed to make the reactions more efficient. Photocatalysts absorb light energy, creating a charge separation that can then drive a chemical reaction. A team of researchers from Imperial College London are researching CO2 conversion using photocatalysis. In work recently published in Chemistry of Materials, they investigated how oxygen doping affects the photocatalytic and optoelectronic properties of boron nitride. Their results provide valuable insights into the photochemistry of boron oxynitride (BNO) at the fundamental level.
Boron nitride has a similar structure to graphite, with layers of hexagonally arranged atoms that can slide over each other. However, unlike graphite, boron nitride is an electrical insulator. As it also has high thermal conductivity and can withstand high temperatures without breaking down, boron nitride is often used in high-performance ceramics, lubricants, and electronics.
Prof Camille Petit from Imperial College London says;
The use of boron nitride as a photocatalyst hasn't been widely studied. In previous research, our group has shown that it can photocatalyse CO2 conversion reactions. But we realised that it is still not efficient enough, and we don't fully understand how it works and what could improve it. So we started studying what happens if we change the composition a bit, for example, by introducing some oxygen groups. We wanted to see how introducing another element changes its properties, how CO2 interacts with the material, and how the material absorbs light. And we wanted to know if those changes depend on how the oxygen is integrated into the structure. So we used the B07 Beamline at Diamond Light Source to help us identify how the oxygen is integrated into the structure and its role in the optoelectronic and photocatalysis properties.
Prof Petit's group had previously used Near Edge X-ray Absorption Fine Structure (NEXAFS) spectroscopy on Diamond's B07-C beamline to investigate the formation of boron nitride, to develop an understanding of how the material forms and how its synthesis can be tuned. As boron nitride is made of light elements rather than metals, getting a good look at its structure is challenging. Prof Petit continues;
We worked closely with the beamline staff through the whole process. They helped us to fine-tune our ideas and develop a plan for the experiments, they supported us during our time at Diamond, and their support was invaluable while we analysed the data. So we knew that we would be able to use NEXAFS to study BNO for these experiments.
In BNO, oxygen doping substitutes oxygen atoms for some of the nitrogen atoms. NEXAFS shows which elements connect to the oxygen atoms. Previous research has shown that oxygen doping can extend light absorption to the visible range, and that BNO has magnetic and semiconducting properties that can be exploited in photocatalysis.
The research team combined NEXAFS results with data from other spectroscopic techniques (X-ray photoelectron spectroscopy (XPS), UV−vis DR, and X-band EPR) and DFT simulations. Their results suggest that paramagnetic, isolated OB3 sites in BNO significantly influence the material's photochemical and optoelectronic properties.
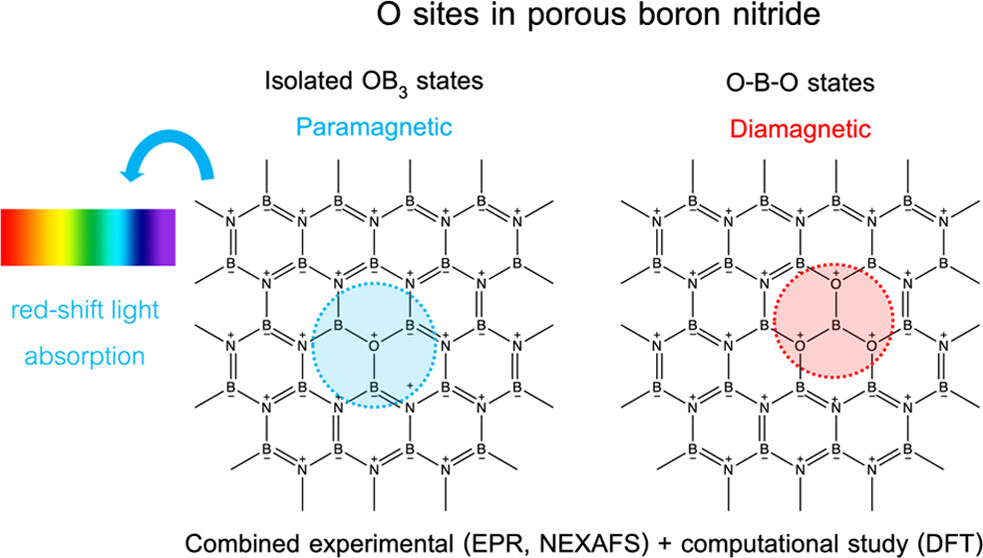
In previous research, the group found a strong inverse relationship between the oxygen content and band gap in BNO. However, using a different synthesis method created a material with the same relative oxygen content, but diamagnetic O−B−O sites in place of isolated OB3 sites. The presence of O−B−O sites caused larger band gaps and reduced photochemistry.
So while it is possible to use doping to tune the optoelectronic properties of BN, a thorough understanding of how different chemical states of oxygen affect the band gap is crucial for designing improved semiconductors.
By clarifying the importance of paramagnetism in BNO semiconductors and providing fundamental insight into their photophysics, this study paves the way to tailoring its properties for CO2 conversion photocatalysis. The group has also recently used a similar methodology to investigate phosphorus doping of boron nitride, which they will explore in a future publication.
To find out more about the B07 beamline or discuss potential applications, please contact Principal Beamline Scientist Georg Held: georg.held@diamond.ac.uk.
Mistry EDR et al. Paramagnetic States in Oxygen-Doped Boron Nitride Extend Light Harvesting and Photochemistry to the Deep Visible Region. Chemistry of Materials 35.5: 1858-1867 (2023). DOI:10.1021/acs.chemmater.2c01646.
L'Hermitte A et al. Formation mechanism and porosity development in porous boron nitride. The Journal of Physical Chemistry C 125.49: 27429-27439 (2021). DOI: 10.1021/acs.jpcc.1c08565.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.