Lithium-ion batteries have powered a global revolution, which now sees us carrying more computing power in our pockets than the Apollo 11 computer needed to land on the Moon. We're so taken with our mobile devices that there are now more of them on the planet than people. However, as we haven't given enough thought to what happens to these batteries at the end of their life, we're creating a mountain of garbage and wasting resources and energy. With a global trend towards electric vehicles powered by lithium-ion batteries, we must get a grip on recycling them. Recovering lithium from spent batteries has received the most attention, as it is a finite, expensive material. However, current recycling methods are energy-intensive. In work recently published in Carbon, an international team of researchers investigated an environmentally-friendly process for recovering graphite from battery anodes. By transforming a waste product into new high-performance anodes, their work takes us a step closer to economically-viable whole-battery recycling.
Graphite anodes are an integral part of lithium-ion batteries (LIBs). However, the graphite required is highly purified, with high production costs. Our ever-increasing demand for LIBs is putting a strain on limited graphite resources. Recycling the graphite from spent batteries is one possible solution.
There are two conventional approaches to LIB recycling: pyro-metallurgical and hydrometallic. The pyro-metallurgical process for LIB recycling uses the graphite anodes as fuel to help reach the high smelting temperatures necessary to recover metals and alloys. The hydrometallic recycling process can recover graphite from LIBs using a combination of solvent extraction and filtration techniques. However, the recovered graphite is of low purity and unsuitable for reuse in batteries.
An international team of researchers from the Indian Institute of Technology Hyderabad, India (Dr Surendra Kumar Martha), The Open University (Prof Satheesh Krishnamurthy), and the University of Cambridge, working with industrial partners, demonstrated an environment-friendly graphite recycling technique using water leaching. Vigorous stirring in distilled water separates the anode from the copper foil current collector. After filtering and drying, water washing removes most of the remaining lithium from the anode surface, restoring electrochemically-active graphite.
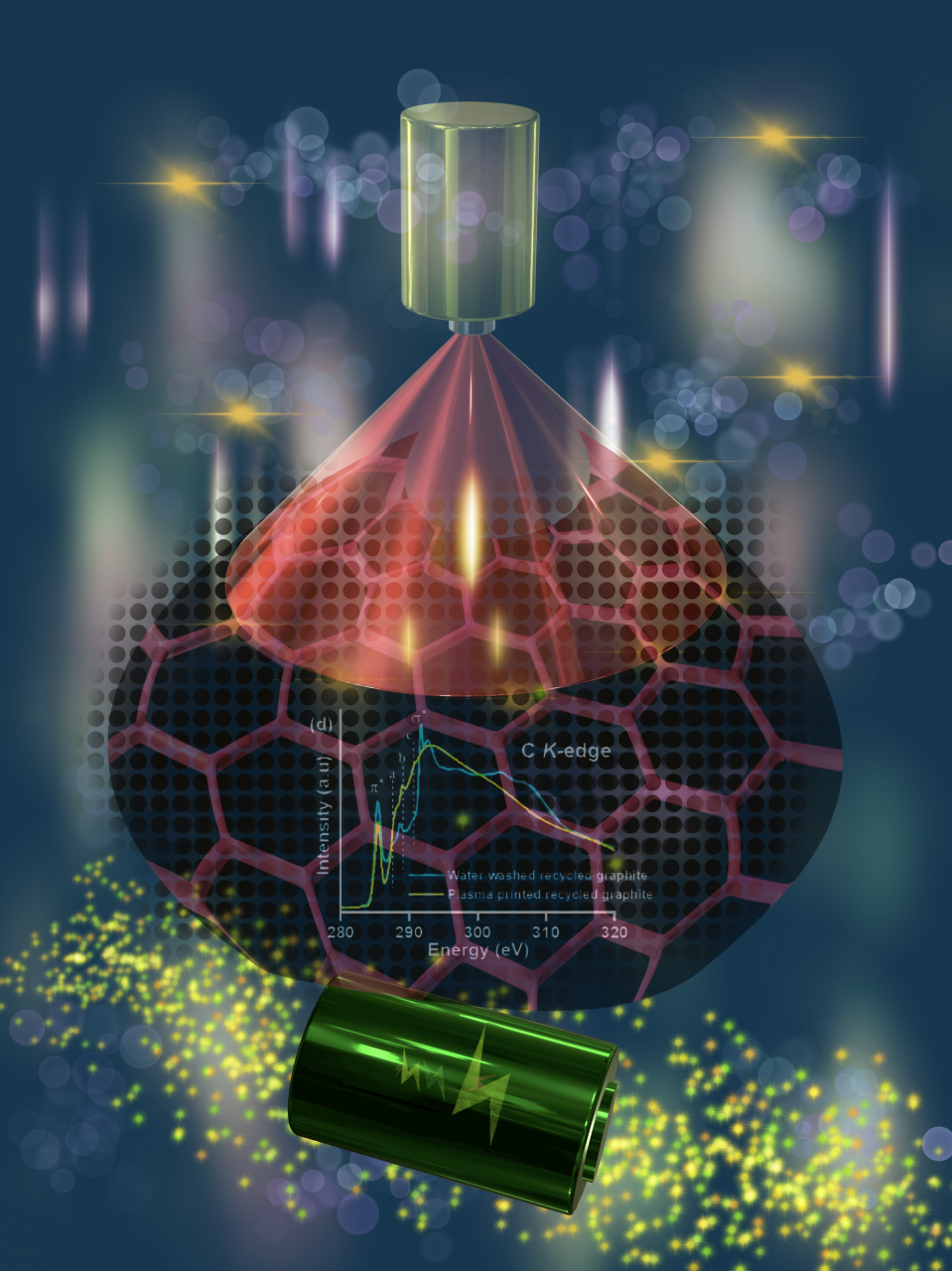
The next step in the process is to turn the recovered graphite back into battery anodes. First, the team dispersed graphite powder into a solvent to create a graphite 'ink'. They then used plasma jet printing, which aerosolises the ink and deposits it onto copper electrodes.
Conventional graphite anode production uses non-conducting binders and conductive additives. These are not required for plasma jet printing, where the deposited graphite sticks well without a binder. The results showed that the plasma jet printed graphite anodes have high specific capacity, good cycling stability, superior rate capability, and excellent electrochemical performance.
Seeking to understand this superior performance, the research team sent their samples to Diamond's B07-C beamline. Dr Satheesh Krishnamurthy of the Open University said;
Due to the Covid pandemic, we carried out our experiments remotely with the help of the beamline staff. We had good discussions with them beforehand and developed this idea with them, so we had complete confidence in using remote access.
We used a combination of synchrotron techniques to understand the chemistry of the plasma jet printed graphite surface. X-ray photoelectron spectroscopy (XPS) shows us the bonding and composition of carbon, and near-edge X-ray absorption fine structure (NEXAFS) spectroscopy offers information on unoccupied density of states.
The results suggest that the improved performance of the plasma jet printed graphite is due to the controlled regeneration of surface chemistry with oxidised graphite and graphitic moieties in the same matrix.
The team will conduct further studies to understand how the plasma jet printing process affects electrode characteristics. This work is part of a larger project to recycle complete LIBs, and even upcycle the materials to produce higher-value products. Efforts to recycle graphite from spent batteries into new anodes is a good step forwards on an essential journey.
To find out more about the B07-C beamline or discuss potential applications, please contact Principal Beamline Scientist Georg Held: georg.held@diamond.ac.uk.
This project is funded by UK India research Initiative and Royal Academy of Engineering, with NILE Limited as Industry partner.
Bhar M et al. Plasma jet printing induced high-capacity graphite anodes for sustainable recycling of lithium-ion batteries. Carbon (2022). DOI:10.1016/j.carbon.2022.07.027.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.