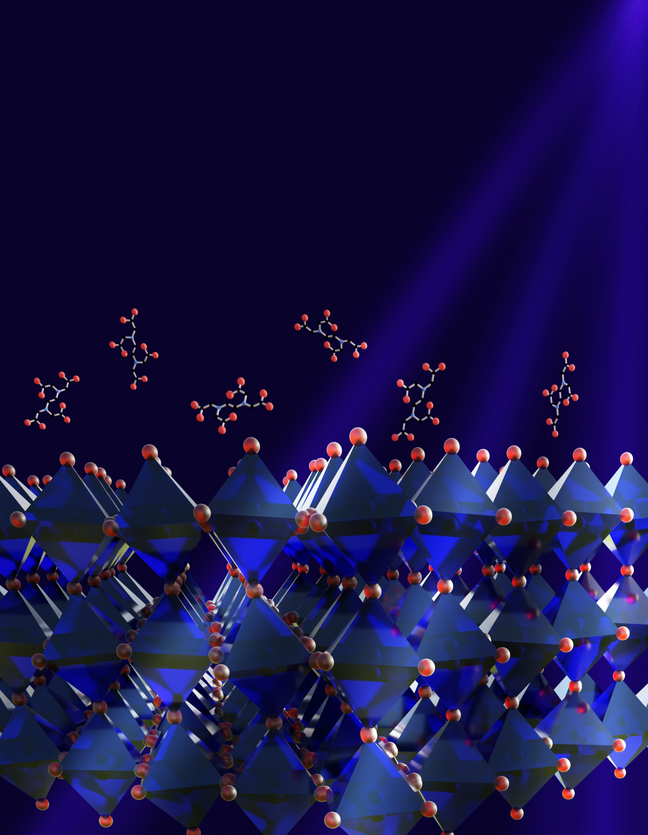
Perovskite materials offer the potential for cheaper optoelectronic devices such as solar cells. Of these, the formamidinium (FA)-based FAPbI3 crystal is one of the most promising - it has a bandgap close to ideal and is very thermally stable. However, photoactive cubic (α)-FAPbI3 perovskite phase is highly unstable and quickly transforms into the non-perovskite yellow phase at room temperature in ambient atmosphere, which affects the performance of photovoltaic devices. Alloying of FA-based perovskite with caesium, methylammonium (MA) cations or a combination of both can keep the perovskite in its more efficient phase at lower temperatures. However, this can give patchy results, leading to power losses. In work recently published in Science, researchers from the University of Cambridge Department of Chemical Engineering and Biotechnology (CEB) and the Cavendish Laboratory investigated the crystal structure of the alloyed perovskite materials to understand why adding cations improved their performance. Their results show that cation alloying induces a minor octahedral tilt that keeps the perovskite material in its highly efficient phase, and is a step towards commercial production of stable and efficient perovskite-based solar cells.
Formamidinium (FA)-based perovskites have much better thermal stability than the methylammonium (MA)-based absorber layers commonly used in early perovskite-based solar cells. FAPbI3 is a particularly promising material, but its photoactive phase is only stable at high temperatures (above 150ºC) in inert atmosphere. It transitions to a hexagonal phase with poor optoelectronic performance at lower temperatures.
It has been shown empirically that alloying FAPbI3 with methylammonium (MA) cations or caesium (or both) improves stability. However, although this approach led to record efficiencies, the mechanism underlying it was not fully understood. It also produces uneven materials with patches of instability that lead to performance losses.
Co-lead author Tiarnan Doherty was a PhD student at the Cavendish Laboratory and is now an Oppenheimer Fellow in CEB. He says:
We wanted to investigate the atomic structure of the alloyed perovskite materials, but they're very sensitive to damage. So we brought the samples to ePSIC for high-resolution electron microscopy with a low electron dose. We also used nano X-ray diffraction on beamline I14. That beamline has very sensitive detectors, which allowed us to achieve our results using low X-ray exposures.
Detailed imaging showed that uneven distribution of the added cations allows the formation of microscopic unstable regions that transition to the hexagonal phase and lead to localised performance losses. It also provided an unexpected discovery of the material structure, which has been widely understood to be cubic. In fact, the researchers found that the alloyed perovskites have a non-cubic structure, with a ~2° octahedral tilting at room temperature. This slight tilt, unresolvable with commonly employed lab-based characterisation techniques, improves the stability of the material by preventing its transition into the hexagonal phase.
Armed with this discovery, the team engineered an octahedral tilt into pure α-FAPbI3 thin films using surface-bound ethylenediaminetetraacetic acid (EDTA). High-resolution imaging showed that the EDTA acts as a template, inducing the growth of octahedral tilt–stabilised FAPbI3 throughout the film without compromising its optoelectronic properties.
Co-lead author Dr Satyawan Nagane explains:
The EDTA templating agent is easily available, stable and non-toxic, and only needed in very small amounts to dramatically improve the stability of the FAPbI3 perovskite films. We will be looking for more generalised understanding of the mechanisms to improve the stabilisation further.
This fundamental study opens up new avenues for further investigation into using organic molecules to improve perovskite stability and performance and is another step towards cheaper solar cells and LEDs.
The senior author on the paper is Dr Sam Stranks, an Assistant Professor at the University of Cambridge. He says:
These are very exciting findings, with important implications for optimising the manufacture of perovskite materials. With the alloying approach, manufacturers need to have precise control over the deposition of the additives to achieve high-quality materials. Using a templating agent has the potential to both simplify and improve the manufacturing process, ensuring a uniform result.
Chris Allen, Principal Electron Microscopist on ePSIC, commented on the group’s use of the electron microscope E02, he said:
We’ve been working closely with the Stranks group for some time now to optimise the imaging conditions on E02 to enable the study of these highly electron beam sensitive materials. This close collaboration between ePSIC staff and the Stranks research team, combined with the excellent performance of our direct electron detectors is enabling new understanding of this fascinating class of materials.
Paul Quinn, Imaging Group Leader at Diamond, said:
The combination of electron microscopy and X-ray nano-diffraction on I14 shows the benefits of multi-scale and multi-technique experiments to understand material properties and scientific impact of combining X-ray and electron microscopy instrumentation at Diamond.
To find out more about ePSIC or discuss potential applications, please contact Principal Electron Microscopist Chris Allen: christopher.allen@diamond.ac.uk. For details about I14, please contact Julia Parker: julia.parker@diamond.ac.uk
Doherty TAS et al. Stabilised tilted-octahedra halide perovskites inhibit local formation of performance-limiting phases. Science 374.6575 (2021): 1598-1605. DOI:10.1126/science.abl4890.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.