The key to keeping organic carbon in our oceans
Jun 10, 2022
Jun 10, 2022
Oceans represent one of the world’s largest reservoirs of organic carbon. Regulating the organic carbon reservoir is critical to the global carbon cycle and is vital to supporting life on Earth; if not maintained this would have substantial impact on the levels of oxygen and carbon dioxide in the Earth’s atmosphere. Minerals are known to protect organic carbon from degradation, conserving organic carbon in soils and sediments. However, there is a lack of understanding of how this protective mechanism actually works.
In recent work published in Nature Communications, a team led by Professor Caroline Peacock, based at University of Leeds School of Earth and Environment, set out to investigate the mechanism behind the conservation of organic carbon in the world’s oceans. The team adopted a multi-faceted approach, incorporating Near Edge X-ray Absorption Fine Structure (NEXAFS) spectroscopy at Diamond’s I08 beamline, to demonstrate that adsorption of organic carbon by clay minerals limits its availability to methane-producing microbes, restricting the conversion of organic carbon into methane. The team’s remarkable discovery sheds light on the mechanism responsible for preserving the oceanic organic carbon reservoir and may have profound implications for our existing understanding of the global carbon cycle.
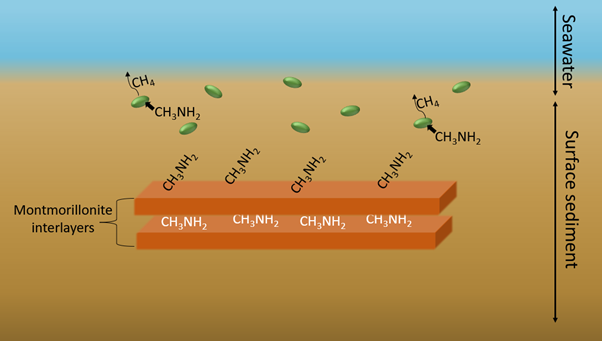
Organic carbon is derived from living organisms, as opposed to inorganic carbon, which originates from ores and minerals and is found in compounds such as carbon dioxide and methane. The balance between the preservation and degradation of organic carbon fundamentally regulates oxygen and carbon dioxide levels in the Earth’s atmosphere. Conversion of organic carbon into methane (CH4) by methane-producing microorganisms (methanogens) that live in marine sediments is responsible for around 29% of global organic carbon degradation occurring below the seafloor. Methylated forms of organic carbon, methylamines, are highly abundant in marine sediments, serving as important sources of carbon and nitrogen for marine microbes. There is widespread assumption that adsorption of organic carbon by minerals in marine sediments protects organic carbon from microbial degradation. However, the mechanisms by which this process may occur have rarely been investigated, where prior work has stopped short of revealing specific chemical and/or physical processes that control uptake of methylamines.
Professor Peacock explains:
We know there is an intimate association between minerals and organic carbon. However, the extent to which minerals protect organic carbon from microbial remineralisation has not been fully tested to a systematic degree. There have been surprisingly few studies which have subjected organic carbon to microbial incubation.

In this work, the team investigated specific mechanistic interactions between particular types of methylamines, clay minerals, and methanogens that co-exist in marine sediments. When methylamines were added to growth medium containing methanogens, methylamines were converted into methane. The introduction of montmorillonite, a clay mineral commonly found in marine sediments, dramatically slowed-down methane production, also reducing the final measured concentration of methane. This effect was seen to increase with increasing montmorillonite concentration, confirming that the clay impeded methane production. Adsorption experiments demonstrated that clay minerals strongly adsorbed methylamines from solution, and in the presence of methanogens, the methylamines were separated into exchangeable and non-exchangeable pools by the montmorillonite.
The team used NEXAFS at Diamond’s I08 beamline to investigate chemical interaction between the methylamines and montmorillonite more closely. NEXAFS can be used to probe how different substances interact by the characteristic appearance of new spectral features when new chemical bonds are formed. Chemical analysis of carbon is particularly challenging, as Professor Peacock explains:
Carbon is notoriously difficult to analyse by a variety of different techniques because it is a very soft scatterer and difficult to chemically see in the presence of hard scattering elements that are often abundant in minerals, like iron. NEXAFS is the only technique with the capacity to spatially map and chemically probe carbon in association with hard minerals at the resolution of the particle sizes we are analysing.
NEXAFS measurements provided evidence for reversible electrostatic bonding of methylamines to clay minerals, with hydrogen bonding accounting for the variation in binding affinity between different classes of methylamine. In addition to reversible adsorption via electrostatic and hydrogen bonds, methylamines were also occluded into clay interlayers. This created a protective barrier, blocking methanogens from reaching the organic carbon, thus forming a pool of methylamines less readily exchanged than those adsorbed at the clay surfaces. Findings also showed that adsorption alone could not fully account for the total reduction in methane concentration; it was suggested that the observed release of iron (II) from iron-rich montmorillonite may further disrupt methanogenesis.
The findings from this study demonstrate some of the first concrete evidence to support the protective adsorption theory i.e. that when organic molecules stick to minerals, the organic molecules are less readily remineralised. Findings suggest that conversion of organic carbon into methane may be further inhibited where montmorillonite concentrations are high, such as the South Pacific Ocean. By revealing how this protective process works, it is anticipated that this discovery may also be widely relevant to interactions between other small organic molecules and microbes, and could have an exciting impact on research into global carbon cycling.
To find out more about I08 or discuss potential applications, please contact Principal Beamline Scientist Burkhard Kaulich: [email protected]
Xiao, K. Q., Moore, O. W., Babakhani, P., Curti, L., & Peacock, C. L. (2022). Mineralogical control on methylotrophic methanogenesis and implications for cryptic methane cycling in marine surface sediment. Nature Communications, 13(1), 1-9.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.