Although we may think that substandard or falsified (SF) medical products are only a problem in the developing world, sales of these products are rising across the globe. Online marketplaces offer easy access to consumers, with counterfeiters labelling products as herbal or food supplements to avoid regulation. Consumers may buy these SF products because they are cheaper or easier to access. However, when taking unregulated products, they risk serious side effects or other health problems. To counter this threat to public health, we need to develop new tools to quickly and accurately detect SF medical products. In work recently published in the International Journal of Pharmaceutics, researchers from the UCL School of Pharmacy investigated three herbal supplements sold online claiming to be natural remedies for erectile dysfunction. Their results showed they all contained sildenafil citrate, the active pharmaceutical ingredient in Viagra®, and were SF products. The study highlights the benefit of employing different analytical techniques to investigate substandard and falsified products, and the need for tighter regulation of therapeutic products on e-commerce websites.
Viagra® is a well-known medication used to treat erectile dysfunction. In the developed world, it's widely available via prescription. In the UK, patients can access the drug over the counter after a quick chat with the pharmacist. So why would they risk buying an unregulated "herbal supplement" online? Some are looking for a cheaper alternative, some are seeking to avoid an embarrassing conversation with a medical professional, and some will have contraindications that mean they might be refused the drug. But there are risks involved in buying online, the first one being - what exactly are they buying?
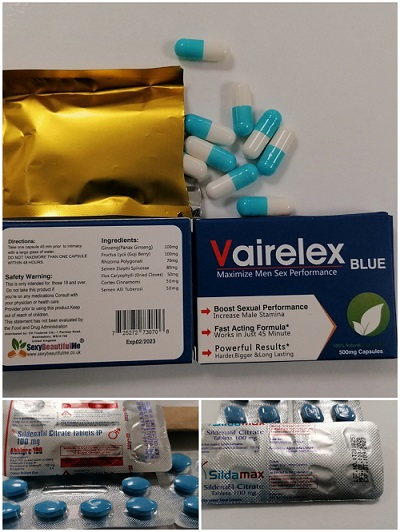
Researchers from the UCL School of Pharmacy purchased three products from a popular e-commerce website, each claiming to be a herbal remedy for erectile dysfunction. Searching the products available, they chose three that had extremely good reviews - more glowing than you would perhaps expect for a herbal supplement.
The WHO classifies three categories of substandard and falsified medicines (SF). Substandard medical products are authorised medications that have failed to meet the specifications or the quality standards. Unregistered/unlicensed products are those distributed without authorisation from the local licensing agency. And falsified medicines deliberately disguise their identity, composition, or source.
In the UK, websites that sell medicines to the public must be registered with the Medicines and Healthcare products Regulatory Agency (MHRA). However, the online sale of herbal or food supplements is not regulated, and that loophole allows the sale of falsified and unlicensed medicines without regulation.
When they arrived, two of the products were packaged as generic sildenafil citrate tablets manufactured in India, different to the advertised packaging. The third came in the same packaging as advertised, proclaiming itself to be a herbal product. But what were they really?

Some counterfeit medicines are produced to be visually identical to the official version. Various lab techniques, including chromatography, spectroscopy and mass spectrometry, can identify and quantify the active pharmaceutical ingredients (APIs). However, the physical properties of the APIs are also important, affecting solubility, bioavailability, dissolution rate and other properties.
More advanced techniques, not commonly used to analyse SF, could provide more comprehensive information, particularly when used in combination. These include powder X-ray diffraction analysis (PXRD), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), dielectric spectroscopy (DS) and Terahertz pulsed spectroscopy (TPS).
Having used lab-based techniques to confirm that all three samples contained sildenafil citrate, researchers from the UCL School of Pharmacy came to Diamond's Joint Engineering, Environment and Processing (JEEP) Beamline I12 to examine them further using simultaneous differential scanning calorimetry-synchrotron X-ray diffraction (DSC-XRD).
The results showed that the two samples labelled as generic sildenafil citrate tablets fulfilled the British Pharmacopeia standards and would be classified as unregistered/unlicensed products. However, the third sample contained around double the maximum daily dose, risking severe side effects for many users. As the packaging was designed to deliberately mislead consumers, it would be classified as a falsified medicine.
Lead author Dr Asma Buanz says:
We've been working in close collaboration with the staff on I12 for around eight years, particularly Dr Oxana Magdysyuk in the past few years, developing techniques to analyse drug samples combining DSC with synchrotron XRD. We began by analysing active ingredients, then polymorphic forms, and now we're using the same techniques to investigate these more complex samples. We've shown that DSC can distinguish fake medicines, and we've verified that result with XRD. The next step will be to find out whether the same result can be achieved with portable DSC equipment, which would make quick 'on the spot' analyses possible.
Taking these SF medicines exposes consumers to serious health risks, with unknown ingredients and inconsistent dosages increasing the risk of overdoses or harmful interactions with other medications. Beyond the individual consumer, counterfeit medicines cost pharmaceutical companies billions in lost sales, with knock-on effects on employment and investment in new products.
The WHO takes a three-fold approach to tackling SF medicines:
This study is a step towards adding new tools to the detection toolbox and making it easier to distinguish between licensed medicines and counterfeit drugs. It also highlights the need for tighter regulation of e-commerce sites and increasing public awareness of the danger of purchasing health products through these websites and how to report fake products.
To find out more about the I12 beamline or discuss potential applications, please contact Principal Beamline Scientist Thomas Connolley: thomas.connolley@diamond.ac.uk.
Ho HMK et al. The Era of Fake Medicines: Investigating counterfeit medicinal products for erectile dysfunction disguised as herbal supplements. International Journal of Pharmaceutics (2022): 121592. DOI:10.1016/j.ijpharm.2022.121592.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.