Zinc oxide (ZnO) is a particularly attractive material for high-tech optoelectronic applications, such as organic LEDs and solar cells, because it is conductive and optically transparent. In addition, zinc oxide is cheap and non-toxic and easy to make and modify. It is also chemically stable, but not inert, and has catalytic and photocatalytic applications. Many of these applications are dependent on how other materials interact with the zinc oxide surface, which is governed by the nature and structure of hydroxyl (OH) groups. Theoretical models assume that the OH groups (formed by zinc oxide reacting with air or water) are found on the zinc oxide surface. However, when researchers investigating the material using a novel technique on Diamond's high-resolution I09 beamline discovered that the OH groups weren't where they were expected to be. Their results, recently published in Physical Review Materials, will lead to a better understanding of this useful material.
Zinc oxide can be tailored for many different applications, many of which depend on how other materials interact with the zinc oxide surface. This interaction is governed by the presence of hydroxyl (OH) groups, which form due to interactions with the air or water. However, investigating the nature and structure of these OH groups requires a highly sensitive instrument that can distinguish between the oxygen atoms in the OH groups and those in the zinc oxide itself. Dr Jens Niederhausen of Helmholtz-Zentrum Berlin and his colleagues from the University of Tübingen and Soochow University brought their ZnO samples to Diamond's I09 beamline, which is designed for high-resolution studies of atomic and electronic structures of surfaces and interfaces.
In the X-ray Standing Waves (XSW) technique, a beam of X-rays directed at the sample under investigation reflects back. The incident and reflected waves combine to form an interference pattern. The resulting waveform appears to be standing still, in that the peaks and troughs do not move in space. For these experiments, Dr Niederhausen combined XSW with X-ray Photo Emission Spectroscopy (XPS), which is a surface-sensitive technique that measures the electrons emitted from a surface in the X-ray beam.
Dr Niederhausen explains:
We use the standing wave like a ruler. Where the X-rays hit the sample, they cause the emission of electrons, and we measure the electron flux. The electron flux is higher if the OH groups are located in an X-ray peak, and lower if they are in a trough, and so we can see where they are relative to the zinc oxide surface.
David Duncan, the senior beamline scientist on I09, carried out a complementary experiment on the same samples, using Photoelectron Diffraction (PhD). Dr Niederhausen continued:
Having PhD available at the same end station is a real bonus, and we were very grateful for David's expertise to help us carry out this additional experiment. The PhD results really helped us to develop our understanding of the surface structure.
Theoretical models tell us that the OH groups are on top of the ZnO surface. However, the results of these experiments did not agree. The OH groups weren't where they were expected to be.
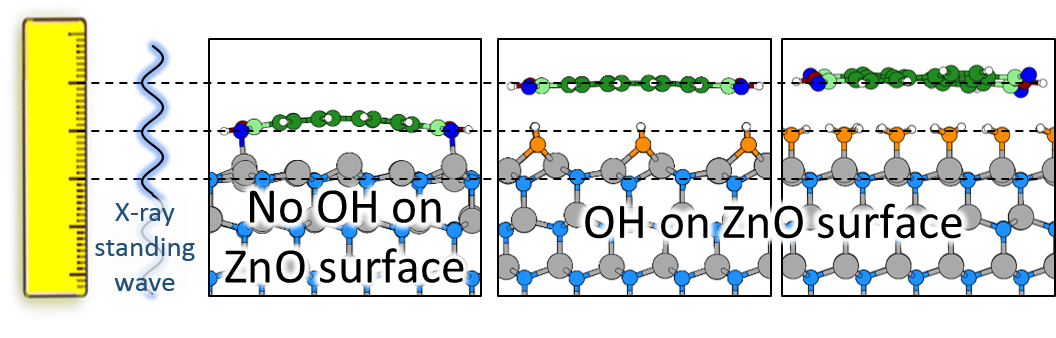
There were no theoretical models that explained the experimental results. However, measuring the vertical position of the OH groups above the ZnO surface assumed that the ZnO surface was perfectly flat. Real-world conditions are a little less ideal, so Dr Niederhausen developed a new technique to get more accurate measurements.
He looked for an organic molecule that could form as complete and pure a monolayer on the zinc oxide substrate as possible. The perfect choice turned out to be PTCDI, which is a stable, planar (2D) molecule. He repeated his experiments with the new samples, measuring the vertical distance between the OH groups and the PTCDI monolayer. Again, the results showed that the OH groups were not on the zinc oxide surface.
Were they acting as a spacer between the two different materials? Researchers from Graz University of Technology in Austria carried out state-of-the-art computational physics to model that scenario. Again, the model did not match the experimental results. In contrast, assuming direct contact between PTCDI and the ZnO surface gave a very good match. So we don't know the structure of the OH groups on this ZnO surface, but we can rule out that they sit on top.
Dr Niederhausen is excited by these unexpected results. He says:
Our best models tell us that the OH groups are on the top of the ZnO surface, but they assume ideal crystals in the most stable state. These results show us that we need to question those assumptions and to develop more advanced theories. This new technique is excellent for surface probes, and could prove useful for other materials – but probing the OH groups in their sub-surface locations will require other clever approaches.
To find out more about the I09 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Tien-Lin Lee: tien-lin.lee@diamond.ac.uk.
Niederhausen J et al. X-ray standing waves reveal lack of OH termination at hydroxylated ZnO (0001) surfaces. Physical Review Materials 4.2 (2020): 020602. DOI:10.1103/PhysRevMaterials.4.020602.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.