The structure of a photosynthetic protein complex that absorbs infrared light in aquatic bacteria has been determined for the first time, using cryo-electron microscopy (cryo-EM) at the Electron Bio-Imaging Centre (eBIC) at Diamond Light Source. The work outlined in the acclaimed journal, Nature, showed every component of the photosynthetic complex to an impressive 2.9 Å resolution.
In photosynthesis, light harvesting (LH) complexes funnel absorbed solar energy to reaction centre (RC) complexes, which transiently trap and store the harvested energy in the form of a charge separated state. Thereafter, a series of electron and proton transfers within the RC converts a quinone acceptor to its reduced form, a quinol.
Plants and algae usually absorb light energy at wavelengths of up to 700 nm, but one particular phototrophic bacterium Blastochloris (Blc.) viridis can absorb and utilise wavelengths far greater than these. Remarkably, these bacteria have adapted RCLH complexes with the capacity to absorb energy above 1000 nm – near infrared light that is inaccessible to other photosynthesisers.
To understand the structural basis of this extraordinary infrared absorption, Dr Pu Qian and Professor Neil Hunter FRS from the University of Sheffield joined forces with Dr Alistair Siebert at eBIC to apply cryo-EM to determine the structure of the RCLH1 complex. Their 2.9 Å resolution structure revealed the positions of all bacteriochlorophyll, carotenoid, haem and quinone cofactors, as well as the protein-protein interactions that form the structural basis for infrared light absorption, charge trapping electron transfers and export of the quinol product.
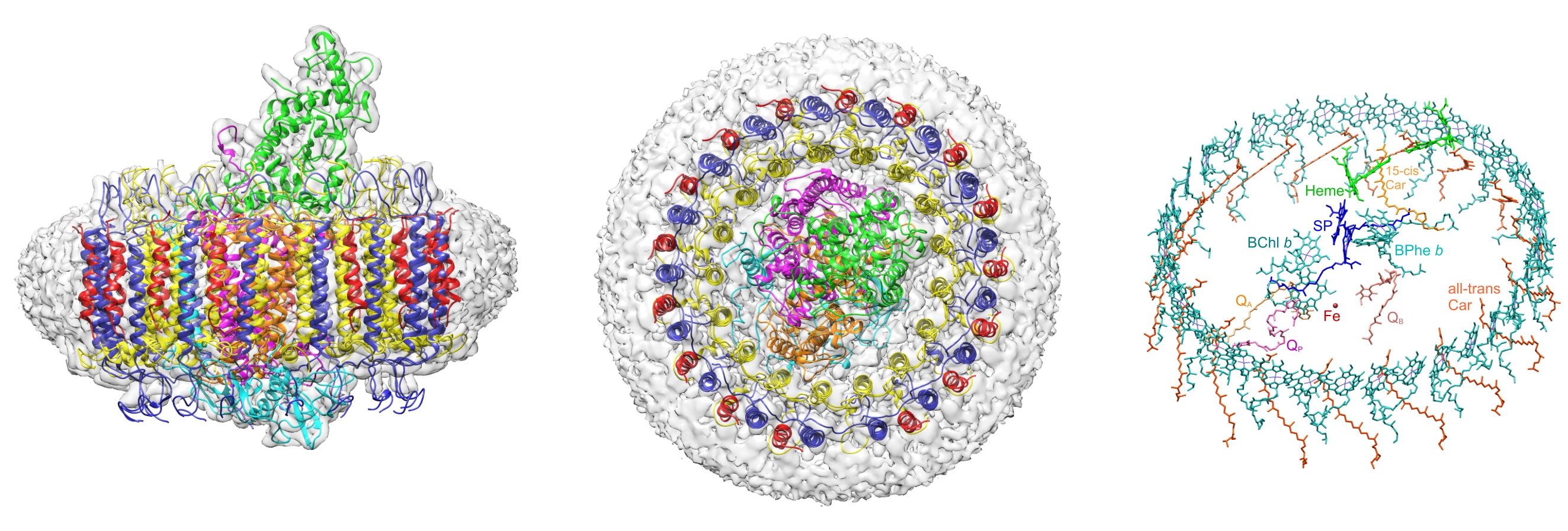
Figure 1. (Left) The cryo-EM structure of the Blc. viridis RCLH1 complex, with the electron density map in grey and ribbon representations of LH1-α (yellow), LH1-β (dark blue), LH1- γ (red), RC-C (green), RC-H (cyan), RC-L (orange) and RC-M (magenta).The LH1 complex is surrounded by a belt of detergent and other disordered molecules. (Middle) Top view of the periplasmic face of the complex. The ‘missing’ LH1- γ in the outer ring at the bottom of the figure is the location of the pore for quinol export and for oxidised quinones to gain access to the RC. (Right) Closely spaced LH1 bacteriochlorophyll b (sea green) and carotenoid (orange) pigments surrounding central RC pigments, as well as haem cofactors (green) for electron transfer to the RC, and quinones (Q) for transport of protons and electrons away from the RC.
Photosynthesis in the near-infrared
Plants and algae use chlorophyll a to absorb solar energy at wavelengths up to 700 nm. However, it has been known for decades that some phototrophic bacteria can make use of other parts of the solar spectrum, by absorbing light in the 750–900 nm region. Most of these bacteria use bacteriochlorophyll a, which absorbs at around 770 nm in methanol, and their RCLH1 complexes are red-shifted to 870–880 nm.
Blc. viridis uses bacteriochlorophyll b (BChl b), which in its pure form in methanol absorbs at 795 nm, and in principle should push photosynthesis further to 900 nm. Remarkably, this organism squeezes a red shift of 220 nm from these pigments, enabling photosynthesis to occupy a spectral niche above 1000 nm. The central RC component of the Blc. viridis photosynthetic complex was the first ever membrane protein to be crystallised; however the structure of the RCLH1 complex in its entirety had eluded scientists until now.
Scientists from the University of Sheffield took a different approach, seeking to solve the structure with the application of cryo-EM. Principal investigator of the study, Prof Hunter, explained their aims: “We wanted to understand the whole complex because the reaction centre alone cannot harvest enough light - it needs the belt of BChl b around it to absorb and deliver the energy. For 30 years, people could not grow crystals of the whole complex, so we used cryo-EM to circumvent this problem.”
Reasons for the red shift
The cryo-EM structure of the Blc. viridis RCLH1 complex showed that the organism employs an intriguing range of strategies to allow it to harvest light at 1000 nm. Just BChl b alone was not sufficient for absorption; the structure showed that H-bonding between BChl b and LH1 Trp residues was responsible for some of the red shift. Close packing between the BChls in the outer ring that surrounds the RC also contributed.
More importantly, the structure revealed that the bacterium had a novel triple ring LH1 complex. “The additional outer gamma subunit acts as a stabilising force that helps promote the red shift, and no other complex ever seen has a comparable subunit,” remarked Prof Hunter.
The great escape
The product of energy trapping is a quinol molecule, which must escape the confines of the LH1 ring that surrounds the RC. The 2.9 Å structure obtained at eBIC showed how a quinol traverses the LH1 ring surrounding the RC.
Prof Hunter described their observations: “On a first look, the LH1 complex appears to completely surround the RC, giving no opportunity for a quinol to escape or for an oxidised quinone to pass the other way and gain access to the RC. However, there is a small pore in the LH1 ring, near a binding pocket, which houses a quinol, designated QP. This binding pocket provides afolding template that imposes a compact conformation and a suitable orientation on QP prior to its entry to the pore in the LH1 ring. Thus, the RC-LH1 complex of Blc. viridis has revealed a new strategy for fostering quinol/quinone exchange across an LH1 ring.”
The structural insights gained from this study could enable near infrared light absorption to be engineered into biosynthetic, bioinspired or biohybrid photosynthetic structures. In particular, bacteria could be adapted to have a wide input of solar energy to be used for solar powered cell factories.
The team will continue looking at this bacterium and they plan to apply genetic engineering to remove the critical outer ring to see if they can force the complex to absorb light towards visible wavelengths. Building on the huge success of this structural study at eBIC they will also explore other photosynthetic systems using cryo-EM.
To find out more about eBIC, or to discuss potential applications, please contact Dr Alistair Siebert: alistair.siebert@diamond.ac.uk
Qian P et al. Cryo-EM structure of Blastochloris viridis LH1-RC complex at 2.9 Å. Nature 2018. DOI:10.1038/s41586-018-0014-5
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.