 Conventional X-ray imaging, as used in hospitals and airports, relies on objects absorbing radiation as it passes through them. Dense features in the object (e.g. bones) absorb most of the radiation, and project a dark shadow on the radiograph, while less dense features (e.g. soft tissue) are more transparent to X-rays and thus create a brighter region on the image.This method works well for many applications, but it is known to fail when details with similar density (e.g. different parts of soft tissues) need to be visualised.
Conventional X-ray imaging, as used in hospitals and airports, relies on objects absorbing radiation as it passes through them. Dense features in the object (e.g. bones) absorb most of the radiation, and project a dark shadow on the radiograph, while less dense features (e.g. soft tissue) are more transparent to X-rays and thus create a brighter region on the image.This method works well for many applications, but it is known to fail when details with similar density (e.g. different parts of soft tissues) need to be visualised.
A different kind of X-ray technique that can show the inner structure of soft tissue in great detail is now available on Diamond’s X-ray Imaging and Coherence beamline, I13. The technique called grating interferometry is a form of X-ray phase contrast imaging (XPCI); a method that analyses changes in the cycle of a beam’s waveform (its ‘phase’) to ascertain variations in a sample’s density.
Due to its higher sensitivity to small density differences, phase contrast imaging often yields superior results compared to absorption imaging, and has become an established method for investigating samples such as biological soft tissues and alloys.
Grating interferometry on I13
X-ray phase contrast imaging (XPCI) is a powerful method to access structures invisible with conventional absorption contrast. This method relies on the high sensitivity of the phase information, usually obtained through interference of the x-ray waves, to generate the image contrast. In this way details with small density differences, such as cancerous areas within healthy tissue, become clearly visible.
Several methods for performing phase-sensitive imaging have been developed in the last decades, including grating interferometry. This technique involves placing a grating between the sample and the detector, and measuring the resultant wave interference pattern with sophisticated image processing algorithms to generate a high-resolution image.
Due to the high coherence of beamline I13, it has been possible for Diamond’s research staff to test and prove its capacity for grating interferometry, giving I13 users an incredibly powerful tool for high-contrast and high-resolution imaging of soft tissue ultrastructures. Two independent operating branches are available for this type of research, the ‘coherence’ and the ‘imaging’ branches. The latter is known as the Diamond Manchester Imaging Branchline (I13-2), co-funded by the University of Manchester.
Who’s been working on this?
Dr Irene Zanette, a Postdoctoral Research Associate on I13, has been working to develop the grating technique with her colleague Marie-Christine Zdora, a joint-funded PhD student between Diamond and University College London (UCL). They have been using mouse embryos in paraffin wax provided by Dr Andrew Cook, Cardiac Morphologist based at UCL, and achieved some exciting results from their grating interferometer experiments. The extremely high-contrast, high-resolution images are comparable to those that can be obtained with commonly used destructive methods such as histopathology and High Resolution Episcopic Microscopy – but with the added bonus of not damaging the samples.
 Fig 1. Experimental setup on I13
Fig 1. Experimental setup on I13
Marie-Christine has recently presented the findings at the 23rd International Congress on X-ray Optics and Microanalysis (ICXOM23), and you can view her poster here.
Irene notes that I13 offers the ideal conditions for developing these phase-contrast methods. “The I13 beam is relatively large and has a very high coherence level. The single phase grating produces a reference interference pattern that is directly resolved by the detector. We now have a stable setup (Fig 1) with phase images of histology-like quality (Fig. 2), and are ready to accept applications for beamtime for this technique”.
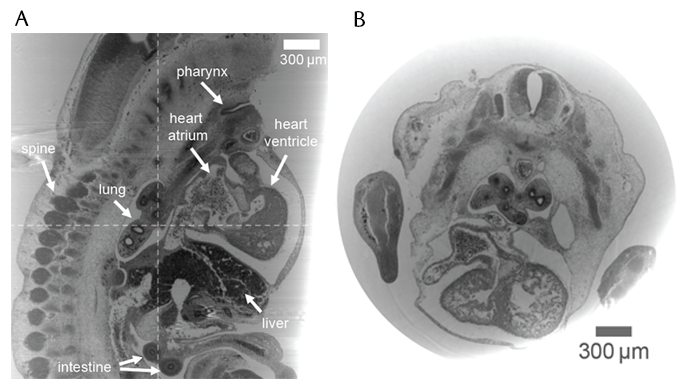
Fig 2. Grating interferometry results on a mouse embryo in paraffin wax: A) sagittal slice, B) axial slice.
Phase contrast imaging of muscle cells in hearts
Samples were provided by Dr Cook, Senior Lecturer at UCL’s Institute of Cardiovascular Science. He works to improve understanding and prenatal detection of congenital heart disease, and has been using XPCI on I13 to look at the detailed structure of cardiac muscle cells (myocytes) in mouse and human hearts. Not much is known about these myocytes as yet, but they are complex branching muscle cells that are organised in a certain spiral pattern in the heart, and change orientation as the heart beats: “I13 has been able to give us the resolution needed to study ultrastructure of the hearts, in particular the way the myocytes are organised, and the direction they take within the myocardium.”
 Dr Andrew Cook from UCL preparing samples in the I13 lab.
Dr Andrew Cook from UCL preparing samples in the I13 lab.
For more information on the I13 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Professor Christoph Rau: christoph.rau@diamond.ac.uk
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.