X-ray spectroscopy techniques at Diamond have given scientists a new insight into the behaviour of uranium during deep disposal of radioactive waste.
Many countries will use geological disposal facilities as the final way to dispose of radioactive wastes and so it is important to be able to predict the behaviour and potential impacts of uranium under alkaline conditions relevant to deep disposal. The effectiveness of iron (oxyhydr)oxides to reduce the mobility of uranium is well known due to their high surface reactivity. However, the fate of surface bond uranium during the crystallisation of these minerals is poorly understood.
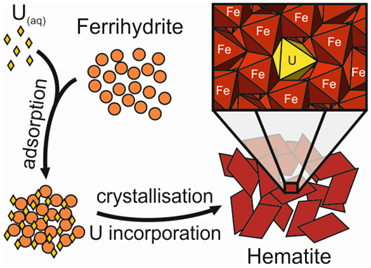
Image: Adsorption of uranium onto the surface of ferrihydrite, an amorphous iron oxyhydroxide, leads to incorporation of uranium into the structure of the iron oxide hematite during crystallisation. EXAFS data obtained at B18 clearly show the changing bonding environment of the uranium, from adsorbed to incorporated, during crystallisation.
The common iron oxide hematite is ubiquitous in waste management scenarios, and is likely to hold a particular importance here due to its long-term stability. With the prospect of exposure to heat-yielding radioactive waste and geothermal fluctuations, hematite could be important in uranium immobilisation as it can be locked up within the mineral structure.
The team of researchers from the University of Manchester and Diamond Light Source used spectroscopy techniques on Diamond’s B18 beamline to explore these processes. Combining X-ray Absorption Spectroscopy alongside chemical extraction and transmission electron microscopy techniques at the labs in Manchester, they have been able to gather the first evidence for how absorbed U(VI) – uranium in its more mobile oxidation state – becomes incorporated into hematite. The Extended X-ray Absorption Fine Structure (EXAFS) technique also allowed the exact mechanism of uranium incorporation to be determined, showing that it directly substitutes for iron, with little distortion of the surrounding crystalline structure.
The combined results of the study have afforded a full mechanistic understanding of uranium incorporation into hematite, and the nature of uranium bonding within the mineral structure. Together these findings highlight that minerals can be used to lock away radioactive contaminants and thus may contribute to controlling the environmental impact of radioactive waste disposal.
Dr Sam Shaw, one of the research scientists on the project said, "Understanding how contaminants interact with the geological environment is central to underpinning the case for nuclear waste disposal. The results generated at Diamond have shown us that uranium can become incorporated inside the actual mineral itself, not just absorbed onto the surface. This is a big step in a global understanding of contaminant behaviours, and is an important contribution to the field."
Dr Sam Shaw, Research Centre for Radwaste Disposal, and Williamson Research Centre for Molecular Environmental Science, University of Manchester
The research was by funded the UK National Environmental Research Council (NERC) BIGRAD consortium, which is led by Prof. Katherine Morris from the University of Manchester and brings together leading researchers from a number of universities and research organisations with the objective of understanding the behaviour and control of radionuclides in geological disposal facilities.
Further beamtime for the BIGRAD Consortium has been allocated for later in the year to analyse, for the first time at Diamond, samples containing the transuranic radionuclide neptunium, which is formed in nuclear reactors. The aim is to understand the fate of neptunium during biogeochemical reactions relevant to management of radioactively contaminated environments.
To find out more about using the B18 beamline, or to discuss potential applications, please contact Dr Andy Dent: [email protected]
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.