Analysing functional DNA nanomaterials with SRCD spectroscopy
Feb 6, 2013
Feb 6, 2013
Brewer, A., Siligardi, G., Neylon, C., Stulz, E. Introducing structural flexibility into porphyrin–DNA zipper arrays, Org. Biomol. Chem. 9, 777–782, (2011)
DNA is, besides displaying its central biological role as bearer of the genetic material, an extremely versatile construction material. DNA has become very attractive for the creation of novel nanosized objects. We are studying the influence of modifications of DNA with the aim to create functional DNA based nano-materials. Our modifications generally consist of large chromophores, which show specific light-induced activity such as energy or electron transfer. We are using modifications which are derived from naturally occurring molecules, such as porphyrins. Both DNA and the porphyrins show specific response when measured using circular dichroism (CD) spectroscopy, where the UV part gives insight into the structure of the DNA, and the visible part displays important electronic information on the porphyrin. By making use of the sensitivity of the beamline B23 SRCD spectroscopy station, we have analysed a number of different DNA strands. The CD spectra are also extremely valuable in determining the presence of different metals within the porphyrins; metallation of porphyrins can be used to change their optoelectronic properties. In addition to the porphyrins, we are also investigating other metal-binding substituents which can be used to cross-link DNA strands together to create extended structures held with complementary interactions.
The synthesis of electronically active organic molecules on the macromolecular scale is of major interest due to their promising applications in light harvesting devices, photovoltaics, logic gates and molecular wires. The limiting factor in the development and refinement of these systems is often the speed of fabrication, since the introduction of a minor change to the final compound may dictate a completely different synthetic pathway. One solution to this is the development of modular building blocks, whereby the system as a whole can be tweaked and optimised through the relatively facile substitution of a different building block during the synthesis. Nature provides us with an abundant, easily modified scaffold upon which to base our building blocks, namely DNA. A great deal of research has been conducted on modified DNA, with many different electronically active molecules being covalently bound 1. Our focus is on the multiple attachments of porphyrins and metal complexes to the DNA scaffold. Porphyrins are an ideal choice for this purpose due to the facile tunability of their electronic properties through modification of the attached substituents or through metallation of the porphyrin macrocycle and they can easily be attached to a variety of scaffolds. The same properties are true for terpyridine based metal complexes, hence provide a second very interesting modification unit.
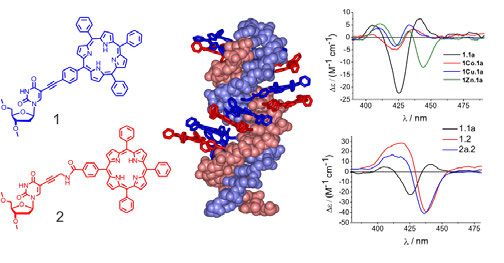 |
| Figure 1: Left: structure of the porphyrin modifications with either rigid (1) or flexible (2) attachment; middle: modelled structure of the modified rigid-flexible hybrid-DNA (1.2) showing the even distribution of the porphyrins along the DNA major groove; right: CD spectra of metallated rigid-rigid DNA (without metal 1.1a, and with metal Co, Cu or Zn; only one of the complementary strands is metallated) (top) and CD spectra of the rigid-rigid (1.1a), rigid-flexible (1.2) and flexible-flexible (2a.2) porphyrin-DNA (bottom). |
Previous research in our group has focused on tetraphenyl porphyrins connected via a rigid acetylene linker (Fig. 1). We are still holding the record for attaching the largest number (11) of such a large and hydrophobic substituent onto DNA.2 Positioning all modifications on one strand leads to a destabilisation of the duplex, whereas when positioned on alternate strands (creating a zipper system) the modifications provide a significant degree of stabilisation to the system3,4. The rationale for an enormous stabilisation effect of > 40 °C in the zipper system is in the π-π-stacking of the hydrophobic aromatic porphyrin molecules, leading to a positive interaction and outweighing the structurally unfavourable changes in the DNA duplex, which are inherently induced by such a large substituent. We have extended the concept to introduce a more flexible and longer propargyl-amide base connection. Molecular modelling shows an even distribution of the porphyrins along the major groove of the DNA (Fig. 1); CD spectroscopy has proven an invaluable tool to determine the structural influence of the new linkers on the DNA. Of particular importance was, in this case, the large electronic difference in the visible range, which arises from both different attachment and different metallation states. These features are not easily detectable using standard UV-vis spectroscopy.
 |
| Figure 2: Terpyridine-DNA building block with DNA sequences (top). UV-vis and CD spectra are displayed at the bottom, showing the spectral differences between the modified and natural DNA in the presence and absence of metal (zinc). |
Alongside the porphyrins, transition metal complexes based on terpyridine (terpy, Fig. 2) ligands are inherently interesting to study due to their large diversity in both electrochemistry and photophysics. We have recently synthesised the corresponding nucleoside-terpy building block and incorporated it site specifically into DNA. To probe their efficiency as metal chelators, we have designed a supramolecular system which makes use of orthogonal binding modes to assemble nano-sized DNA structures (Fig. 3)5.First, the DNA strands were designed to be in part self-complementary with overhanging 'sticky ends'. Those sticky ends, which are again complementary to each other, will selectively anneal to form the DNA duplex according to the highly selective and specific base pairing. The result is that the strands, once mixed, will spontaneously form an almost infinitely long DNA strand. Or so we thought: AFM pictures revealed that the linear terpy-DNA actually folds into well-defined nano-rings of 50 to 100 nm diameter. By strategically placing terpy units into the strands, the metal binding units can now be used to connect different strands, and with it form an extended array of DNA. Again, CD spectroscopy has proven very valuable to follow the metal binding event. Furthermore, since the optical window of the chelator is outside that of the DNA, and seems very sensitive to the environment of the structure, it will provide a structural handle for further exploration. However, the combination of the base pairing and selective metal binding leads to the formation of nano-tubes, which are 50-200 nm wide and 2-50 nm high, and B23have a length of several micro meters. This result can, to a certain extent, be explained by the fact that the terpy units are not positioned at exactly 180° in the DNA duplex, but rather form an angle of about 130°, thus facilitating the formation of tubular structures rather than flat sheets. Nevertheless, the directed formation of DNA nano-arrays through orthogonal self-assembly is still a very promising approach to obtain new functional materials.
 |
| Figure 3: Putative representation of the annealing of long DNA strands through sticky-end recognition and formation of tubular assemblies by metal complexation (including AFM picture). |
The modifications, which we attach to the DNA and successfully characterise using the excellent facilities available at Diamond Light Source, are now under further investigation as optical handles to analyse DNA in more biological environments. As an example, we have designed molecular rulers based on porphyrin-DNA, which allows the analysis of DNA on a nano-meter resolution; this ruler is now being applied to the study of DNA protein interactions.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.