De Santis, E., Hjelmgaard, T., Faure, S., Roy, O., Didierjean, C., Alexander, B. D., Siligardi, G., Hussain, R., Javorfi, T., Edwards, A. A. & Taillefumier, C. Cyclic α,β-peptoid octamers with differing side chain patterns: synthesis and conformational investigation. Amino Acids. 41 (3), 663-672 (2011)
The three dimensional structure (i.e. conformation) of natural proteins and peptides plays an important role in their biological activity and has led to the development of non-natural backbones which mimic such conformations and offers therapeutic advantages. Peptoids are peptidomimetics where the chemical modification is responsible for desirable chemical, physical and biological properties. The presence of tertiary amides in the peptoid backbone results in complex NMR spectra due to cis/trans isomerism of the amide bond. As a result, chiroptical techniques become essential tools for conformational investigation. Our study focuses on the conformational investigation of novel alternating α,β-peptoids to develop improved peptidomimetic scaffolds.
Peptoids are peptidomimetics, where the side chain is moved from the α-carbon to the adjacent amide nitrogen, generating a tertiary amide. This chemical modification is responsible for desirable chemical, physical and biological properties, e.g. different interactions along the backbone, flexibility, resistance to proteases and higher cell permeability. The presence of the tertiary amide is also responsible for the low energy barrier for the cis/trans isomerism of the amide bond, thus complicating NMR spectra. As a result, chiroptical techniques become essential tools for conformational investigation.
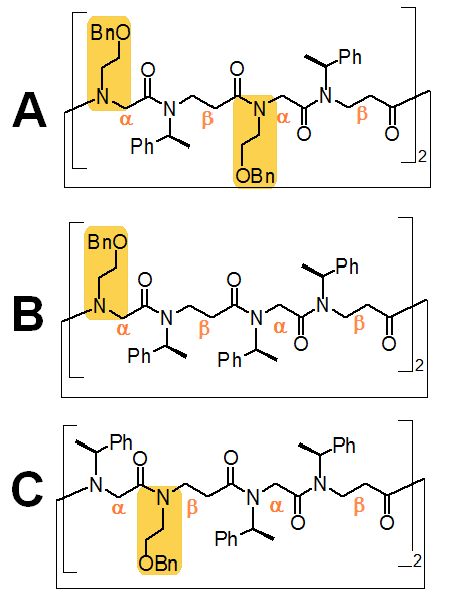 |
| Figure 1: Cyclic peptoid octamers A, B and C with different side chain patterns. (Bn: benzyl; Ph: phenyl). |
Our study focuses on the conformational investigation of novel alternating α,β-peptoids to develop improved peptidomimetic scaffolds for the display of carbohydrate antigens. The use of circular dichroism was essential for the conformational analysis of these novel compounds as the far UV below 200 nm was the most characteristic region and the high signal/noise ratio in this spectral region was essential for the understanding of the data.
Peptoids are structurally related to natural α-peptides, where the side chain is moved along the backbone from the α-carbon to the adjacent amide nitrogen.1 Compared to peptides, they have key advantages; such as resistance to enzymatic hydrolysis1 and improved cell permeability.2 In addition, peptoids are not immunogenic3 and this is essential for the development of peptoid-based therapeutics. Due to these properties, peptoids have a vast potential for therapeutic application, some of which have already been considered to date and include mimicry of antimicrobial peptides and lung surfactant protein4 C.
The biological activity of natural peptides and proteins is related to their sequence composition as well as to their three dimensional structure. Therefore, when generating synthetic peptidomimetics, their ability to fold into a three dimensional structure (i.e. conformation) needs to be considered and their conformational preference investigated. However, the conformational investigation of peptoids is complicated by their inherent flexibility due to the presence of tertiary amide bonds which are devoid of H-bonding donors. Thus the H-bonding potential of the peptoid backbone is significantly reduced. Also, the energy barrier for cis/trans isomerisation of the tertiary amide is decreased relative to the secondary amide in peptides.5 As a result, interpretation of NMR spectra is generally complicated, and chiroptical techniques, such as circular dichroism, become essential tools for the conformational investigation of peptoids.
The use of SRCD was essential for the conformational analysis of these novel compounds as the far UV below 200 nm was the most characteristic region and the high signal/noise ratio in this spectral region was essential for the understanding of the data.
Our research has focused on investigation of the conformational preference of novel α,β-peptoids, which are composed of alternating α- and β- peptoid residues. In our previous work we have shown that, despite the decreased hydrogen-bonding potential of the peptoid backbone, linear and cyclic α,β-peptoids can adopt more than one ordered conformation6 and these can be perturbed by different solvent environments and temperatures.
In this study, we investigated the effect of different side chain patterns on the spectral features and conformational preference of α,β-peptoids.7 Three cyclic peptoids with different side chain compositions were selected for the study (Fig. 1).
Preliminary 1H NMR studies were undertaken in CDCl3, CD3CN and CD3OD but NMR signals could not be assigned due to the complexity deriving from the presence of cis and trans isomers of the tertiary amide in the NMR time scale. Despite this, octamer C had the greatest resolution in the 1H NMR spectra and particularly in CD3OD and CDCl3. This was indicative of a smaller area of conformational space being sampled by C, i.e. a reduced conformational heterogeneity thus an increased population of some conformers. Notably, C also crystallised from methanol. To further investigate these observations, synchrotron radiation circular dichroism (SRCD) studies were undertaken. The use of SRCD was essential for the conformational analysis of these novel compounds as the far UV below 200 nm was the most characteristic region and the high signal/noise ratio in this spectral region was essential for the understanding of the data.
  |
| Figure 2: SRCD spectra of (a) cyclic octamers (A-C) with differing side chain sequences in MeOH and (b) octamer C in different solvents. All spectra were recorded at 20 °C at known concentrations in the range 205 - 255 μM using a cylindrical cell of 0.01 cm pathlength. |
SRCD studies of A-C in MeCN showed similar spectral features (data not shown). By contrast, the SRCD spectra in MeOH showed that C had the greatest intensity of the molar ellipticity compared to other cyclic peptoids with different sequence patterns (Fig. 2a). This indicated that the side chain pattern promoted adoption of a preferred conformation or an ensemble of closely related conformations in MeOH. The same trend was also observed in 2,2,2-trifluoroethanol (TFE), whilst different spectral features were observed in 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP) (Fig. 2b).
Octamers B and C, which have the same percentage of chiral side chains and only differ by the position of the N-2-(benzyloxy)ethyl side chain respectively on the α- or β- residue (Fig. 1), were studied at variable temperatures (data not shown) to give further insight into the effect of the side chain composition. Temperature studies in MeOH showed a greater change in molar ellipticity upon heating for C by contrast to B. A similar result was obtained in the other protic solvents investigated (TFE and HFIP). By contrast, the degree of change in MeCN (aprotic) was similar for B and C. This confirmed that the sequence pattern of C (with the N-2-(benzyloxy)ethyl on the β-residue) determined the adoption of a more ordered conformation.
 |
| Figure 3: Titration of compound C in MeOH with increasing percentage of MeCN. All spectra were recorded at 20 °C in the solvent stated at the concentration of 229 μM using a cylindrical cell of 0.01 cm pathlength. |
To further explore the greater conformational order demonstrated for C in MeOH by contrast to MeCN, a solvent titration from MeCN to MeOH was undertaken. Interestingly, rather than the anticipated gradual increase of ellipticity with increasing MeOH, the most intense ellipticity was observed in 1:1 and 1:3 mixtures of MeCN : MeOH, and not in neat MeOH (Fig. 3). This indicated that the greatest conformational stabilisation occurred in the presence of a mixture of protic/aprotic solvents and can be explained by an intricate balance of inter- (i.e. solvent-peptoid) and intra-molecular interactions determining a favourable ‘ordered solvation’ where the conformational stabilisation is enhanced in a mixture of MeOH and MeCN rather than for each solvent in isolation.
  |
| Figure 4: Titration of compound (a) B and (b) C in MeCN with increasing percentage of HFIP. All spectra were recorded at 20 °C in the solvent stated at 214 μM for B and 208 μM for C using a cylindrical cell of 0.01 cm pathlength. |
Solvent titrations with MeCN and HFIP were also undertaken for both B and C (Fig. 4). It was found that in both cases the spectral features in MeCN were largely maintained until 100% HFIP where a conformational change was then observed, most clearly for C (Fig. 4b). However, a slight increase of ellipticity was observed for the 1:1 and 1:3 mixtures of MeCN : HFIP, suggesting that a combination of protic/aprotic solvents can increase conformational order but that a small amount of HFIP was not sufficient to induce the significant conformational change observed in neat HFIP.
The results of this study clearly demonstrated that the side chain patterns can affect the conformational preference of the α,β-peptoid backbone. B23In particular, positioning of the N-2-(benzyloxy)ethyl side chain on the β-residue was found to increase conformational order in protic solvents. This observation is vital for understanding the conformational preference of these novel systems and how this can be directed by the chemical structure. This work has significantly furthered efforts towards the engineering of compact ordered secondary structures of α,β-peptoids.
The authors acknowledge the Medway School of Pharmacy for the PhD scholarship for Ms De Santis.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.