Accurate detection and classification of materials, such as diseased tissues or illicit substances, is critical and misclassification can sometimes have life threatening consequences. Infrared (IR) absorption spectroscopy has been widely adopted as a simple but powerful characterisation tool, effectively producing a fingerprint of the samples’ molecular composition and aiding classification. IR absorption spectroscopy is quantitative and highly sensitive, but some measurement configurations, particularly in microspectroscopy1, can suffer from optical artefacts2,3. Misleading results can be obtained if changes in the IR absorption spectrum are not directly associated with chemical changes in the sample. Whether caused by the optical arrangement of the measurement or something intrinsic to the sample morphology itself, it is important to understand, and ideally correct for, any source of variance in the spectrum which is not due to the molecular composition. In this work a severe artefact is addressed which is present for samples on IR reflective slides due to the drop in IR intensity near a metallic surface.
There is a linear relationship between the IR light absorbed and the amount of absorbing material or chemical concentration in the sampled volume, which is expressed in the well known Beer-Lambert law. Molecular composition can, for example, help determine whether a histological tissue sample is healthy or diseased. In order to correctly and consistently classify samples it is crucial that the quantitative accuracy of the IR absorption spectra is maintained in all measurement configurations, especially as there is no standard measurement protocol.
The most common modes of measuring IR absorption spectra with a microscope are transmission and transflection. In transmission, the quantitative nature of IR spectroscopy is maintained, samples are mounted on a transparent window and the amount of light that passes through the sample and window is measured. In the transflection geometry, samples are mounted on reflective substrates, the IR light passes through the sample, is reflected off the substrate and passes back out through the sample, doubling the absorption path length. This configuration is popular in the biomedical sciences because it produces the strongest absorption signal for thin transparent samples such as cytology or histology specimens, it works over a broad spectral range and the substrates are relatively inexpensive.
In this work, it is shown that despite its advantages, the transflection measurement geometry induces complex optical effects which destroy the quantitative linear relationship between IR absorption and sample composition in a way which varies strongly with the IR wavelength.
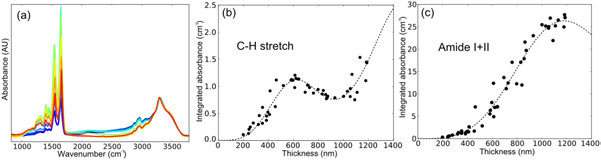 |
| Figure 1: Deviation of transflection IR absorbance data from the Beer-Lambert law, (a) Spectra of different thickness BSA films normalised to the N-H stretching band at 3300cm-1, (b) Integrated C-H stretch absorbance vs. thickness, (c) Integrated amide I+II absorbance vs. thickness. The dashed lines are the predictions of the model described in the text ( see equation). |
The first investigation used a standard, homogenous material, (Bovine Serum Albumin, BSA) with a well characterised IR absorption spectrum, to determine the relationship between absorbance and sample thickness. Measurement of several samples of BSA gel with different thicknesses in transmission showed the material was indeed homogeneous and that the transmission configuration maintains the Beer-Lambert relationship. In the transflection geometry, however, clear non-linear behaviour is observed. Typical transflection spectra for BSA gels of thickness 200-1200 nm are shown in Fig. 1(a) with absorbance normalised to the N-H stretching peak at 3300 cm-1. If the absorbance was linear with thickness in transflection these spectra would all be identical. The non-linearity is illustrated by integrating two spectral regions: the C-H stretching band (Fig. 1 (b), 2830-3010 cm-1); and the amide I+II bands (Fig. 1 (c), 1480-1760 cm-1), and plotting them against the sample thickness determined by atomic force microscopy (AFM). The non-linearity of absorbance with thickness is caused by heterogeneity in the IR intensity at the reflective surface. When light is reflected from a perfect conductor, the reflected wave undergoes a phase shift of ~180°, causing the incident and reflected light to interfere with each other. This interference creates a sinusoidal standing-wave in the light intensity which drops close to zero at the reflective surface4. The spacing between intensity maxima and minima depends on the refractive index of the sample and the wavelength of the light. The dashed lines in Fig. 1 (b) and (c) show the predicted variation of the absorbance with sample thickness based on a new phenomenological model of the optical electric-field standing wave (EFSW):
![]()
where A is the absorbance of light of wavelength λ by a sample of thickness l and refractive index n in the presence of the EFSW, g is a scaling parameter for the optical geometry, a0 is a spectrum of scaling coefficients for the absorbance - proportional to the true absorption coefficients, z is the distance from the reflective surface and R is a positive valued interference coefficient at each wavelength which is strongly dependent on the reflectivity of the sample/air interface. The integral term describes how much of the sinusoidal EFSW interacts with the sample and the term in the square brackets describes optical interference effects due to internal reflections.
Having used the BSA standard material to quantify the relationship between absorbance and thickness for a homogenous sample, the results were compared with a heterogeneous biological material, of the kind commonly studied by IR microscopy.
 |
| Figure 2: Comparison of IR images of six MCF-7 cells, three measured in transmission and three in transflection, (a) Visible microscope images, (b) IR absorption images, (c) AFM topography images, (d,e) Integrated C-H stretch and Amide I+II absorbance vs. thickness in transmission, (f,g) Integrated C-H stretch and Amide I+II absorbance vs. thickness in transflection. The symbols in (d)-(g) represent different cells. |
Breast adenocarcinoma cells, MCF-7, were grown on IR transmission and transflection substrates (Fig. 2 (a)) and analysed using an IR microscope equipped with a Focal Plane Array (FPA) imaging detector. The FPA has 64×64 pixels for simultaneous collection of 4096 IR spectra. The IR absorbance image of six cells was measured, three in transmission and three in transflection (Fig. 2 (b)). AFM topography images (Fig. 2 (c)) were measured to determine the thickness of the cells at each point in the FPA images. The C-H stretch and amide I+II bands of the IR spectra were again integrated and plotted against the sample thickness (Fig. 2(d-g)) . The integrated absorbance values were segregated into 100 nm thickness bins and averaged for plotting. Sample regions over 1 μm thick were rejected from the analysis because the spectra were few in number and corresponded to small, localised regions below the spatial resolution of the IR measurement. Integrated absorbance vs. thickness plots for the cells are shown at the bottom of Fig. 2. The transmission data (Fig. 2(d) and (e)), shows a linear relationship between the sample thickness and absorbance, i.e. it obeys the Beer-Lambert law, demonstrating also that there is a negligible change in component concentration in different cell regions. The cell transflection data (Fig.2 (f) and (g)) shows a very similar trend to the BSA standard transflection data (Fig. 1 (b) and (c)), especially the almost quadratic relationship between the absorbance and thickness below 600 nm.
The results of the full analysis of these six cells confirm that the EFSW artefact has a profound effect on the transflection spectra of such materials, and that the main spectral variations can be related to the sample thickness rather than any chemical differences. Understanding to what extent the EFSW artefact has affected the conclusions of previous investigations using the transflection geometry (of which there are many) and whether it can be corrected is the focus of ongoing work.
Electric field standing wave artefacts in FTIR micro-spectroscopy of biological materials, Filik, J., Frogley, M. D., Pijanka, J. K., Wehbe, K., and Cinque, G., Analyst. 137(4), 853 (2012)
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.