Structural insights into formation of an active signalling complex between Rac and phospholipase C-γ2
Oct 6, 2010
Oct 6, 2010
Prof Lawrence Pearl, Institute of Cancer Research
Rho family GTPases are important cellular switches and control a number of physiological functions. Understanding the molecular basis of interaction of these GTPases with their effectors is crucial in understanding their functions in the cell. Here we present the crystal structure of he complex of Rac2 bound to the split pleckstrin homology (spPH) domain of phospholipase C-γ2 (PLCγ2). Based on this structure, we illustrate distinct requirements for PLCγ2 activation by Rac and EGF and generate Rac effector mutants that specifically block activation of PLCγ2, but not the related PLCβ2 isoform. Furthermore, in addition to the complex, we report the crystal structures of free spPH and Rac2 bound to GDP and TPγS. These structures illustrate a mechanism of conformational switches that accompany formation of signaling active complexes and highlight the role of effector binding as a common feature of Rac and Cdc42 interactions with a variety of effectors.
We have previously shown that, of the two PLCγ isoforms, only PLCγ2 was directly activated by Rac. We also found that an unusual pleckstrin homology domain, the split PH (spPH) domain, is both necessary and sufficient to effect activation of PLCγ2 by Rac. As these experiments established the spPH domain as the site of interaction with Rac, our further studies aimed to understand the structural basis of this interaction using X-ray crystallography. Data for this project was collected on the MX beamlines I02 and I03.
To identify suitable protein variants for structural analysis of the complex, we tested spPH domain proteins with different point mutations that have affinities for GTP-loaded Rac2G12V similar to those of wild-type spPH. We were able to cocrystallise spPHY495F with Rac2G12V bound to GTPγS and determine the structure of the complex at 2.3 Å resolution. The interaction surface involves predominantly residues from the β5-strand and α-helix of the spPH domain, and residues present in both the switch I (amino acids 27 to 40) and switch II (amino acids 56 to 71) regions of Rac2. The total surface area buried at the interaction interface is approximately 900 Å2. The interaction is predominantly hydrophobic, with Phe37 and Val36 from Rac2 forming a two-pronged plug fitting into a double hydrophobic cleft on the surface of the spPH domain. Rac2 residues Ile33, Trp56, Tyr64, Leu67, and Leu70 intercalate with spPH residues Phe872, Val893, Leu896, Phe897, and Phe900 to extend this hydrophobic pocket. The spPH residue Lys862 makes multiple interfacial contacts with Rac2.
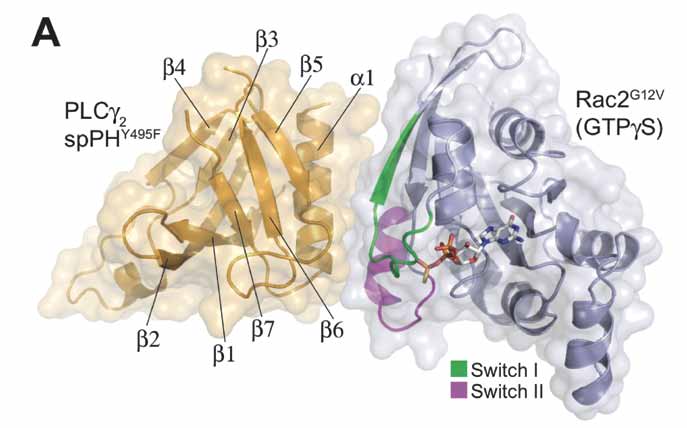 | Fig A: Overview of the Rac2G12V (GTPγS)/PLCγ2spPH complex structure. SpPH domain in orange and Rac2 in blue. The Rac2 switch I region is shown in green and the switch II in magenta. |
As we obtained structures of Rac2G12V(GDP), Rac2G12V(GTPγS), and spPHY495F as the free proteins, as well as in the complex, it was possible to identify conformational changes that accompany complex formation. The crystal structure of the free form of the PLCγ2 spPH contained 12 subunits in the asymmetric unit. We observed no significant difference in the conformation of the Cα backbone chain between the free and complexed forms. Interestingly though, the different subunits within the asymmetric unit of the free PLCγ2spPH show some heterogeneity, particularly with respect to the side chain orientation of Phe897, the key residue involved in Rac binding. In half the subunits, this orientation was the same as that in the complex with Rac2, which permits insertion of Rac2 residue Phe37. However, in the other molecules, the side chain of spPH residue Phe897 is rotated by 90°, blocking the hydrophobic socket. This suggests that a “soft switch” could exist on the surface of the spPH that opens or closes the hydrophobic cleft that ultimately accommodates the Rac switch I residue, Phe37.
| Fig B: Ribbon diagram of superimposed Rac2(GDP) (purple), Rac2 (GTPγS) free form (yellow) and the Rac2 (GTPγS) from the Rac2G12V (GTPγS)/PLCγ2spPH complex (blue). The two switch regions have been boxed. |  |
Comparison of the Rac2 structures shows some differences in the orientation of the Cα backbone of the switch II region and highlights an obvious change in the switch I effector loop. The GDP-bound and GTPγS-bound free forms of Rac superimpose well, but upon binding the spPH domain, the Rac switch regions are clearly repositioned. Strikingly, the positioning of the side chain of the key effector-interacting residue Phe37 is shifted upon binding, whereas the nature of the bound nucleotide in the free form of Rac does not influence the position of this residue. Effector binding also influences the Rac amino acid residues that coordinate the γ-phosphate on GTPγS. In stark contrast to the situation with crystal structures of several members of Ras family bound to GTP-analogs, where the γ-phosphate is coordinated by Thr35 and Gly60 when the protein is free or bound to effectors, in the case of Rac2, this coordination is seen only in the complex with spPH. The absence of this interaction in free Rac2(GTPγS) would imply that the structures of GDPand GTPγS -bound Rac2 should be similar. Indeed, the comparison with Rac2-GDP shows that both Thr35 and Gly60 are similarly positioned. Although coordination of the γ-phosphate by Thr35 is facilitated by changes in conformation of switch I in the Rac2/spPH complex, the direct interaction with Gly60 from switch II results from a movement of the GTPγS γ-phosphate that is shifted to the vicinity of Gly60.
Together, these data suggest that the signaling-active conformation of Rac2(GTP), resembling that of noncomplexed Ras (GTP), is greatly stabilised through interaction with its effector, PLCγ2. An important feature of its mechanism is likely to be the formation of a hydrophobic pocket that buries Phe37 and Val36 of the switch I region of Rac. This, in turn, reverses orientation of the key residues within the switch I region so that the coordination of the γ-phosphate is achieved. A similar mechanism is likely to underlie formation of a PLCγ2/Rac complex and complexes formed by Rac/Cdc42 proteins and several other effectors. As discussed further below, these structural insights provide a basis to consider more generally dynamic conformational states of small GTPases and the implications for their interaction with and specific recognition of effector proteins.
Principal Publications and Authors
T.D. Bunney, O. Opaleye, S.M. Roe, P. Vatter, R.W. Baxendale, C. Walliser, K.L. Everett, M.B. Josephs, C. Christow, F.Rodrigues-Lima, P. Gierschik, L.H. Pearl and M. Katan, Structural Insights into Formation of an Active Signaling Complex between Rac and Phospholipase C-γ2, Molecular Cell, 34, 223-233 (2009).
Funding Acknowledgement
Cancer Research UK (CRUK), the Deutsche Forschungsgemeinschaft (DFG), the Association pour la Recherche sur le Cancer and Association Française contre les Myopathies.
Research carried out at Diamond on I02 and I03, and at the ESRF on ID29.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.