Growing wires for a nanoscale circuit board
Sep 29, 2010
Sep 29, 2010
Electronic devices are getting smaller all the time, and the challenge is to make the individual components as small as possible without affecting how they function. This includes the need for nanometre scale wires to be placed on electrically insulating substrates. We used a technique called metal vapour deposition to grow palladium nanostructures on a dielectric support. We then used Scanning Tunnelling Microscopy (STM) to identify two distinct structures: roughly hexagonal islands and long thin wires just a few nanometres wide. Although STM gave us a good idea of the shapes of the nanostructures, it does not provide any information on their chemical make-up. We turned to the Nanoscience beamline at Diamond (I06). Using a technique called Low Energy Electron Microscopy we were able to identify the same nanoparticles after growing them at Diamond. We then used X-ray Photoemission Electron Microscopy (XPEEM) to monitor the chemical state of individual nanoparticles on a particle by particle basis. By scanning the kinetic energy and creating a movie, we determined that the nanoparticles were composed of metallic Pd, identifying a route for growing metallic nanowires on a dielectric substrate.
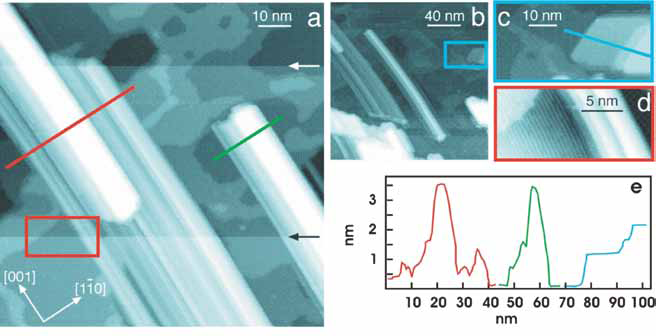 |
| Figure 1: STM images of TiO2(110) after depositing Pd at 900 K. a (100×100 nm2). b (200×200 nm2). Pseudohexagonal islands (blue box, expanded in c (~55×27 nm2)) are observed in addition to rod like structures. The red box in a highlights the area of image d (~20×9 nm2), in which atomic Ti rows can still be imaged. Arrows indicate tip changes during scanning. Line profiles measured over the nanoparticles are shown in e ; the line colours correspond to those used in the images |
Directed self-assembling nanostructures offer considerable potential for nanoscale electronics. A crucial component of these electronic devices will be quasi-1D wires supported on a dielectric support. In this study we have used the structurally sensitive technique of scanning tunneling microscopy (STM) at UCL and chemical sensitive spectro-microscopy [1] to investigate the growth of metallic nanowires supported on a model dielectric substrate, namely Pd on TiO2(110)-1×1. Fig. 1 shows STM images; Fig. 1a shows two nanowires separated by ~15 nm. Fig. 1b contains two types of nanostructures: nanowires and large pseudo-hexagonal islands. The image of the latter is expanded in Fig. 1c. Between these nanostructures, the TiO2 substrate can be seen with clear terraces and steps. Furthermore, in high resolution images, such as in Fig. 1d, atomic scale rows with a periodicity of 0.65 nm can clearly be seen running in the [001] direction. These rows are characteristic of the TiO2(110)-1×1 surface [2] and indicate that the nanowires align in the [001] direction. The isolated nanowire in the centre of Fig.1b is ~170 nm long and ~4 nm high, while the flat top of the nanowire on the left of Fig. 1b indicates a width of about 5 nm. The apparent uniform profile of the differing nanowires is a convolution of the surface features with the tip morphology. The second type of Pd nanostructure observed (Fig. 1c) consists of flattopped pseudo-hexagonal islands, similar to those previously observed on TiO2(110)- 1x2. [3]
Initial characterisation employed Low Energy Electron Microscopy (images not shown), which confirmed the presence of both nanowires and islands. The apparent width of the islands varied between ~80- 160 nm. Nanowires have a consistent apparent width of 120 ± 20 nm, and extend up to ~1 µm in length. The apparent width of features will be dominated by instrumental broadening. The actual width of features is expected to be in-line with that observed with STM. XPEEM images were recorded at a single photon energy, collecting emitted photoelectrons with a narrow energy dispersion (0.6 eV).
 |
| Figure 2: XPEEM images of TiO2(110) after depositing 0.4 ML Pd at 900 K. (a-d) XPEEM images recorded at ~13 µm field of view for different binding energies (see text for details). (e) Pd 3d µXPS acquired from Pd at hυ = 640 eV (red curve) with Doniac-Sunjic function fit (grey line). Also shown is the spectroscopy of 10 Pd dot-like nanoparticles (blue squares) and nanowires (green circles). Blue and green circled areas in frames a-d show the regions sampled to generate the spectra. Dashed lines correspond to the binding energies used for frames a-d. |
This energy-filtered mode allows the chemical state of individual nanostructures to be monitored through the chemical shift of photoemission features. By scanning the kinetic energy, a sequence of images can be collected that is analogous to X-ray photoelectron spectroscopy (XPS), but which is also spatially resolved. In Fig. 2a-d, four images are shown recorded over the Pd 3d3/2 and 3d5/2 core levels. The kinetic energy of photoelectrons was scanned over 8 eV in 0.2 eV steps, corresponding to binding energies (EB) of 342.1 to 334.1 eV.
The four images displayed in Fig. 2a-d are individual frames that demonstrate the essence of the XPS spectral information contained in the movie. Frame 2a is recorded at EB = 342.1 eV, just above the Pd 3d3/2 core level. Frame 2b is located exactly on the Pd 3d3/2 core level at EB = 340.1 eV. Frame 2c is recorded at EB = 337.1 eV and lies between Pd 3d3/2 and 3d5/2. Finally, frame 2d is on the Pd 3d5/2 core level at EB = 334.7 eV. Fig. 2e shows three XPS spectra recorded using two different measurement modes. The first mode (red spectrum, Fig. 2e) is performed by imaging the dispersive plane of the energy analyzer, sampling electrons from the entire field of view, and is known as micro-XPS (µXPS) due to the micrometer-scale sampling areas used. The second method (green and blue spectra, Fig. 2e) is built up pixel-by-pixel from the spectral- XPEEM image sequence, selecting the sampling area over individual features.[4] The photon energy was selected to generate photoelectrons of ~70 eV KE which enhances the surface sensitivity of the measurements. The red curve in Fig. 2e is a micro-XPS (mXPS) spectrum of the Pd 3d core levels recorded over the entire 20 µm field of view, hence integrating signal from both the substrate and the nanoparticles. The Pd 3d core levels display asymmetry and Pd 3d5/2 was measured at a binding energy of 334.8 ± 0.5 eV, which corresponds to the lower end of values for metal Pd reported in the literature, 335.2 ± 0.4 eV. The blue and green curves (Fig. 2e) are averaged spectra recorded from 10 dot-like nanoparticles and 2 nanowires, respectively. The lower signal to noise compared to the µXPS spectrum (red) is due to the much smaller areas sampled. The total areas sampled for the dotlike structures and the nanowires are ~400 and ~2000 times smaller than for the red spectrum, respectively. The key result is that peaks for the nanoparticles correspond to those of the mXPS spectrum, the latter being consistent with metallic Pd. The dominating role in the formation of elongated islands, which have an aspect ratio that is unparalleled, is probably the anisotropic lattice mismatch of the lowest energy plane termination of Pd, Pd(111) with TiO2(110).
References
[1] A. Locatelli, E.J. Bauer, Phys.: Condens. Matter, 20, 093002 (2008).
[2] O. Bikondoa, C.L. Pang, R. Ithnin, et al., Nat. Mater. 5, 189 (2006).
[3] R.A. Bennett, C.L. Pang, N. Perkins, et al., Phys. Chem. B, 106, 4688 (2002).
[4] A. Locatelli, L. Aballe, T.O. Mentes, M. Kiskinova, E. Bauer, Surf. Interface Anal, 38, 1554 (2006).
Principal Publications and Authors
Self assembled metallic nanowires on a dielectric support: Pd on rutile TiO2(110), D. S. Humphrey, G. Cabailh, C. L. Pang, C. A. Muryn, S. Cavill, H. Marchetto, A. Potenza, S. S. Dhesi, G. Thornton, Nano Letters, 9,155 (2009).
Funding Acknowledgement
Engineering and Physical Sciences Research Council in the Figure 1: STM images of TiO UK and the European Union.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.