Researchers into from Imperial College London and Harvard University have used Diamond to solve a 20-year-old puzzle about how HIV establishes itself in the human body. They have used the synchrotron to determine the structure of an enzyme called integrase, used by the HIV and similar viruses to copy their genetic information into the DNA of their hosts. Antiretroviral drugs have already been developed which work by blocking integrase, but the mechanism behind this was not fully understood until now. Understanding the mechanism could lead to better treatments for AIDS. This research has been published in the journal Nature.
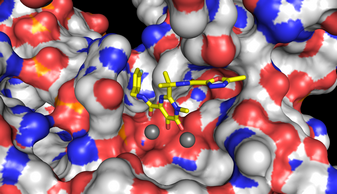 |
| Isentress (Raltegravir) bound to the active site of the integrase-DNA complex |
Retroviruses are a family of viruses that carry their genetic material as RNA (ribonucleic acid), rather than DNA. This is converted into DNA by enzymes they bring with them as they enter a host cell. For infection to be effective, the viral DNA has to be inserted into the genome of the cell, thus making it a permanent carrier of the viral genetic material. This process requires integrase, the essential viral enzyme that cuts a host cell chromosome and pastes in the viral DNA. HIV integrase is an important drug target, and disrupting its function has already produced potent antivirals. In addition, a greater understanding of retroviral integration will aid the development of gene therapy applications, which utilize modified retroviruses as vehicles for delivery of genetic material.
The challenge to understanding integrase lies with the difficulty of growing suitable crystals. For this study, researchers grew a crystal using a version of integrase borrowed from a little-known retrovirus called Prototype Foamy Virus (PFV), which is very similar to its HIV counterpart. Producing just one crystal of high enough quality to determine the three-dimensional structure took four years and over 40,000 trials.
Dr Peter Cherepanov, from the Department of Medicine at Imperial College London, led the study.
"It is a truly amazing story. When we started out, we knew that the project was very difficult, and that many tricks had already been tried and given up by others long ago. Therefore, we went back to square one and started by looking for a better model of HIV integrase, which could be more amenable for crystallization. Despite initially painstakingly slow progress and very many failed attempts, we did not give up and our effort was finally rewarded."Dr Peter Cherepanov, Imperial College London
The scientists found that retroviral integrase has quite a different structure to that which had been predicted based on earlier research. Researchers also soaked the crystals in solutions of clinical integrase inhibitors Isentress (also known as Raltegravir) and Elvitegravir and observed for the first time how these drugs bind to and inactivate the viral integration machinery. This exciting breakthrough means that researchers can begin to fully understand how existing drugs that inhibit integrase are working, how they might be improved, and how to stop HIV developing resistance to them.
Retroviral intasome assembly and inhibition of DNA strand transfer Stephen Hare, Saumya Shree Gupta, Eugene Valkov, Alan Engelman, Peter Cherepanov, Nature January 2010
DOI: 10.1038/nature08784
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.