Find out more about our ambitious upgrade project, delivering more brightness, more coherence, and greater speed of analysis to UK science. More about Diamond-II
![]()
Find out more about Diamond's response to virus research.
![]()

Research undertaken at Diamond has allowed scientists to create a super-enzyme that degrades plastic bottles six times faster than before.
The super-enzyme, derived from bacteria that lives on a diet of plastic, enables the full recycling of plastic bottles.
Plastic pollution is a global threat as plastics are rarely biodegradable and they can remain in the environment for centuries. One of the most abundant plastics that contributes hugely to this dire situation is poly(ethylene terephthalate) (PET).
PET is used largely in textiles, where it is commonly referred to as polyester, but it is also used as packaging for liquids and foodstuffs. PET’s excellent water-repellent properties led to it being the plastic of choice for soft drink bottles. However, the water resistance of PET means that they are highly resistant to natural biodegradation and can take hundreds of years to break down in the environment.
In 2018, researchers discovered that a unique bacterium (Ideonella sakaiensis 201-F6) was found feeding on waste from an industrial PET recycling facility. The bacterium had the amazing ability to degrade PET and use it to provide carbon for energy. Central to this ability was the production of a PET-digesting enzyme, known as PETase.
Diamond contributed to the original study of PETase using the world's unique Long-Wavelength Macromolecular Crystallography (MX) beamline, as well as other Diamond MX beamlines (I03 and I04) to successfully solve the structure of the bacterial enzyme. This created a potential low-energy solution to tackle plastic waste. The team engineered the natural PETase enzyme to be around 20 percent faster at breaking down PET.
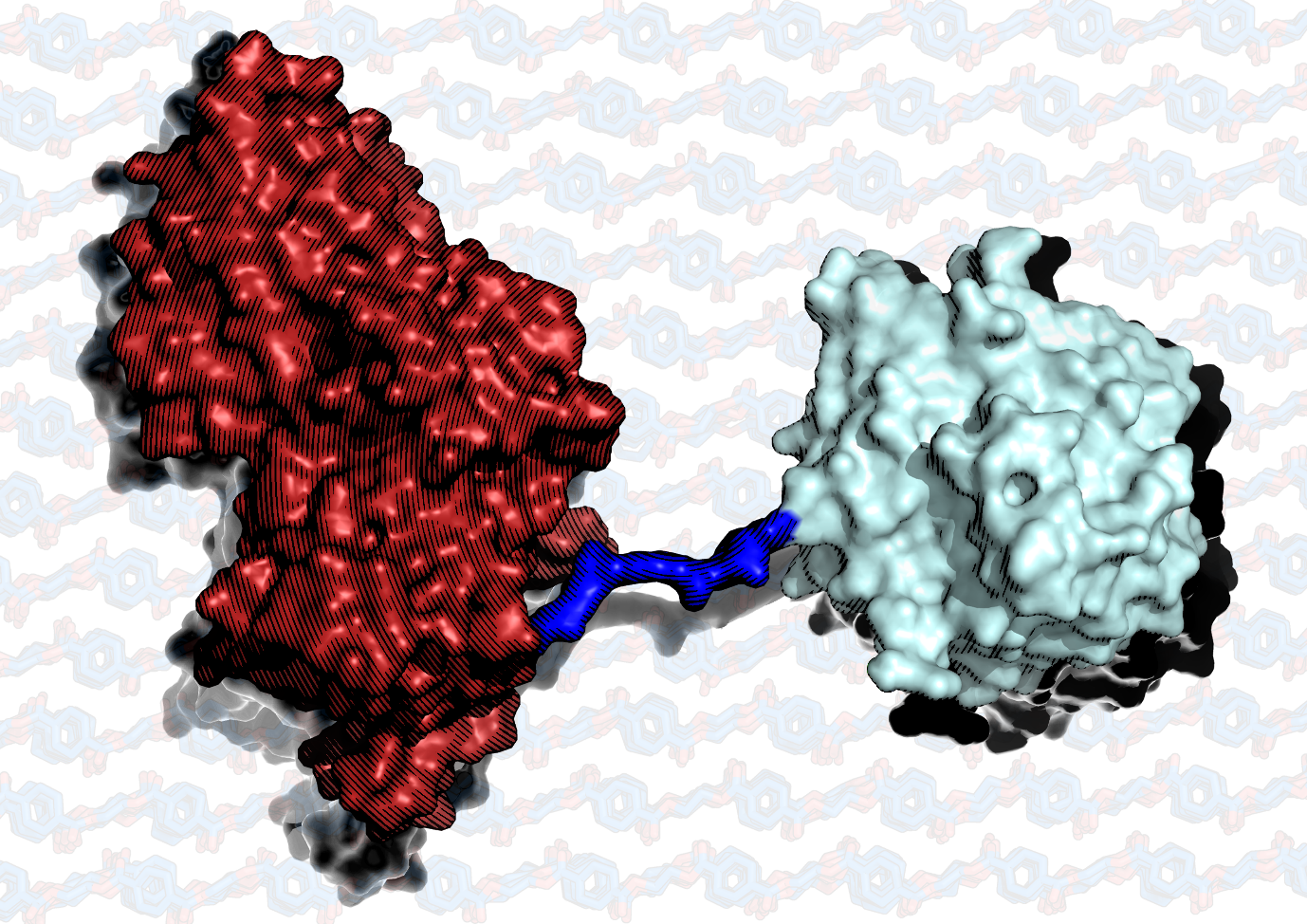
The same international team, co-led by Professor John McGeehan, Director of the Centre for Enzyme Innovation (CEI) at the University of Portsmouth, and Dr Gregg Beckham, Senior Research Fellow at the National Renewable Energy Laboratory (NREL) in the US, have combined PETase with MHETase to generate much bigger improvements. Simply mixing PETase with MHETase has doubled the speed of PET breakdown and engineered a connection between the two enzymes to create a ‘super-enzyme’.
Diamond X-rays were also used in this research and helped solve the 3D structure of MHETase. This gave researchers the molecular blueprints to begin engineering a faster enzyme system.
Professor John McGeehan, said:
Our first experiments showed that they did indeed work better together, so we decided to try to physically link them, like two Pac-men joined by a piece of string. It took a great deal of work on both sides of the Atlantic, but it was worth the effort – we were delighted to see that our new chimeric enzyme is up to three times faster than the naturally evolved separate enzymes, opening new avenues for further improvements.
The original PETase enzyme discovery was the first hope that a solution to the global plastic pollution problem might be within grasp. Combining it with a second enzyme, and finding together they work even faster, means another leap forward has been taken towards finding a solution to plastic waste.
All X-ray data collections were performed at one of Diamond’s macromolecular crystallography (MX) beamlines, I03.
John E. McGeehan et al. Characterization and engineering of a two-enzyme system for plastics depolymerization. Proceedings of the National Academy of Sciences Sep 2020, DOI: 10.1073/pnas.2006753117
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.