Scientists from the UK and USA have used the intense X-rays generated at Diamond Light Source to gain important insights into how target molecules in skin cancer treatment can become resistant to drugs designed to fight the cancer, leading to ineffective treatment. Their results are published today in
Nature.
In a developing embryo, a group of proteins known as the ‘hedgehog pathway’ send signals that help cells to grow in the right place and in the right way – these proteins can also control the growth of blood vessels and nerves. In adults, hedgehog pathway proteins are not usually active. But in some people, changes in a gene can switch them on, potentially leading to the growth of skin cancer and other types of cancer.
A type of anti-cancer drugs known as ‘hedgehog pathway blockers’ are designed to switch off these proteins so as to stop the growth of the cancer. However in some instances, the proteins can develop resistance to these drugs, so that the treatment no longer works.
But now, scientists from Oxford, Diamond, Stanford and St. Louis have revealed the structure of a complex molecule – known as the Smoothened protein receptor – which plays an important signalling role in the hedgehog pathway. It is this protein that the anti-cancer drug, ‘vismodegib’ acts on to switch off the hedgehog pathway and prevents cancer growth.
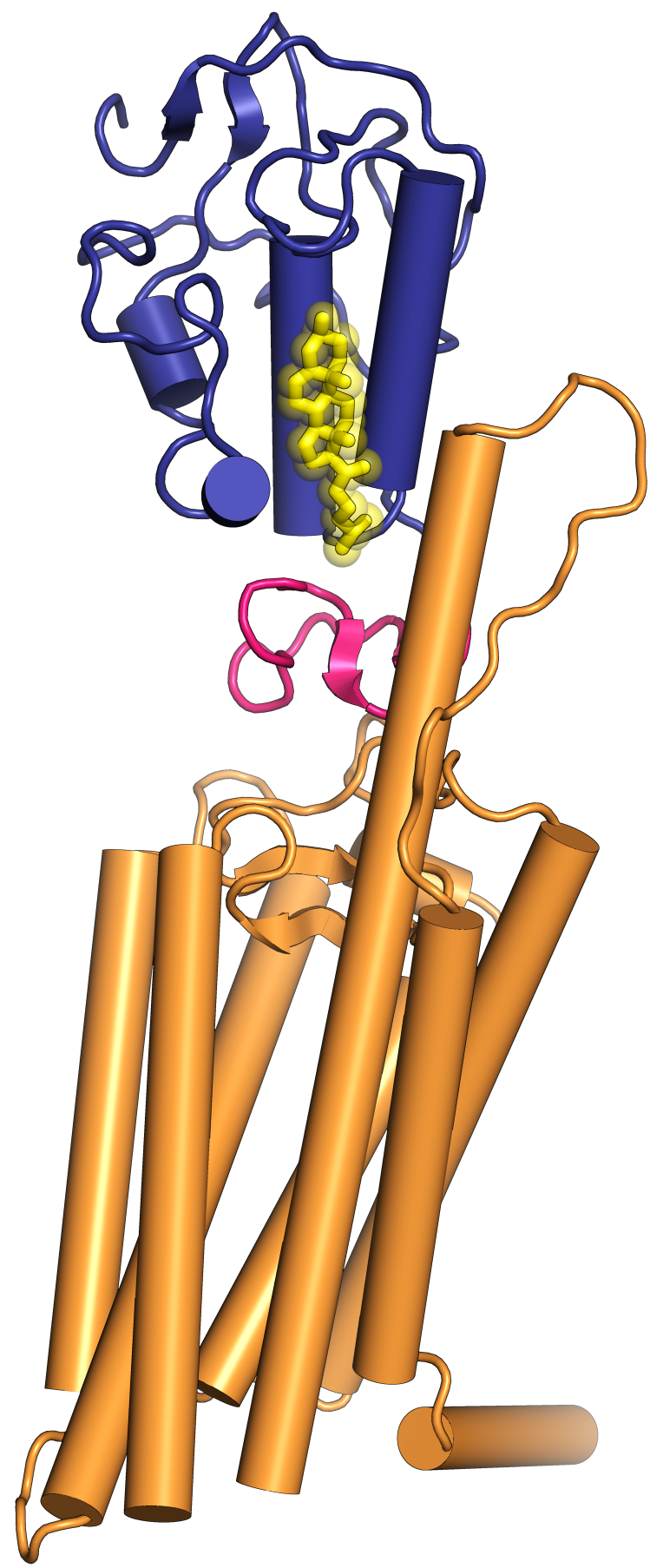
Structure of human Smoothened bound to cholesterol.
Blue: cysteine-rich ectodomain. Pink: juxtamembrane linker domain. Orange: hepta-helical transmembrane domain
Using cutting-edge scientific techniques, the group were also able to observe the way in which the molecule mutates so as to prevent the anti-cancer drug from binding with it – rendering the treatment useless.
Prof Christian Siebold, from the Division of Structural Biology (STRUBI) at the University of Oxford, explains the significance of the findings: “We are extremely excited by our results because the Smoothened molecule is a very unusual protein and it was technically very challenging to determine its crystal structure. Beyond this, we managed to see how the molecule changes and mutates to prevent the anti-cancer drug, vismodegib, from binding.”
This research was funded by Cancer Research UK, the US National Institutes of Health and the Wellcome Trust.
Dr Emma Smith, Cancer Research UK’s science information manager, said: “By revealing the precise shape of the molecule that vismodegib acts on, this research could lead to new ways of tackling skin cancers that become resistant to the drug. It could also stimulate new research into more effective treatments to destroy cancers driven by these faulty molecules.”
To gain their results, the team made use of complementary techniques at Diamond. The
Microfocus Crystallography beamline I24, enabled them to work with very small crystal samples of the Smoothened molecule. It took some 10 sessions of data collection to gather enough information to build up the entire structure of the molecule. When it came to seeing the structural changes of the Smoothened molecule, and the impact these changes had on the ability of small molecules to bind to it, Diamond’s
High Throughput Small Angle Scattering (SAXS) beamline B21 came into play.
Dr Robert Rambo, Principal Beamline Scientist for B21, said: “This research demonstrates how SAXS experiments can reveal differences in molecular states that cannot be observed by traditional X-ray crystallography. This research shows SAXS can be a powerful drug discovery tool for membrane proteins and if we can grow our knowledge of structural changes in membrane proteins such as this one, we can advance new drugs that can potentially remain effective despite these changes.”

Dr Robert Rambo