Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Related publication: Hollingshead S., Jongerius I., Exley R. M., Johnson S., Lea S. M. & Tang C. M. Structure-based design of chimeric antigens for multivalent protein vaccines. Nat. Commun. 9, 1051 (2018). DOI: 10.1038/s41467-018-03146-7
Publication keywords: Meningococcal; Vaccine; Serogroup B; fHbp; PorA
A major World Health Organisation objective is to develop vaccines against pathogenic bacteria. However, vaccine development is often hindered by variation in the molecules at the surface of the bacteria, and the manufacturing challenges inherent to working with such molecules. Researchers conducted a proof-of-principle study to assess the feasibility of taking portions of intractable molecules and creating chimeras by splicing them into well-behaved molecules, with the ultimate goal of producing vaccines that raised immune responses against both types of molecule.
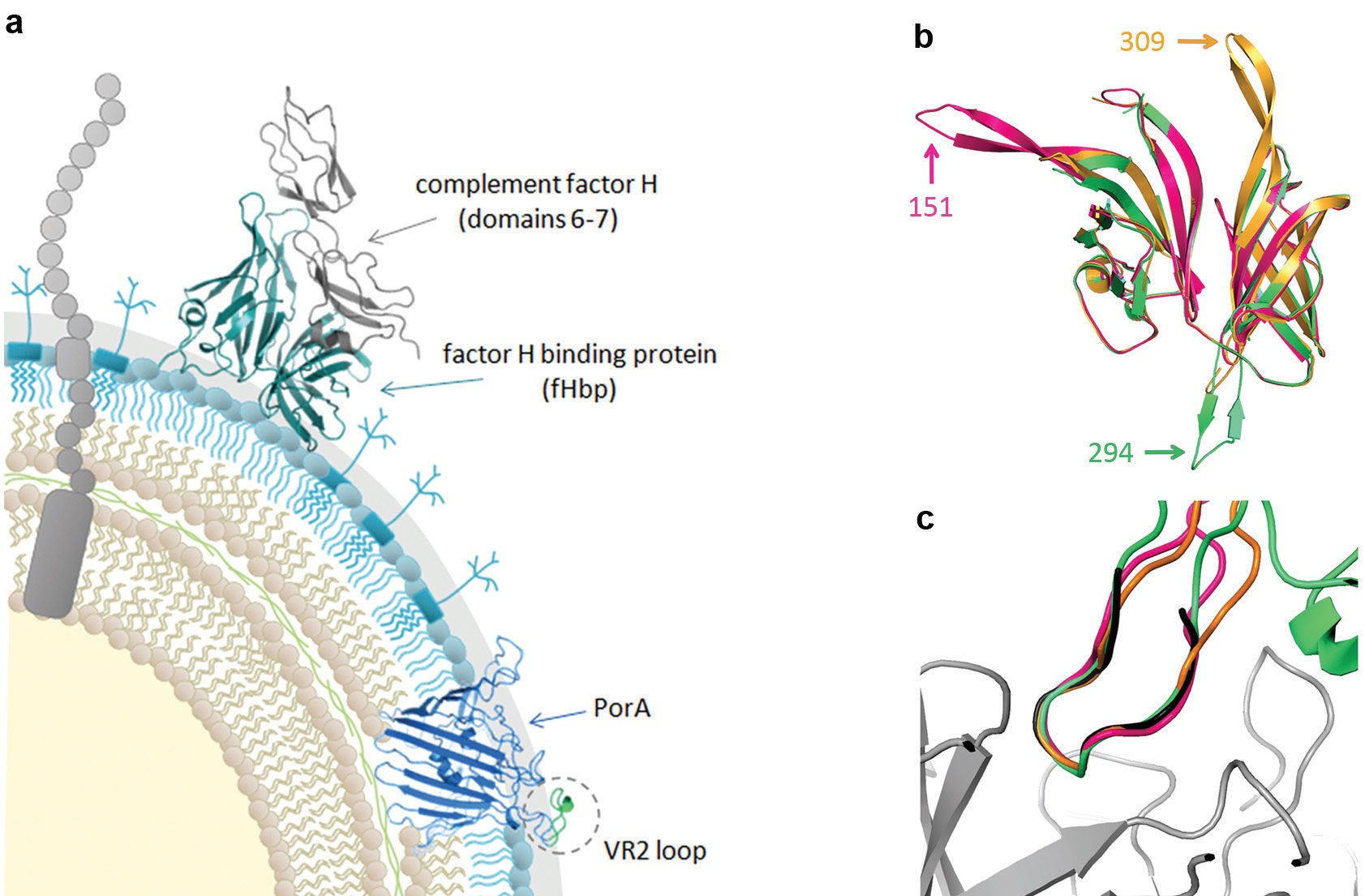
To combat the rise in multi-drug resistant bacteria, the World Health Organisation is advocating the development of vaccines against bacterial pathogens. Historically, vaccines were based on protein toxoids, polysaccharide capsules, or outer membrane vesicles. However, these approaches are not feasible for many pathogens, and immune evasion mechanisms, such as antigenic diversity, further limit the strategies available to develop successful vaccines. In addition, many vaccine candidates are integral membrane proteins which can present manufacturing problems, and issues such as generating immune responses against epitopes that are masked in the whole organism. To circumvent these issues, we used structure-based design to engineer chimeric antigens (ChAs), and developed a next generation vaccine for prevention of meningococcal disease.
The meningococcal ChAs produced for our study are composed of two major N. meningitidis antigens, factor H binding protein (fHbp), a surface exposed lipoprotein1, and PorA, an integral outer membrane porin2 (Fig. 1a). In the ChAs, fHbp is exploited as a molecular scaffold to display an immunogenic surface exposed loop from PorA, known as the variable region 2 (VR2, Fig. 1a). Both fHbp and the VR2 loop exhibit antigenic diversity, with fHbp peptides falling into three variant groups or two subfamilies depending on the classification system: V1 (subfamily B), V2, and V3 (both subfamily A). In general, immunisation with a particular fHbp induces cross-protection against strains that express an fHbp belonging to the same, but not a different, variant group, although there can be cross-protection between fHbp variant groups V2 and V3 (subfamily A). Unlike fHbp, the PorA VR2 offers limited or no cross-protection against strains expressing a PorA peptide with a different VR2. Therefore to optimise coverage of the vaccine, the fHbp and PorA VR2 peptides used must be carefully selected. This was achieved using the comprehensive epidemiology data available in the meningococcal genome library3.
Figure 2: Serum bactericidal assay titres. (a) α-PorA SBA titres generated using ChA/alum antisera and serogroup B N. meningitidis isolates with mismatched fHbp variants. (b) α-fHbp SBA titres generated using ChA/alum antisera and serogroup B N. meningitidis isolate H44/76 ΔporA.
References:
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.